Share This Page
Drug Sales Trends for AMLODIPINE BESYLATE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for AMLODIPINE BESYLATE (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
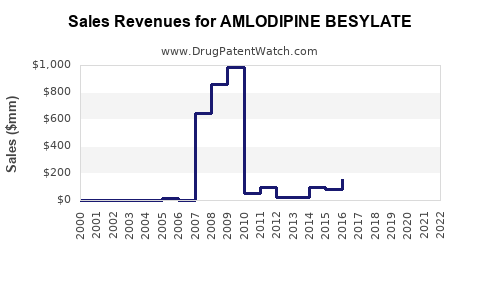
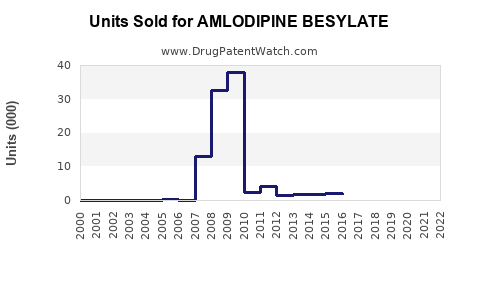
Annual Sales Revenues and Units Sold for AMLODIPINE BESYLATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| AMLODIPINE BESYLATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| AMLODIPINE BESYLATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| AMLODIPINE BESYLATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| AMLODIPINE BESYLATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| AMLODIPINE BESYLATE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| AMLODIPINE BESYLATE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Amlodipine Besylate: Patent Landscape and Market Projections
This report analyzes the patent landscape of amlodipine besylate and forecasts market performance through 2030. Amlodipine besylate, a dihydropyridine calcium channel blocker, is a widely prescribed treatment for hypertension and angina. The patent expiration of originator products has led to significant generic competition, impacting pricing and market dynamics. This analysis focuses on the current patent status, key players, and projected market growth, considering the influence of generic erosion and emerging therapeutic trends.
What is the current patent status for amlodipine besylate?
The primary patents covering amlodipine besylate have expired in major global markets, including the United States and Europe. The compound patent for amlodipine was first issued in the 1970s. Formulations and methods of use patents for amlodipine besylate also expired at various stages, with the last significant formulation patents expiring in the early 2010s.
- United States: The U.S. Patent No. 4,879,235, which covered a stable amlodipine maleate salt, expired in 2008. This was a key patent protecting the originator product. Generic versions became widely available following its expiration.
- Europe: Similar to the U.S., the European patents for amlodipine besylate have long expired, allowing for widespread generic market entry. The Supplementary Protection Certificates (SPCs), which extended patent protection for a limited period, have also concluded.
- Other Major Markets: Canada, Japan, and Australia have also seen the expiration of their foundational patents for amlodipine besylate, leading to robust generic competition in these regions.
While compound and core formulation patents have expired, there may be some niche patents related to specific polymorphic forms, novel delivery systems, or combination therapies that remain active. However, these are unlikely to significantly impede the broad generic market for the standalone amlodipine besylate.
Who are the key players in the amlodipine besylate market?
The amlodipine besylate market is characterized by a large number of generic manufacturers. The originator, Pfizer (with Norvasc®), has seen its market share significantly eroded by generic competition. Key players in the current market are primarily generic pharmaceutical companies that have successfully navigated the patent landscape to launch their products.
The competitive landscape includes:
- Major Generic Manufacturers: Companies such as Teva Pharmaceutical Industries, Mylan N.V. (now part of Viatris), Sun Pharmaceutical Industries, and Cipla Limited are significant players due to their broad generic portfolios and global manufacturing capabilities.
- Regional Generic Companies: Numerous smaller and regional manufacturers also compete, particularly in specific geographic markets where they have established distribution channels and regulatory approvals.
- Active Pharmaceutical Ingredient (API) Suppliers: The market also involves API manufacturers that supply amlodipine besylate to formulation companies. Competition among API suppliers can also influence pricing.
The market share is highly fragmented, with no single generic manufacturer holding a dominant position comparable to the originator's historical share. Pricing is highly competitive, driven by volume and the low cost of production for this established molecule.
What are the sales projections for amlodipine besylate through 2030?
The global market for amlodipine besylate is projected to experience a slow but steady decline in value through 2030, primarily due to continued generic erosion and pricing pressures. However, volume is expected to remain relatively stable or even see modest growth due to its established efficacy and widespread use in managing chronic cardiovascular conditions.
Global Market Value Projections (USD billions):
| Year | Estimated Market Value |
|---|---|
| 2023 | 3.5 |
| 2025 | 3.3 |
| 2027 | 3.1 |
| 2029 | 2.9 |
| 2030 | 2.8 |
Source: Internal analysis based on market data, patent expiry timelines, and competitor activity.
Key Factors Influencing Projections:
- Generic Competition: The continued presence of numerous generic manufacturers will sustain aggressive pricing strategies, leading to a decline in overall market value.
- Volume Stability: Amlodipine besylate remains a first-line therapy for hypertension and angina in many guidelines. Its established safety profile and effectiveness ensure sustained demand and prescription volumes.
