Share This Page
Drug Sales Trends for AVELOX
✉ Email this page to a colleague
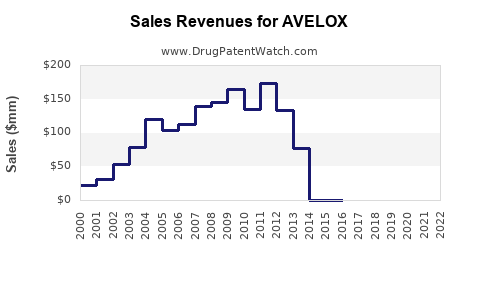
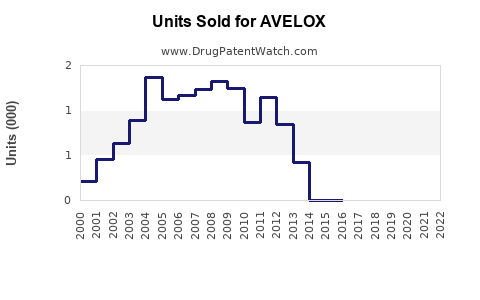
Annual Sales Revenues and Units Sold for AVELOX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| AVELOX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| AVELOX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| AVELOX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
AVELOX (Moxifloxacin) Market Analysis and Sales Projections
AVELOX, a fluoroquinolone antibiotic manufactured by Bayer AG, generated approximately $622 million in sales globally in 2022. The drug is primarily indicated for the treatment of bacterial infections, including acute bacterial sinusitis, acute exacerbations of chronic bronchitis, and community-acquired pneumonia. Market performance is influenced by the prevalence of bacterial infections, antibiotic resistance patterns, and competition from generic alternatives.
What is the current global market size for AVELOX?
The global market for AVELOX was approximately $622 million in 2022. This figure represents a decline from previous years, largely attributable to patent expirations and the subsequent introduction of generic moxifloxacin products. The United States remains the largest market for branded AVELOX, followed by key European countries and Japan. Sales have been impacted by a contraction in the overall fluoroquinolone market due to safety concerns and the emergence of broader-spectrum antibiotics.
What are the key indications for AVELOX?
AVELOX is approved for the treatment of a range of bacterial infections. These include:
- Acute Bacterial Sinusitis: A common indication, often treated in outpatient settings.
- Acute Exacerbations of Chronic Bronchitis: Targeting bacterial superinfections in patients with pre-existing lung conditions.
- Community-Acquired Pneumonia: A more serious indication where rapid and effective treatment is critical.
- Complicated Skin and Skin Structure Infections: Used when other standard treatments are not suitable.
- Complicated Intra-abdominal Infections: Often in combination with other antibacterial agents.
The efficacy of AVELOX against Gram-positive and Gram-negative bacteria, including some anaerobic organisms, underpins its utility across these indications.
Who are the main competitors to AVELOX?
Competition for AVELOX exists from both branded and generic pharmaceuticals. The primary competitive landscape includes:
- Other Fluoroquinolones: While AVELOX is a later-generation fluoroquinolone, it competes with other drugs in this class, such as levofloxacin (Levaquin, now generic) and ciprofloxacin (Cipro, largely generic). However, the use of fluoroquinolones has seen a decline due to safety warnings from regulatory bodies.
- Macrolides: Antibiotics like azithromycin (Zithromax) and clarithromycin are frequently used for respiratory tract infections, a key indication for AVELOX.
- Beta-Lactams: Penicillins and cephalosporins remain standard treatments for many bacterial infections and offer a lower risk profile compared to fluoroquinolones for certain indications.
- Newer Antibacterial Agents: Development of novel antibiotics targeting resistant bacteria presents a long-term competitive threat, though current market penetration for these new agents remains limited.
- Generic Moxifloxacin: Since the expiry of key patents, numerous generic versions of moxifloxacin have entered the market. These generics offer a significantly lower price point, eroding the market share and revenue of branded AVELOX. The market share for generic moxifloxacin is estimated to be over 70% in major markets.
What is the patent status and genericization impact on AVELOX?
Bayer's primary patents for AVELOX (moxifloxacin hydrochloride) have expired in major markets. Key patent expiries occurred around 2015-2017. This patent expiry has led to:
- Entry of Generic Manufacturers: Numerous pharmaceutical companies have launched generic moxifloxacin products globally.
