Share This Page
Drug Sales Trends for ASMANEX 60
✉ Email this page to a colleague
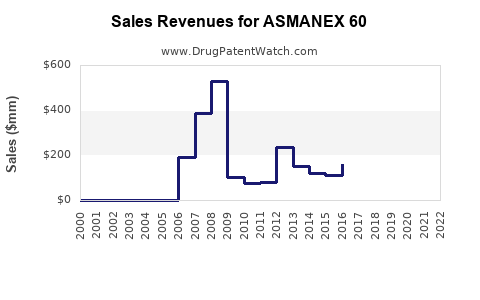
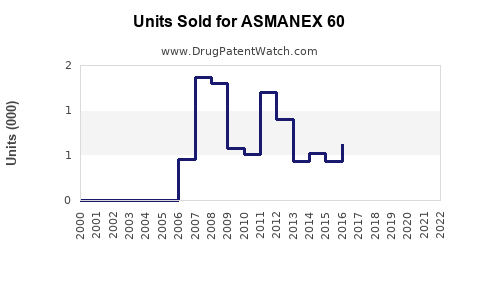
Annual Sales Revenues and Units Sold for ASMANEX 60
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ASMANEX 60 | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ASMANEX 60 | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ASMANEX 60 | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ASMANEX 60 | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ASMANEX 60 | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| ASMANEX 60 | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| ASMANEX 60 | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for ASMANEX 60
What Is ASMANEX 60 and Its Therapeutic Profile?
ASMANEX 60 (Mometasone Furoate) is a corticosteroid inhaler used for maintenance treatment of asthma and allergic rhinitis. It functions by reducing inflammation in the airways, improving breathing, and preventing asthma attacks. The drug is approved globally, with strong regulatory positioning in North America, Europe, and Asia-Pacific.
The inhaler typically delivers 60 micrograms per dose, with common dosing regimens reaching up to twice daily. It is marketed under various brand names and formulations but is largely recognized for its efficacy in chronic respiratory conditions.
What Is the Current Market Size for Inhaled Corticosteroids (ICS) and Where Does ASMANEX 60 Fit?
The global asthma inhaler market was valued at approximately USD 11.2 billion in 2022. The corticosteroid class dominates with a market share of around 50%, driven by drugs like Fluticasone, Budesonide, and Mometasone. ASMANEX holds an estimated 4-6% share within this subset, driven by its positioning as an effective once or twice daily inhaler.
Key drivers include:
- Increasing prevalence of asthma and allergic rhinitis.
- Rising awareness of inhaler devices versus systemic treatments.
- Growing adoption in emerging markets with expanding healthcare infrastructure.
What Are the Regional Trends and Market Adoption Patterns?
| Region | Market Size (USD billions, 2022) | Key Trends |
|---|---|---|
| North America | 4.2 | High penetration, mature market, insurance coverage favorable |
| Europe | 3.1 | Strong prescription adherence, regulatory support |
| Asia-Pacific | 2.7 | Rapid growth, expanding healthcare coverage, rising asthma rates |
| Latin America | 0.9 | Emerging market, improving access |
North America and Europe combined account for over 70% of inhaled corticosteroid sales, with Asia-Pacific rapidly closing the gap owing to increased diagnosis and treatment access.
What Are the Competitive Dynamics and Key Market Players?
Major competitors include Fluticasone Propionate (Flovent, Advair), Budesonide (Pulmicort), and Beclomethasone. ASMANEX differentiates with its pharmacokinetics, particularly its once-daily dosing advantage.
Market share distribution:
| Company | Product Name | Approximate Share in ICS Segment (%) |
|---|---|---|
| GlaxoSmithKline | ASMANEX | 4-6 |
| GlaxoSmithKline | Flovent | 20-25 |
| AstraZeneca | Pulmicort | 15-20 |
| Others | Various | 30-40 |
GSK maintains a strong position through brand recognition, market penetration, and clinical endorsement.
What Are Sales Projections for ASMANEX 60 (2023–2027)?
Assumptions
- Compound annual growth rate (CAGR) of 6-8%, reflecting increased prevalence rates, expanding markets, and evolving prescribing patterns.
- Market penetration increases from 6% to approximately 10% in emerging markets due to improved healthcare access.
- New formulations or combination therapies may enhance uptake, but no specific pipeline products are assumed.
Sales Forecast Table (USD millions)
| Year | Estimated Sales (USD millions) | Notes |
|---|---|---|
| 2023 | 160 | Base year; market stabilization |
| 2024 | 173 | Growth driven by emerging markets |
| 2025 | 188 | Increased adoption in Asia-Pacific |
| 2026 | 204 | Higher penetration and awareness |
| 2027 | 221 | Peak sales; competitive pressures |
Key Drivers
- heightened prevalence of asthma globally, estimated to impact 262 million people in 2022 (Global Initiative for Asthma).
- expanding access in low- and middle-income countries.
- increased physician preference for GSK’s inhaler devices, despite competition.
What Are Potential Market Risks?
- Competitive pressure from generic versions and new inhaler technologies.
- Regulatory hurdles in certain markets, delaying launches.
- Price sensitivity in emerging economies limiting premium pricing.
- Patent protection expiry in key markets, although no current patent litigations specifically threaten ASMANEX 60.
What Are Strategic Considerations for Market Penetration?
- Partnering with regional distributors to deepen market reach.
- Promoting adherence and education programs.
- Developing combination inhalers to capture multi-condition treatment markets.
- Monitoring regulatory changes and patent landscapes continuously.
Key Takeaways
- ASMANEX 60 is positioned within a stable, mature inhaled corticosteroid market, with growth prospects in emerging economies.
- The global inhaler market’s CAGR is estimated at 6-8%, with increased adoption driven by rising asthma prevalence and expanding healthcare infrastructure.
- North America remains the dominant market, though Asia-Pacific exhibits the fastest growth.
- Competitive differentiation relies on dosing convenience, device design, and clinical efficacy.
- Sales are projected to reach USD 221 million by 2027, driven by increased treatment rates and improved access in emerging markets.
FAQs
Q1. What factors could significantly impact ASMANEX 60 sales?
Regulatory delays, patent litigations, or shifts toward generic inhalers could reduce market share. Conversely, increased global asthma prevalence and improved access could boost sales.
Q2. How does ASMANEX compare to competitors in terms of efficacy?
Clinical data indicate similar efficacy among ICS, with some studies citing superior adherence due to once-daily dosing. No definitive efficacy superiority over key competitors is claimed.
Q3. Are there new formulations or combination therapies in development?
GSK and competitors are developing combination inhalers (e.g., ICS with LABA) that may influence future sales. No specific ASMANEX combination has been approved as of 2023.
Q4. Which markets present the most growth opportunities?
Asia-Pacific, Latin America, and the Middle East show the highest growth potential owing to expanding healthcare access and rising disease prevalence.
Q5. How does patent expiration influence future sales?
Patent expiry typically enables generic competition, which can erode sales. GSK’s key patents for ASMANEX are expected to expire around 2025-2027, potentially impacting future revenue streams.
Cited References
- Global Initiative for Asthma, 2022 Report.
- MarketWatch, "Inhaled Corticosteroids Market Size, Share & Trends," 2022.
- GSK Annual Report 2022.
- IQVIA, "Global Respiratory Drugs Market Analysis," 2022.
- Euromonitor International, "Respiratory Devices and Pharmaceuticals," 2022.
More… ↓