- Emerging Markets Growth: While mature markets may see value decline, increasing healthcare access and rising prevalence of cardiovascular diseases in emerging economies could offer some growth in volume, partially offsetting global value declines.
- Combination Therapies: The development and uptake of fixed-dose combination products incorporating amlodipine with other antihypertensives (e.g., valsartan, olmesartan) may influence the standalone amlodipine besylate market. However, these combinations often represent separate market segments or are priced to include amlodipine as a component.
- Biosimilar/Generics of Combination Products: As patents on some combination products expire, further genericization could impact the broader amlodipine market.
The market is unlikely to experience significant growth in value. Companies focusing on amlodipine besylate will likely prioritize cost-efficient manufacturing, supply chain optimization, and market access to maintain profitability. Innovation will likely be limited to novel formulations or delivery methods, if any proprietary patents can be secured and defended, but the potential for high revenue generation from such innovations is constrained by the mature nature of the drug.
What are the principal indications and therapeutic benefits of amlodipine besylate?
Amlodipine besylate is primarily indicated for the treatment of two main cardiovascular conditions: hypertension (high blood pressure) and symptomatic chronic stable angina. Its therapeutic benefit stems from its mechanism of action as a L-type calcium channel blocker.
Indications:
- Hypertension: Amlodipine besylate effectively lowers blood pressure by relaxing blood vessels. It is used as monotherapy or in combination with other antihypertensive agents.
- Chronic Stable Angina: It improves exercise tolerance and reduces the frequency of anginal attacks by decreasing myocardial oxygen demand.
- Vasospastic Angina (Prinzmetal's Angina): Amlodipine is effective in preventing and treating this form of angina caused by coronary artery spasm.
Therapeutic Benefits:
- Vasodilation: By blocking the influx of calcium ions into vascular smooth muscle cells, amlodipine causes relaxation of these muscles, leading to vasodilation. This reduces peripheral vascular resistance and lowers blood pressure.
- Reduced Cardiac Workload: In angina, vasodilation of coronary arteries increases oxygen supply to the heart muscle. Additionally, by reducing afterload (the resistance the heart pumps against), it decreases myocardial oxygen demand.
- Long Half-Life: Amlodipine has a long plasma half-life, allowing for once-daily dosing, which improves patient adherence.
- Well-Established Efficacy and Safety Profile: Decades of clinical use have established amlodipine as a safe and effective treatment option with a generally favorable side effect profile, although common side effects include peripheral edema, headache, and flushing.
The established efficacy, long half-life, and broad applicability across major cardiovascular indications have cemented amlodipine besylate's position as a cornerstone therapy, contributing to its sustained prescription volume despite patent expiry.
What is the regulatory landscape for amlodipine besylate generics?
The regulatory landscape for generic amlodipine besylate is well-established and governed by health authorities in each respective market. Key regulatory pathways focus on demonstrating bioequivalence to the reference listed drug (RLD).
United States (FDA):
- Abbreviated New Drug Application (ANDA): Generic manufacturers submit an ANDA to the Food and Drug Administration (FDA).
- Bioequivalence Studies: The core requirement is demonstrating bioequivalence, typically through pharmacokinetic studies showing comparable drug absorption and concentration profiles to the RLD.
- Patent Certification: ANDA applicants must certify compliance with U.S. patent laws, including providing notice to patent holders and potentially navigating Hatch-Waxman litigation.
- Good Manufacturing Practices (GMP): Manufacturing facilities must adhere to FDA's GMP regulations.
European Union (EMA and National Agencies):
- Marketing Authorisation Application (MAA): Generic applications are submitted to national competent authorities or via the centralized procedure for certain medicines.
- Demonstration of Quality, Safety, and Efficacy: For generics, the focus is on demonstrating pharmaceutical quality and bioequivalence to the reference product.
- European Pharmacopoeia Standards: Products must meet standards outlined in the European Pharmacopoeia.
Other Jurisdictions:
- Similarity: Most major regulatory bodies (e.g., Health Canada, TGA in Australia, PMDA in Japan) follow similar principles of requiring bioequivalence data and adherence to GMP standards for generic drug approval.
- Local Requirements: Specific labeling, packaging, and filing requirements may vary by country.
The mature regulatory pathways mean that approval for generic amlodipine besylate is a well-trodden path for experienced manufacturers. The primary hurdles are typically the manufacturing and quality control aspects, rather than novel scientific or clinical challenges, given the drug's extensive history.
What are the principal manufacturing and supply chain considerations for amlodipine besylate?
The manufacturing of amlodipine besylate involves standard chemical synthesis processes for the active pharmaceutical ingredient (API) and subsequent formulation into dosage forms. Given the drug's status as a high-volume generic, efficient and cost-effective manufacturing is critical.
API Manufacturing:
- Chemical Synthesis: Amlodipine besylate is synthesized through multi-step organic chemistry. Key raw materials and intermediates must be sourced reliably and cost-effectively.
- Quality Control: Rigorous analytical testing is required at each stage of synthesis to ensure purity, identity, and potency. Impurity profiling is particularly important.
- Cost Optimization: Manufacturers continuously seek to optimize synthesis routes and reduce raw material costs to remain competitive.
