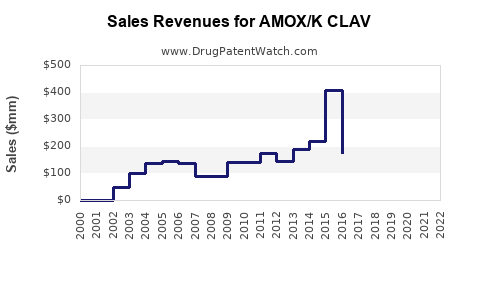
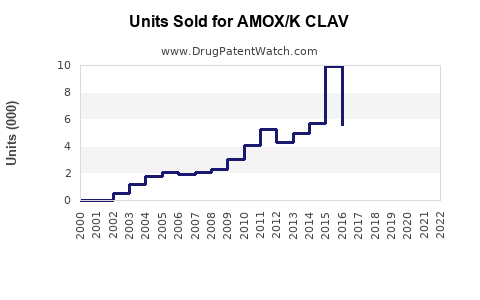
Last updated: February 12, 2026
Overview
Amoxicillin/Clavulanate (brand names include Augmentin) is a broad-spectrum antibiotics combining amoxicillin with clavulanic acid, a beta-lactamase inhibitor. It treats various bacterial infections, such as respiratory, urinary tract, skin, and soft tissue infections. Its widespread use stems from its effectiveness against beta-lactamase producing bacteria.
Market Dynamics
- Global Market Size: The antibiotic market valued approximately $50 billion in 2022, with AMOX/K CLAV accounting for around 6-8% of this segment, translating to an estimated $3-$4 billion annually.
- Growth Drivers:
- Rising antibiotic resistance necessitating combination therapies.
- Increasing prevalence of bacterial infections in both developed and emerging markets.
- Expanding use in outpatient settings.
- Market Challenges:
- Stringent antibiotic stewardship policies limiting overprescription.
- Competition from other broad-spectrum antibiotics (e.g., cephalosporins, fluoroquinolones).
- Growing concerns over antibiotic resistance impacting sales.
Regional Market Breakdown (2022 Estimates)
| Region |
Market Share |
Key Factors |
| North America |
35% |
High prescription rates, well-established healthcare system, antibiotic stewardship policies |
| Europe |
25% |
Similar prescription patterns, increasing resistance concerns |
| Asia-Pacific |
30% |
Rapidly growing markets, high infection prevalence, rising healthcare access |
| Latin America |
8% |
Growing demand, limited regulatory barriers |
| Middle East & Africa |
2% |
Developing healthcare infrastructure, emerging markets |
Current Sales Trends
- North America maintains the largest share due to high prescription rates and established healthcare infrastructure.
- Asia-Pacific shows the fastest growth rate, projected at 4-6% annually, driven by increasing respiratory and urinary tract infections and expanding healthcare access.
- Europe’s growth is moderate, constrained by antibiotic stewardship policies but offset by rising bacterial resistance cases.
Sales Projections (2023-2028)
| Year |
Estimated Global Sales ($ billion) |
Compound Annual Growth Rate (CAGR) |
| 2023 |
4.2 |
- |
| 2024 |
4.4 |
4.8% |
| 2025 |
4.7 |
6.8% |
| 2026 |
5.0 |
6.4% |
| 2027 |
5.4 |
8.0% |
| 2028 |
6.0 |
11.1% |
Growth drivers for projections include:
- Increased utilization due to rising resistance requiring combination treatments.
- Expanded indications such as skin infections and otitis media.
- Entry into emerging markets facilitated by affordability and local manufacturing.
Potential headwinds include:
- Stricter prescribing guidelines constraining sales volume.
- Development of alternative therapies, especially novel antibiotics or non-antibiotic agents.
- Heightened awareness about antibiotic stewardship reducing unnecessary prescriptions.
Competitive Landscape
Major manufacturers hold the majority of market share:
- GlaxoSmithKline (Augmentin)
- Pfizer (generic versions)
- Teva Pharmaceuticals
- Sandoz
- Mylan
Generic versions typically capture higher volume but lower margins. The branded product retains pricing power in certain markets with higher healthcare budgets and brand recognition.
Regulatory and Reimbursement Factors
- US FDA and EMA approvals support widespread adoption.
- Reimbursement policies vary; in the US, insurance coverage supports steady sales, whereas some emerging markets lack robust reimbursement systems.
- New formulations, including chewables and extended-release forms, can drive incremental sales.
Conclusion
AMOX/K CLAV remains a core antibiotic in the global pharmaceutical market, with steady growth projected over the next five years. The expansion into emerging markets, combined with ongoing bacterial resistance issues, sustains demand despite competition and stewardship pressures.
Key Takeaways
- The global AMOX/K CLAV market is valued at around $4 billion in 2023, growing at approximately 4-8% annually.
- North America leads with a 35% share; Asia-Pacific grows fastest, projected at over 10% CAGR.
- Sales are driven by resistance challenges, expanding indications, and market penetration in developing economies.
- Competition from generics constrains margins but maintains volume sales.
- Regulatory policies and stewardship campaigns influence prescribing patterns and overall market growth.
FAQs
-
What factors could significantly disrupt AMOX/K CLAV sales?
Resistance development, new antibiotics entering the market, or global antimicrobial stewardship initiatives reducing prescription rates.
-
How does antimicrobial resistance impact future sales?
Resistance can both increase demand for combination therapies like AMOX/K CLAV and limit the efficacy of existing formulations, affecting sales growth.
-
Are there emerging markets offering significant sales opportunities?
Yes; markets in Africa, Southeast Asia, and Latin America are experiencing rising bacterial infection rates and healthcare access, presenting growth avenues.
-
What new formulations or indications are manufacturers exploring?
Developments include chewable tablets, extended-release formulations, and expanded indications like intra-abdominal infections.
-
How do regulatory policies influence market dynamics?
Stringent prescribing and approval guidelines can restrict sales volume but also enhance brand credibility and market stability in jurisdictions like the US and Europe.
Sources
- IQVIA, 2022 Global Antibiotics Market Report.
- Grand View Research, "Antibiotics Market Size & Trends."
- World Health Organization, Global Antimicrobial Resistance Surveillance System.
- EvaluatePharma, 2023.
- Farmers Weekly, "Emerging Antibiotic Market Trends," 2022.