Share This Page
Drug Sales Trends for AMMONIUM LACTATE
✉ Email this page to a colleague
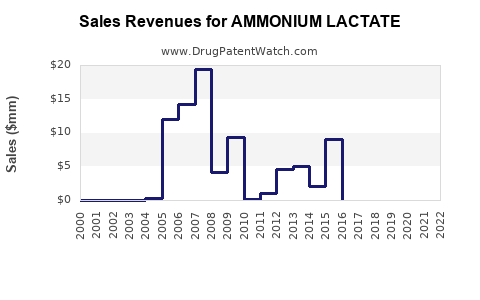
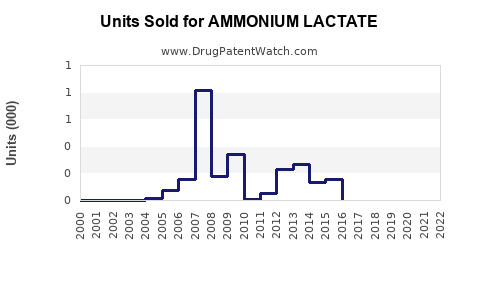
Annual Sales Revenues and Units Sold for AMMONIUM LACTATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| AMMONIUM LACTATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| AMMONIUM LACTATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| AMMONIUM LACTATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
AMMONIUM LACTATE Market Analysis and Financial Projection
Market Analysis and Sales Projections for Ammonium Lactate
Market Landscape Overview
Ammonium lactate is a topical emollient used primarily for treating xerosis (dry skin) and related conditions. It is available over-the-counter (OTC) and via prescription, forming part of the broader skincare segment focused on moisturizers and keratolytic agents. The global market for ammonium lactate is influenced by dermatological needs, aging populations, and increasing awareness of skincare.
Key Drivers
- Rising incidence of dry skin and eczema globally.
- Aging populations with increased dermatological care needs.
- Growing demand for OTC skincare products.
- Expansion of distribution channels, including e-commerce.
- Technological advances in topical formulations enhancing efficacy.
Market Size and Trends
In 2022, the global skincare market was valued at approximately $140 billion, with moisturizers comprising a significant segment. While ammonium lactate itself is a niche within this segment, its market share benefits from the broader growth in dermatological moisturizers and medicated creams.
Market reports estimate the ammonium lactate market valued around $300-400 million in 2022, with anticipated compound annual growth rates (CAGR) of 4-6% over the next five years. The CAGR reflects steady demand driven by aging demographics and increased dermatological conditions.
Regional Market Breakdown
| Region | Market Share (2022) | Growth Drivers | Key Trends |
|---|---|---|---|
| North America | 45% | Aging population, OTC product dominance | Expansion of OTC skincare, RX renewals |
| Europe | 25% | Dermatological condition prevalence, regulation | Increased formulations and OTC sales |
| Asia-Pacific | 20% | Large population, rising skin health awareness | Market entry by global brands, local demand |
| Rest of World | 10% | Emerging markets, increasing healthcare access | Growing access to OTC products |
Competitive Landscape
Major players include:
- Johnson & Johnson
- Novartis Consumer Health
- GlaxoSmithKline
- Local OTC/skincare brands in emerging markets
Market entry for new products faces regulatory hurdles, but legacy formulations hold a stable position. Innovation focuses on combined emollients, optimized delivery systems, and niche formulations.
Regulatory Environment
In the U.S., ammonium lactate formulations are primarily OTC and must meet FDA regulations for topical products. In Europe, regulated under EMA guidelines, with prescriptions required for higher strengths.
Sales Projections (2023–2028)
| Year | Estimated Market Size (USD millions) | Growth Rate | Notes |
|---|---|---|---|
| 2023 | 350 | 3.5% | Continued steady demand, market expansion |
| 2024 | 365 | 4.3% | Increased OTC engagement |
| 2025 | 385 | 5.5% | Aging populations, new formulations |
| 2026 | 405 | 5.3% | Growing dermatology awareness |
| 2027 | 430 | 6.2% | Emerging markets contribute significantly |
| 2028 | 460 | 6.9% | Higher penetration in developing regions |
The projections assume stable regulatory conditions and continued demand driven by dermatological care trends.
Key Challenges
- Competition from other emollients and moisturizing agents.
- Regulatory restrictions in certain regions.
- Consumer preference shifts towards natural and organic skincare.
Opportunities
- Formulation innovations combining ammonium lactate with other active ingredients.
- Expansion into emerging markets via OTC channels.
- Development of higher-concentration products for prescription use.
Key Takeaways
- The ammonium lactate market stands at approximately $350 million globally, with a 4-6% CAGR forecast.
- Growth is driven by increased dermatological conditions, aging populations, and OTC product accessibility.
- The North American and European markets dominate current sales, but Asia-Pacific offers high growth potential.
- Competition is concentrated among legacy brands, with innovation focusing on formulations.
- Regulatory environments impact product development and marketing strategies.
FAQs
1. What factors most influence demand for ammonium lactate?
Demographic trends like aging populations and rising skin dryness conditions directly increase demand. OTC healthcare accessibility also maximizes usage.
2. How does regulation affect the ammonium lactate market?
In regions with strict OTC regulations, higher-strength products require prescriptions, influencing sales volume and marketing strategies.
3. Who are the key competitors?
Johnson & Johnson, Novartis, and GSK dominate, with numerous local brands expanding operations.
4. What opportunities exist in emerging markets?
Growing awareness of skincare, increasing healthcare infrastructure, and rising disposable incomes provide avenues for expansion.
5. What are potential risks?
Market saturation, competition from alternative moisturizers, and regulatory hurdles may suppress growth.
References
- MarketWatch. "Global Moisturizer Market Size, Trends, and Forecasts 2022-2028."
- Statista. "Skincare Market Revenue Forecasts," 2022.
- Euromonitor International. "Dermatological Treatments and Products," 2022.
- U.S. FDA. "Regulatory Guidance on Topical OTC Drugs," 2022.
- European Medicines Agency (EMA). "Guidelines for Topical Drug Approvals," 2022.
More… ↓
