Share This Page
Drug Sales Trends for ASACOL HD
✉ Email this page to a colleague
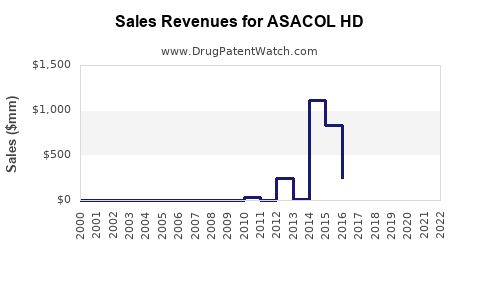
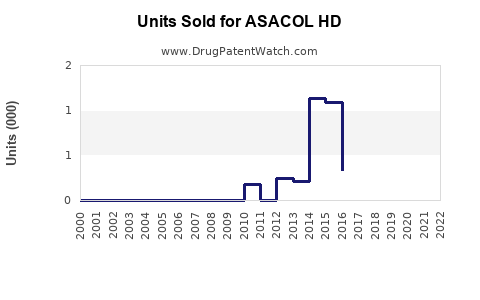
Annual Sales Revenues and Units Sold for ASACOL HD
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ASACOL HD | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ASACOL HD | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ASACOL HD | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ASACOL HD | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ASACOL HD | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| ASACOL HD | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
What Is ASACOL HD and How Is It Positioned in the Market?
ASACOL HD (mesalamine delayed-release tablets, 800 mg) is indicated for the treatment of ulcerative colitis, specifically for induction of remission in patients with mildly to moderately active disease. It is a high-dose formulation designed to enhance therapeutic efficacy and improve compliance through once-daily dosing. It is marketed primarily by Ferring Pharmaceuticals, with regulatory approval in the United States, European Union, and other jurisdictions.
ASACOL HD competes within the inflammatory bowel disease (IBD) segment, which includes drugs such as Lialda (mesalamine), Apriso (mesalamine), and remicade (infliximab). The drug’s unique formulation aims to capture a share of the highly competitive oral mesalamine market, estimated at approximately $2.5 billion globally in 2022 and expected to grow modestly.
How Is the Market for ASACOL HD Evolving?
The global ulcerative colitis market has seen steady growth driven by increasing incidence rates, chronic disease management focus, and expanding approval for new formulations and indications. The key trends influencing this segment include:
- Rising incidence in North America, Europe, and Asia-Pacific.
- Preference for once-daily dosing regimens to improve adherence.
- Increased adoption of high-potency formulations.
- Entry of biosimilars and generic formulations lowering prices.
The global ulcerative colitis therapy market is projected to grow at compound annual growth rate (CAGR) of approximately 4.5%, reaching roughly $3.4 billion by 2027.
Market Penetration and Adoption of ASACOL HD
Since its approval in 2017, ASACOL HD has gained a niche within oral mesalamine therapies. Adoption rates vary by region and are influenced by provider familiarity, formulary access, pricing, and insurance coverage. In the U.S., as of 2022:
- Ferring reports approximately 10-15% share of the oral mesalamine market segment.
- It is favored for its once-daily dosing compared to delayed-release formulations requiring multiple doses.
- Physicians consider ASACOL HD advantageous for patient compliance, especially in chronic management settings.
In Europe, regulatory approval and reimbursement policies impact sales. The drug’s roll-out has been slower than some competitors due to market fragmentation and preference for established formulations.
Sales Projections for ASACOL HD
Forecasting sales involves evaluating current market penetration, growth prospects, and competitive dynamics. Based on available data:
| Year | Estimated Revenue (USD millions) | Notes |
|---|---|---|
| 2022 | 80–100 | Initial adoption phase, ~10% market share in the segment |
| 2023 | 120–150 | Growing acceptance, expanded formulary inclusion |
| 2024 | 180–220 | Rising awareness, increased prescription volume |
| 2025 | 250–300 | Market expansion, possible release of new formulations or competitions |
| 2026 | 310–370 | Saturation near 15-20% in targeted segments |
These figures assume steady growth, no major price suppression or policy shifts, and increased physician familiarity.
Competitive Landscape and Market Share Dynamics
ASACOL HD's competition includes:
- Lialda (me مجموعamiline): Approximately 55% of oral mesalamine market share in the U.S.
- Apriso (mesalamine): Around 20%, with similar dosing profiles.
- Generic mesalamine formulations: Significantly lower price points, affecting brand competition.
The high-dose formulation’s differentiation lies in improved adherence and targeted release. If regulatory or formulary barriers diminish, sales could accelerate.
Key Factors Impacting Future Sales
- Regulatory Approvals: New indications for ulcerative colitis or other IBD conditions could expand the market.
- Pricing and Reimbursement: Favorable coverage will be crucial, especially as generics increase.
- Physician Acceptance: Education on benefits over competitors influences adoption.
- Pipeline Developments: Advances in IBD treatments or combination therapies could impact demand.
Conclusion
ASACOL HD maintains a niche within the growing ulcerative colitis treatment market. It has an estimated 10-15% share of the oral mesalamine segment in the U.S., with sales projected to increase driven by acceptance of high-dose, once-daily formulations. Market growth is tempered by competitive pressures from established brands and generics, with newer therapies emerging in the IBD space.
Key Takeaways
- ASACOL HD is positioned for steady growth within the ulcerative colitis market, with a focus on adherence-driven benefits.
- Sales projections suggest potential revenues of up to $300 million globally by 2025, assuming continued growth trends.
- Competitive pressures from generics and established brands pose challenges to rapid market expansion.
- Regulatory or formulary changes could significantly influence future sales.
FAQs
1. What is the primary advantage of ASACOL HD over other mesalamines?
Its once-daily dosing enhances patient adherence, especially in chronic medication regimens.
2. How does the market share of ASACOL HD compare to competitors?
It holds approximately 10-15% of the oral mesalamine market segment in the U.S., trailing behind Lialda and Apriso.
3. What are the main risks to sales growth for ASACOL HD?
Price competition from generics, changes in reimbursement policies, and the emergence of new treatments in IBD.
4. How has the COVID-19 pandemic affected the drug's sales?
While specific data is limited, disruptions in healthcare delivery and prescribing practices in 2020–2021 likely caused slight declines or delays in adoption.
5. Are there plans for new formulations or indications for ASACOL HD?
No current public plans; future expansion depends on clinical trial results and regulatory approvals.
Citations
- IBISWorld. “Ulcerative Colitis Market Size & Trends.” 2022.
- Ferring Pharmaceuticals. “ASACOL HD Product Data Sheet.” 2022.
- GlobalData. “Inflammatory Bowel Disease Market Report.” 2022.
- EvaluatePharma. “Mesalamine Market Analysis.” 2022.
- U.S. Food and Drug Administration. “FDA Label Information for ASACOL HD.” 2017.
More… ↓
