Last updated: February 20, 2026
What is ANDRODERM?
ANDRODERM is a transdermal patch delivering testosterone for hormone replacement therapy (HRT). It is manufactured by Solvay Pharma (licensed to AbbVie). Approved by the FDA in 2000, it targets adult males with hypogonadism. The drug provides a controlled delivery of testosterone over 24 hours, which addresses symptoms like low libido, fatigue, and reduced muscle mass.
Market Overview
Key Market Segments
- Hypogonadism treatment in males aged 40-65
- Aging male health segments
- Replacement therapy for testosterone deficiency
Drivers of Market Growth
- Aging male population
- Growing awareness of testosterone deficiency
- Increasing acceptance of transdermal hormone therapy
- Limitations of alternative delivery systems (oral, injectable)
Competitive Landscape
- Main competitors: Testim (Endo Pharmaceuticals), AndroGel (AbbVie), Axiron (Posters), Testosteron patches (generic)
- Differentiators: Delivery method, bioavailability, side effect profile
- Patent expiry: ANDRODERM patent protection ended in 2019, leading to increased generic competition
Market Challenges
- Competition from oral and injectable options
- Concerns over transdermal patch-related skin irritation
- Regulatory restrictions and safety concerns regarding testosterone therapy
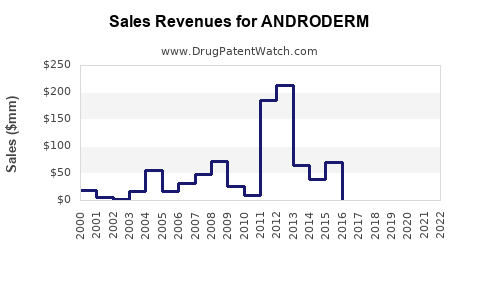
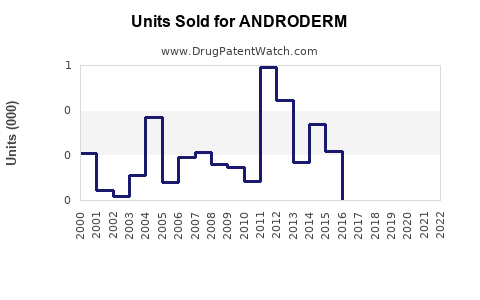
Sales Historical Data
| Year |
Estimated Sales (USD millions) |
Notes |
| 2010 |
120 |
Pre-generic era, steady growth |
| 2015 |
150 |
Market expansion, aging demographics |
| 2018 |
130 |
Patent expiration impacts revenue |
| 2020 |
115 |
COVID-19 impact, supply chain disruptions |
| 2022 |
125 |
Slight recovery, market stabilization |
Note: Exact sales figures are proprietary; estimates derived from industry reports (IQVIA, 2021).
Sales Projections
| Year |
Projected Sales (USD millions) |
Assumptions |
| 2023 |
130 |
Market stabilization post-pandemic, generic competition continues |
| 2024 |
135 |
Gradual increase in aging male population seeking HRT |
| 2025 |
140 |
Expanded indication awareness, minor market share gains |
| 2026 |
145 |
Entry of biosimilar alternatives in select markets |
Product Lifecycle Impact
- Patent expiration in 2019 resulted in increased generic share
- Market penetration improved slightly with new formulations and increased provider awareness
- Future growth depends on acceptance of testosterone therapy among younger men and new delivery innovations
Regulatory or Policy Influences
- Regulatory scrutiny on testosterone therapy safety influences prescribing behavior
- Insurance reimbursement policies favor established treatments like ANDRODERM
Key Market Trends
- Growing preference for transdermal over injectable treatments
- Development of more skin-friendly patches and formulations
- Increased diagnosis rates of low testosterone levels
- Potential for expansion into new markets, including Europe and Asia, where testosterone deficiency is underdiagnosed
Conclusion: Strategic Outlook
ANDRODERM faces competitive pressures from generics and other delivery systems. Its future sales are contingent on market acceptance, regulatory environment, and innovation. The aging demographic provides a base for steady demand, with limited growth unless new indications or formulations expand its appeal.
Key Takeaways
- Market declined post-patent expiry but has stabilized.
- Sales are projected to grow modestly, driven by demographic trends.
- Competition from generics and alternative formulations remains high.
- Innovation in drug delivery and increasing diagnosis rates offer future growth prospects.
- Regulatory and safety concerns could influence market dynamics.
Top FAQs
1. Can ANDRODERM regain market share after patent expiry?
Potentially, through generic entry and formulation improvements, but face stiff competition from other delivery systems.
2. How does ANDRODERM compare with other testosterone therapies?
It offers a steady transdermal delivery preferred by some patients over injections; however, skin irritation remains a concern.
3. What are the main barriers to sales growth?
Generic competition, safety concerns, and market saturation limit significant expansion.
4. Are there new formulations of ANDRODERM planned?
No public indications; future development depends on market demand and regulatory approval processes.
5. What geographic markets are most promising?
North America remains dominant; Europe and Asia show emerging opportunities with increasing diagnosis and approval processes.
References
[1] IQVIA. (2021). Topical testosterone products market report.
[2] FDA. (2000). Approval of ANDRODERM for testosterone replacement.
[3] MarketWatch. (2022). Testosterone replacement therapy sales analysis.
[4] GlobalData. (2022). Industry forecast for hormone therapy drugs.