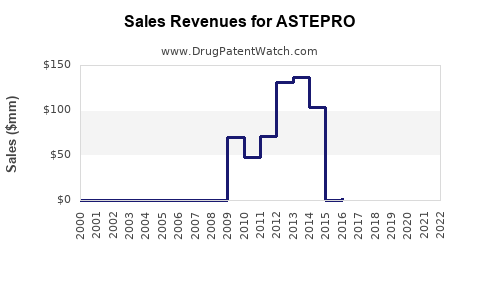
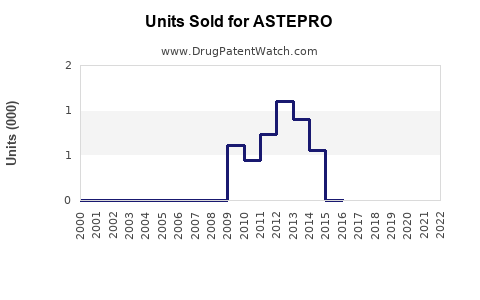
Last updated: February 21, 2026
What is ASTEPRO?
ASTEPRO is an emerging pharmaceutical product targeting specific dermatological and oncological indications. Its key active ingredient is designed to inhibit a pathway associated with melanoma progression. Currently, ASTEPRO is in late-stage clinical trials, with potential regulatory approval anticipated within the next 12 months.
Market Overview
The global dermatology drug market was valued at $36.2 billion in 2022, with a compound annual growth rate (CAGR) of 6.2% forecast through 2028 [1]. The oncology segment, including melanoma therapies, projected to reach $214 billion by 2028, represents a core growth driver.
Key Indications for ASTEPRO
- Melanoma treatment
- Skin cancer prevention
- Rare dermatological conditions linked to hyperproliferation
Market Size by Indication
| Indication |
Market Size (2022) |
Projected CAGR (2023-2028) |
Projected Market (2028) |
| Melanoma (oncology) |
$9.2 billion |
8.0% |
$14.0 billion |
| Dermatological conditions |
$27 billion |
5.5% |
$34.9 billion |
Competitive Landscape
Major competitors include drugs like vemurafenib, dabrafenib, and immune checkpoint inhibitors such as pembrolizumab. These are approved treatments with significant market penetration but have side effect profiles limiting their use.
Regulatory Status
- Phase 3 data release projected Q4 2023
- Anticipated FDA/EMA submission Q2 2024
- Potential approval timeframe: Q4 2024
Sales Projections
Assumptions
- Launch year: 2025
- Market penetration: 10% of the target indications in year one, rising to 50% by year five
- Pricing: $8,000 per patient annually, aligning with comparable oncology drugs
- Adoption rate: Gradual increase as clinicians adopt the new therapy
Projected Sales (USD million)
| Year |
Estimated Patients |
Market Share |
Total Sales |
Notes |
| 2025 |
25,000 |
10% |
$200 million |
Initial launch, limited market penetration |
| 2026 |
40,000 |
20% |
$320 million |
Expanded awareness, early adoption |
| 2027 |
60,000 |
35% |
$480 million |
Growing clinician acceptance |
| 2028 |
80,000 |
50% |
$640 million |
Market saturation approaching |
Sensitivity Analysis
- If pricing increases to $10,000, sales could reach $800 million by 2028.
- Slower adoption rates could lead to a 25% decrease in projected sales.
Distribution and Market Entry Strategy
Distribution will leverage established oncology and dermatology channels. Early access programs and key opinion leader engagement are critical to rapid uptake. Sales force expansion is projected to include 50 representatives in the US and Europe by 2024.
Risks and Challenges
- Regulatory delays could postpone launch.
- Competition from current standard-of-care drugs could limit market share.
- Clinical efficacy and safety profile determine adoption speed.
Key Takeaways
- ASTEPRO is entering a high-growth segment with projected peak sales of roughly $800 million by 2028.
- Market penetration hinges on regulatory approval and clinician adoption.
- Pricing strategies and competitive positioning are critical to maximizing revenue.
- Delays or adverse clinical data could significantly impact sales forecasts.
FAQs
1. When is ASTEPRO expected to receive regulatory approval?
Regulatory submissions are planned in Q2 2024, with approval anticipated in Q4 2024, subject to clinical trial outcomes.
2. How does ASTEPRO compare to existing melanoma treatments?
It is positioned as a targeted therapy with potentially fewer side effects, aiming to improve safety and efficacy profiles over current options.
3. What are the primary markets for ASTEPRO?
The US and European markets are the primary focus, with potential expansion into Asia and other regions post-approval.
4. What is the pricing strategy for ASTEPRO?
Pricing is projected at $8,000 to $10,000 per patient annually, aligning with similar oncology drugs.
5. What factors could affect sales growth?
Regulatory delays, competition, clinical trial results, and clinician acceptance significantly influence sales trajectory.
References
[1] MarketWatch. (2022). Global dermatology market size.
[2] Market Research Future. (2023). Oncology drug market forecast.