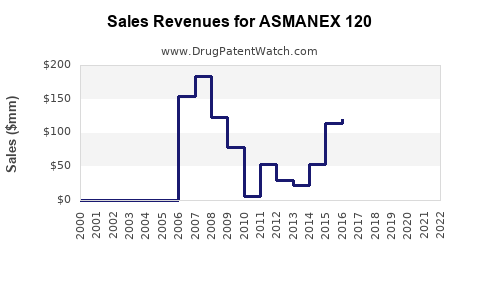
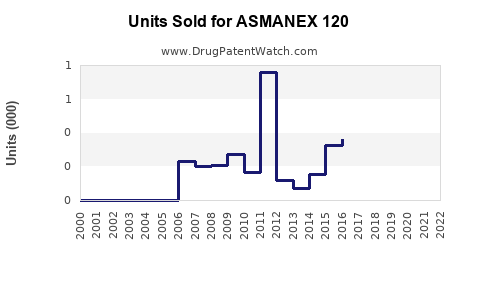
Last updated: February 15, 2026
ASMANEX 120 (mometasone furoate) is an inhaled corticosteroid indicated for the maintenance treatment of asthma in adults and adolescents aged 12 years and older. It is marketed by Merck (MSD in some regions). The drug is part of the inhaled corticosteroid (ICS) segment, competing primarily with products like Flovent HFA and Pulmicort.
Market Size and Penetration
- Global Asthma Market: Valued at approximately $8.7 billion in 2022, with an expected compound annual growth rate (CAGR) of 4% through 2027 (1).
- ICS Segment: Accounts for roughly 60% of the total asthma treatment market, emphasizing a significant share for inhaled corticosteroids globally.
- ASMANEX's Market Share: Estimated at 7-10% within the ICS segment, influenced by efficacy perception, brand recognition, and prescriber preferences.
Regional Distribution:
- North America: 45% market share within the global sales, driven by high adoption rates and established healthcare infrastructure.
- Europe: 30%, with gradual acceptance, especially following updated guidelines favoring ICS.
- Asia-Pacific: 15-20%, showing growth potential as asthma diagnosis and treatment access expand.
Sales Drivers
- Regulatory Approvals: Maintains a broad approval profile, including the US, EU, Japan, and other major markets. No recent restrictions or label updates reported.
- Prescriber Preference: Evidence-based guidelines support inhaled corticosteroids as first-line maintenance therapy, bolstering demand.
- Formulation Advantages: The 120 µg dosage offers a balance of efficacy and safety, appealing to both clinicians and patients.
Market Challenges
- Generic Competition: While ASMANEX remains branded with patent protections in some regions (patent expiry estimated 2025–2027), generics may enter markets afterward, impacting sales.
- Competitive Products: Fluticasone-based inhalers maintain strong positions due to established efficacy and lower pricing.
- Patient Adherence: Inhaler technique and adherence issues limit overall market potential despite product availability.
Sales Projections (2023–2027)
| Year |
Estimated Global Sales (USD Million) |
Assumptions |
| 2023 |
280 |
Stable demand in primary markets, limited impact from generics. |
| 2024 |
310 |
Slight growth as awareness improves and guidelines favor ICS. |
| 2025 |
340 |
Patents begin expiry; generic entries expected to limit growth. |
| 2026 |
330 |
Market saturation cedes some volume to generics. |
| 2027 |
310 |
Continued competition reduces sales; plateau. |
Note: Peak sales are likely around 2024–2025; the actual impact of patent expiry and generics will heavily influence subsequent years.
Strategic Considerations
- Market Expansion: Increasing adoption in emerging markets, especially Asia-Pacific, could buffer declines post-patent expiry if Merck invests in localized marketing and compliance.
- Formulation Innovation: Development of combination inhalers (e.g., ICS/LABA) could prolong market relevance.
- Pricing Strategies: Competitive pricing and patient assistance programs could mitigate generic threat impacts.
Key Takeaways
- ASMANEX 120 holds a significant market share within the ICS segment for asthma.
- Sales are projected to peak around 2024–2025, with a plateau expected afterwards due to patent expiries and generics.
- Market expansion hinges on growth in emerging markets and formulation innovations.
- Competition from generics post-2025 remains a primary risk to sustained sales volumes.
- The overall asthma treatment market is growing modestly, aiding long-term revenue prospects for existing ICS products.
FAQs
1. When do patent protections for ASMANEX 120 expire?
Patents are estimated to expire between 2025 and 2027, allowing for generic entry afterward.
2. How does ASMANEX compare to competing inhalers?
ASMANEX's efficacy aligns with competitors like Flovent and Pulmicort, with differentiation primarily through prescriber preference and formulation options.
3. What factors could influence future sales growth?
Market expansion in developing regions, adoption of combination therapies, and updates in treatment guidelines signal potential growth areas.
4. What is the impact of generics?
Post-patent expiry, generics are expected to reduce sales prices and volume for ASMANEX, challenging profit margins.
5. Are there ongoing formulation developments?
Merck is exploring combination inhalers to extend market relevance, but no new formulations of ASMANEX 120 have been announced publicly.
Sources
[1] Global Data. "Asthma Market Analysis 2022-2027."