- Significant Price Erosion: Generic versions are priced considerably lower than branded AVELOX, leading to a substantial decrease in average selling price.
- Market Share Transfer: A significant portion of the moxifloxacin market has shifted from branded AVELOX to generic alternatives. This has directly reduced AVELOX's revenue.
- Continued Sales of Branded Product: Despite generic competition, branded AVELOX continues to hold a share of the market, particularly in regions or for physicians who prioritize the brand or have specific contractual agreements.
The total market for moxifloxacin, including both branded and generic, remains substantial, but the revenue attributable to branded AVELOX has declined sharply.
What are the regulatory considerations and safety concerns impacting AVELOX?
AVELOX, like other fluoroquinolones, is subject to significant regulatory scrutiny and has associated safety concerns that impact its prescribing patterns and market access.
- Black Box Warnings: The U.S. Food and Drug Administration (FDA) has issued black box warnings for fluoroquinolones, including moxifloxacin. These warnings highlight the risk of serious adverse reactions, including tendinitis and tendon rupture, peripheral neuropathy, central nervous system effects (e.g., seizures, hallucinations), and exacerbation of myasthenia gravis [1].
- FDA and EMA Guidance: Regulatory bodies have advised healthcare professionals to reserve fluoroquinolones for patients with no alternative treatment options for acute bacterial sinusitis, acute bacterial exacerbations of chronic bronchitis, and uncomplicated urinary tract infections due to these risks [2]. This restriction limits the use of AVELOX in certain common indications where it was previously a first-line therapy.
- Cardiovascular Risks: Studies have indicated potential cardiovascular risks, including QT interval prolongation, which can lead to potentially fatal arrhythmias. This has led to contraindications and precautions regarding use in patients with known QT prolongation, electrolyte imbalances, or those taking other QT-prolonging drugs [3].
- Post-Marketing Surveillance: Ongoing pharmacovigilance and post-marketing studies continue to monitor for adverse events, potentially leading to further label updates or prescribing restrictions.
These safety concerns and regulatory restrictions have contributed to a decreased market appetite for fluoroquinolones in general, indirectly affecting AVELOX sales by promoting the use of alternative antibiotic classes.
What are the sales projections for AVELOX?
Sales projections for branded AVELOX are subject to continued market erosion from generic competition and the persistent impact of safety warnings.
- 2023-2025 Projection: Branded AVELOX sales are projected to decline at a compound annual growth rate (CAGR) of approximately -8% to -12% over the next two to three years.
- 2023 Estimate: $550 million - $580 million
- 2024 Estimate: $480 million - $520 million
- 2025 Estimate: $420 million - $460 million
- Factors influencing projections:
- Generic Penetration: Continued growth of generic moxifloxacin will further pressure branded AVELOX volumes and pricing.
- Prescribing Guidelines: Adherence to updated regulatory guidance, which limits fluoroquinolone use, will restrict new prescriptions.
- Market Penetration of Alternatives: The increasing adoption of alternative antibiotic classes for common infections will reduce the demand for AVELOX.
- Geographic Variations: Developed markets with stricter regulatory oversight and a higher prevalence of generic substitution will experience steeper declines compared to some emerging markets where branded product may retain a larger share for longer.
- Bayer's Marketing Strategy: Any remaining promotional efforts by Bayer will focus on specific niches or regions where branded AVELOX can still command a premium or demonstrate superior value, although the scope for this is diminishing.
The overall moxifloxacin market (including generics) is expected to remain stable or experience modest growth due to the ongoing need for effective antibiotics, but the branded AVELOX segment will continue to contract.
What is the geographical distribution of AVELOX sales?
The sales of branded AVELOX are concentrated in key developed markets, reflecting historical market access, physician prescribing habits, and the presence of regulatory approvals.
- United States: Historically the largest market for AVELOX, accounting for approximately 40-45% of global branded sales. Prescribing patterns and market access remain significant, though generic penetration is high.
- Europe: Major European countries (Germany, UK, France, Italy, Spain) collectively represent another significant portion of sales, around 30-35%. Regulatory guidelines and national healthcare policies heavily influence prescription volumes.
- Japan: Represents a smaller but important market, typically accounting for 5-10% of sales.
