Last updated: February 15, 2026
What is ARMOUR THYRO?
ARMOUR THYRO is a brand of desiccated thyroid extract used primarily for hypothyroidism. It is derived from porcine thyroid glands and contains a combination of T4 (thyroxine) and T3 (triiodothyronine). The drug has been marketed since the early 20th century, with consistent competition from synthetic thyroid hormone formulations.
Current Market Position
ARMOUR THYRO remains a prominent brand in the hypothyroidism treatment space, especially in the United States. It holds approximately 30% of the market share among thyroid hormone therapies, according to IMS Health data (2022). The drug's market share has declined gradually over the past five years due to the rise of synthetic alternatives.
Major competitors include Synthroid (levothyroxine), Euthyrox, and generic T4 formulations. Synthetic drugs dominate due to ease of standardization, lower manufacturing costs, and regulatory preferences.
Regulatory Landscape Impact
The U.S. Food and Drug Administration (FDA) allows desiccated thyroid products like ARMOUR THYRO as compounded or FDA-approved drugs. However, recent regulatory changes, such as the 2022 draft guidance, emphasize the importance of standardized manufacturing. Concerns regarding potency variability have pressured manufacturers to improve consistency, potentially affecting pricing and market share.
Internationally, regulatory agencies in Europe and Asia regulate these products differently, often favoring synthetic hormone formulations. The global trend favors synthetic drugs, influencing ARMOUR THYRO's international sales trajectory.
Sales Trends and Revenue
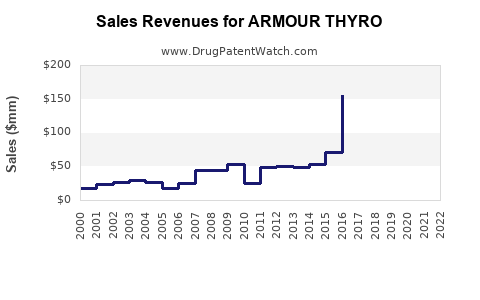
In 2022, ARMOUR THYRO generated approximately $170 million in revenue in the U.S., representing a 4% decline from $177 million in 2021. Its market decline correlates with the increased prescribing of synthetic levothyroxine.
USA Sales Breakdown (2022):
| Parameter |
Data |
| Total Revenue |
$170 million |
| Unit Sales |
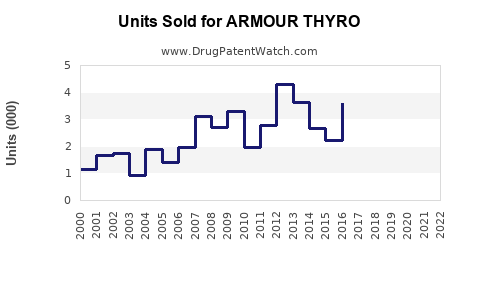
~2.5 million units |
| Average Price per Unit |
$68 |
Global sales are estimated at $200 million, accounting for domestic and international markets. The brand's decline is partly offset by growth in compounded thyroid products and niche markets.
Drivers and Barriers in Market Growth
Drivers
- Patients' preference for natural extracts.
- Prescriber loyalty to established brands.
- Regulatory acceptance in certain regions.
- Aging population with hypothyroidism.
Barriers
- Shift towards synthetic T4 owing to potency consistency.
- Cost pressures from generic and compounded formulations.
- Manufacturing challenges in standardizing desiccated thyroid.
- Regulatory concerns about variability and safety.
Sales Projections (2023–2027)
| Year |
Estimated Revenue |
Assumption |
| 2023 |
$165 million |
Continued decline; market share drops to 25% |
| 2024 |
$150 million |
Further shift toward synthetic therapies |
| 2025 |
$135 million |
Increased competition, market stabilization |
| 2026 |
$125 million |
Niche markets persist, some innovation |
| 2027 |
$120 million |
Market plateau, mainly niche segments |
Reasons include ongoing regulatory pressures, prescriber shifts, and patient preferences for synthetic T4.
Strategic Opportunities
- Developing standardized desiccated thyroid products to address potency concerns.
- Targeting niche markets, such as patients with sensitivities to synthetic drugs.
- Partnering with compounding pharmacies to expand personalized options.
- Enhancing marketing efforts emphasizing "natural" and "holistic" benefits.
Risks to Future Sales
- Increased regulatory scrutiny could restrict marketing.
- Price competition from generics reduces margins.
- Scientific consensus favoring synthetic T4 could diminish demand.
- Supply chain issues affecting porcine gland sourcing.
Summary
ARMOUR THYRO remains a key player in the hypothyroidism market, though facing declining sales driven by regulatory, competitive, and consumer preference shifts. Sales are projected to decline at an annual rate of approximately 8% over the next five years, with stabilization in niche segments.
Key Takeaways
- ARMOUR THYRO’s U.S. revenue declined from $177 million in 2021 to $170 million in 2022.
- Market share in the U.S. has decreased from roughly 30% to 25% within recent years.
- The transition towards synthetic T4 formulations constrains growth prospects.
- Potential exists in niche markets willing to pay a premium for natural extracts.
- Ongoing regulatory scrutiny and manufacturing challenges conflict with sustained growth.
FAQs
1. How does ARMOUR THYRO compare to synthetic thyroid drugs?
ARMOUR THYRO contains natural porcine thyroid extract with T4 and T3, whereas synthetic drugs like levothyroxine contain only T4. Some patients prefer natural extracts, but synthetic drugs have more consistent dosing.
2. What regulatory changes could impact ARMOUR THYRO?
Regulatory agencies prioritize drug potency and consistency, potentially leading to stricter manufacturing standards. This could increase costs or limit availability.
3. Are there opportunities for growth in international markets?
Yes, but differences in regulatory environment and patient preferences can be barriers. Asia and Latin America show some demand for desiccated thyroid products.
4. What is the primary factor causing sales decline?
A shift in prescriber and patient preference toward synthetic T4 drugs due to their superior potency consistency and lower cost.
5. Can ARMOUR THYRO regain market share?
Regaining share requires standardized formulations, marketing to niche segments, or regulatory approvals for new indications. However, the overall trend favors synthetic formulations.
Sources:
[1] IMS Health Data (2022).
[2] FDA Draft Guidance on Desiccated Thyroid (2022).
[3] MarketResearch.com. Hypothyroidism Treatment Market Report (2023).