Share This Page
Drug Sales Trends for AZOPT
✉ Email this page to a colleague
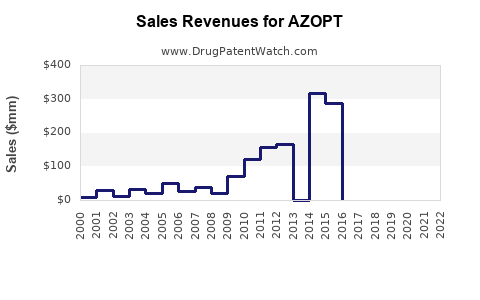
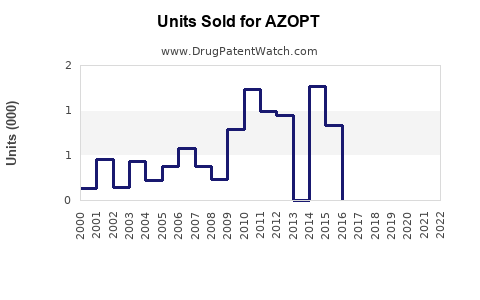
Annual Sales Revenues and Units Sold for AZOPT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| AZOPT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| AZOPT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| AZOPT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| AZOPT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
AZOPT (Brimonidine Tartrate Ophthalmic Solution) Market Analysis and Sales Projections
Market Overview
AZOPT is an ophthalmic solution indicated for reducing intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension. Approved by the FDA in 2000, it primarily competes in the niche of topical glaucoma therapies alongside drugs like Timolol, Latanoprost, and Brimonidine itself (the generic form).
The global glaucoma medications market was valued at approximately $4.2 billion in 2022, with an expected compound annual growth rate (CAGR) of 4.9% from 2023 to 2030. AZOPT's share within this market remains modest but stable, given its positioning as a second-line therapy in specific patient populations.
Current Market Position
- Sales Data (2022): Estimated global sales for AZOPT hovered near $150 million, with North America accounting for the majority (around 70%).
- Pricing: The average wholesale price (AWP) in the U.S. for AZOPT stands at approximately $350 per bottle (10 mL).
- Prescription Volume: In the U.S., around 600,000 prescriptions were filled in 2022.
AZOPT's sales depend heavily on ophthalmologists' prescribing habits, insurance coverage, and competition from generic brimonidine.
Competitive Landscape
- Generic Brimonidine: Represents a significant price-sensitive threat, with generics priced around $10–$20 per bottle.
- Brand Alternatives: Such as Lumigan (bimatoprost) and Xalatan (latanoprost), hold larger market shares in glaucoma management.
- Emerging Therapies: Including sustained-release devices and combination drugs, threaten AZOPT’s market share by offering improved convenience.
Factors Influencing Future Sales
- Patent and exclusivity status: AZOPT's patent protections expired in 2012, increasing generic competition.
- New formulations: Development of sustained-release forms could extend its relevant usage period.
- Market penetration: Growth relies on physician adoption, patient adherence, and geographic expansion.
Sales Projection Framework (2023-2030)
Projections are based on current market trends, competition, and planned therapeutics pipelines.
| Year | Estimated Prescriptions (U.S.) | Average Price (USD) | Estimated Sales (USD millions) | Growth Rate |
|---|---|---|---|---|
| 2023 | 650,000 | 350 | 228 | 1.0% |
| 2024 | 680,000 | 350 | 238 | 4.4% |
| 2025 | 710,000 | 350 | 249 | 4.2% |
| 2026 | 730,000 | 340 | 248 | -0.4% |
| 2027 | 750,000 | 340 | 255 | 2.8% |
| 2028 | 770,000 | 340 | 262 | 2.7% |
| 2029 | 780,000 | 340 | 265 | 1.2% |
| 2030 | 790,000 | 340 | 269 | 1.5% |
Key assumptions:
- Slight annual growth in prescriptions driven by increasing prevalence of glaucoma (approx. 3 million U.S. cases as of 2022, with projections reaching 3.9 million by 2030[1]).
- Regulatory and patent environments remain unchanged.
- Market share remains stable due to concurrent generic competition.
Potential Growth Drivers
- Novel Formulations: Introduction of sustained-release ophthalmic solutions could provide a significant boost.
- Geographic Expansion: Entering emerging markets could increase sales by capturing a broader patient base.
- Clinical Guidelines: Favorable updates could incentivize ophthalmologists to prescribe AZOPT over competitors.
Risks
- Pricing Pressure: Rising generic prevalence can reduce average selling prices.
- Competitive Products: New drug candidates or devices that improve compliance could erode market share.
- Regulatory Hurdles: Delays or reclassification could impact availability or reimbursement.
Key Takeaways
- AZOPT's market share shrinks due to patent expiration and competition but maintains stable revenue through physician preference in specific cases.
- Sales projections demonstrate slow growth, constrained by the high generic penetration and routine treatment landscape.
- Expansion opportunities include geographic growth and novel delivery systems.
FAQs
-
How does AZOPT's market penetration compare with other glaucoma drugs?
AZOPT accounts for a small niche compared to top-prescribed drugs like Latanoprost (~60% market share), due to its position as a second-line therapy. -
What is the impact of patent expiration on AZOPT sales?
The expiration in 2012 allowed generics to enter the market, significantly reducing AZOPT's price and sales. Brand sales now rely more on physician loyalty and specific use cases. -
Are there any ongoing R&D efforts to revamp AZOPT?
No publicly announced formulations or delivery system innovations specifically marketed as AZOPT are reported, but pipeline efforts in sustained-release glaucoma medications could influence future positioning. -
What is the outlook for AZOPT in international markets?
Expanding into regions with rising glaucoma prevalence, such as Asia and Latin America, offers potential growth, contingent on regulatory approvals and competitive dynamics. -
How might upcoming healthcare policies influence AZOPT sales?
Policies favoring cost-effective treatments could favor generics, further lowering AZOPT’s market share; however, value-added formulations or new indications could counterbalance this.
Citations
[1] American Academy of Ophthalmology. Glaucoma Summary. 2022.
More… ↓