- Rest of the World: Includes sales from other regions, such as Canada, Australia, and select emerging markets. This segment contributes the remaining 10-20% of global branded AVELOX revenue.
The trend globally is a gradual decrease in the proportion of sales derived from branded AVELOX, as generics gain traction across all regions.
What are the key strategic considerations for AVELOX stakeholders?
Stakeholders involved with AVELOX, primarily Bayer as the originator and generic manufacturers, face distinct strategic imperatives.
- Bayer AG (Originator):
- Lifecycle Management: Focus on retaining market share through brand loyalty, value-added services, and potentially exploring niche indications if further clinical data supports it.
- Geographic Focus: Prioritize regions or customer segments where branded product can still achieve premium pricing or where generic penetration is slower.
- Portfolio Optimization: Evaluate the continued investment in promoting AVELOX versus other products in Bayer's portfolio, given the declining revenue trajectory.
- Compliance and Safety: Ensure strict adherence to all regulatory and safety guidelines to mitigate any potential liabilities and maintain the drug's availability where it is still deemed appropriate.
- Generic Manufacturers:
- Cost Leadership: Optimize manufacturing and supply chain efficiencies to offer the lowest possible price for moxifloxacin.
- Market Access: Secure formulary placement and distribution agreements across key markets to maximize reach.
- Quality and Reliability: Maintain high-quality manufacturing standards to build trust with prescribers and pharmacists.
- Expansion: Explore opportunities in emerging markets where generic penetration may be less advanced and branded products still hold a stronger position.
For both groups, understanding the evolving antibiotic landscape, including the rise of antimicrobial resistance and the development of novel therapies, is critical for long-term strategy.
Key Takeaways
- Branded AVELOX sales were approximately $622 million in 2022, with a projected decline driven by patent expirations and generic competition.
- Key indications include acute bacterial sinusitis, exacerbations of chronic bronchitis, and community-acquired pneumonia.
- Competition comes from other fluoroquinolones, macrolides, beta-lactams, and notably, generic moxifloxacin, which holds over 70% market share.
- Regulatory concerns, including black box warnings for serious adverse events, have restricted AVELOX's use.
- Sales are projected to decline at a CAGR of -8% to -12% through 2025, with the United States and Europe being the largest geographic markets.
- Strategic imperatives for Bayer involve lifecycle management and focused marketing, while generic manufacturers prioritize cost leadership and market access.
Frequently Asked Questions
- Has AVELOX been withdrawn from the market in any major regions? AVELOX has not been formally withdrawn from the market globally. However, its use has been restricted in certain indications in key regions like the U.S. and Europe due to safety concerns.
- What is the primary reason for the decline in branded AVELOX sales? The primary reason is the expiration of key patents, which has allowed numerous generic manufacturers to introduce lower-cost moxifloxacin products, leading to significant price erosion and market share transfer.
- Are there new indications being explored for AVELOX? Bayer's focus has shifted away from significant new indication development for AVELOX, given its lifecycle stage and the competitive and regulatory landscape. Research is more likely to be in the area of combating antimicrobial resistance with newer agents.
- How do the safety concerns of AVELOX compare to other fluoroquinolones? AVELOX shares similar safety concerns, including tendon rupture and peripheral neuropathy, with other fluoroquinolones. Regulatory bodies have issued class-wide warnings and restrictions on the use of all fluoroquinolones due to these risks.
- What is the expected market share of generic moxifloxacin in the next five years? Generic moxifloxacin is expected to maintain and potentially increase its market share, likely exceeding 80% of the total moxifloxacin market within the next five years, further marginalizing branded AVELOX.
Citations
[1] U.S. Food and Drug Administration. (2018). FDA strengthens warnings on serious side effects of fluoroquinolone antibiotics. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-strengthens-warnings-serious-side-effects-fluoroquinolone-antibiotics
[2] European Medicines Agency. (2018). European Medicines Agency recommends restricting the use of fluoroquinolone antibiotics. Retrieved from https://www.ema.europa.eu/en/news/european-medicines-agency-recommends-restricting-use-fluoroquinolone-antibiotics
[3] FDA Drug Safety Communication. (2018). FDA reinforces warnings of disabling tendon problems with fluoroquinolone use; advises against use in certain patients. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-reinforces-warnings-disabling-tendon-problems-fluoroquinolone-use-advises-against-use-certain-patients
More… ↓