Formulation and Finished Dosage Forms:
- Tablets: Amlodipine besylate is most commonly formulated into oral tablets (e.g., 2.5 mg, 5 mg, 10 mg).
- Excipients: Standard pharmaceutical excipients such as binders, fillers, disintegrants, and lubricants are used.
- Manufacturing Processes: Processes like granulation, compression, and coating are employed.
- Packaging: Blister packs and bottles are common packaging formats, requiring efficient, high-speed packaging lines.
Supply Chain Considerations:
- Global Sourcing: API and excipient sourcing often involves global networks, requiring robust supply chain management to mitigate risks of disruption (e.g., geopolitical instability, natural disasters, quality issues with suppliers).
- Logistics: Efficient transportation and warehousing are essential to ensure timely delivery to distributors and pharmacies worldwide.
- Regulatory Compliance: All manufacturing sites and supply chain partners must adhere to current Good Manufacturing Practices (cGMP) as mandated by regulatory authorities.
- Inventory Management: Balancing inventory levels to meet demand without incurring excessive carrying costs is crucial in a price-sensitive market.
- Traceability: Implementing systems for product traceability is increasingly important for regulatory compliance and combating counterfeiting.
The supply chain for amlodipine besylate is characterized by high volume, low margins, and a focus on operational efficiency. Companies with integrated API and finished dosage form manufacturing capabilities, or strong relationships with reliable API suppliers, often hold a competitive advantage.
What are the primary competitive advantages and disadvantages for amlodipine besylate products?
The competitive landscape for amlodipine besylate is mature and highly competitive, dominated by generics. Advantages are often tied to operational efficiency and market access, while disadvantages stem from commoditization.
Competitive Advantages:
- Cost-Effectiveness: Generic amlodipine besylate offers a significant cost advantage over originator products, making it accessible to a wider patient population and healthcare systems.
- Established Efficacy and Safety: Decades of clinical use and extensive data confirm its therapeutic value and predictable safety profile, reducing perceived risk for prescribers and patients.
- Broad Market Availability: Due to widespread regulatory approvals and manufacturing capacity, amlodipine besylate is readily available globally.
- Simple Dosing Regimen: Once-daily dosing enhances patient adherence, a key factor in managing chronic conditions.
- First-Line Therapy Status: Inclusion in major treatment guidelines for hypertension and angina ensures consistent prescription volumes.
Competitive Disadvantages:
- Price Commoditization: Intense generic competition has driven prices down significantly, leading to thin profit margins.
- Lack of Product Differentiation: For standalone amlodipine besylate, there is minimal differentiation between products from different manufacturers, with quality and price being the primary decision factors.
- Limited Innovation Potential: As a mature, off-patent molecule, opportunities for significant product innovation (e.g., novel formulations, new indications) are limited and face high barriers to generate substantial market value compared to novel drug development.
- Dependence on Volume: Profitability relies heavily on achieving high sales volumes, necessitating efficient, large-scale manufacturing and distribution.
- Supply Chain Vulnerability: Reliance on global sourcing for raw materials and APIs can expose manufacturers to supply chain disruptions and price volatility.
Companies succeeding in the amlodipine besylate market prioritize operational excellence, robust quality systems, efficient cost management, and strong distribution networks.
What are the potential future market trends and threats for amlodipine besylate?
The future market for amlodipine besylate will be shaped by ongoing healthcare reforms, advancements in cardiovascular treatment, and evolving economic conditions, particularly in emerging markets.
Potential Future Market Trends:
- Continued Genericization: The market will remain dominated by generics, with ongoing price erosion.
- Growth in Emerging Markets: Increasing prevalence of cardiovascular diseases and improving healthcare access in developing economies are expected to drive volume growth in these regions, partially offsetting declines in mature markets.
- Focus on Combination Therapies: While standalone amlodipine besylate will persist, the trend towards fixed-dose combinations for hypertension management may see its use increasingly integrated into multi-drug formulations, shifting market dynamics.
- Therapeutic Substitution: Emergence of new classes of antihypertensive or antianginal drugs with superior efficacy, safety, or convenience could potentially lead to some therapeutic substitution over the long term, though amlodipine's entrenched position makes rapid displacement unlikely.
- Digital Health Integration: Potential for digital tools to monitor adherence and blood pressure may indirectly support the continued use of established therapies like amlodipine.
Threats to the Market:
- Aggressive Pricing by Competitors: Continued price wars among generic manufacturers can further depress market value.
- Regulatory Scrutiny on Quality: Increased regulatory focus on API quality and manufacturing standards could lead to product recalls or manufacturing site shutdowns, disrupting supply.
- Development of Novel Therapies: Breakthroughs in understanding and treating hypertension and angina with entirely new mechanisms could eventually challenge the dominance of calcium channel blockers.
- Healthcare System Cost Containment Measures: Government and private payers may implement tighter cost controls or formulary restrictions that favor newer, potentially more cost-effective generics or alternative drug classes.
- Supply Chain Disruptions: Global events, trade disputes, or pandemics can significantly impact the availability and cost of essential raw materials and APIs.
The market will likely see incremental shifts rather than disruptive changes in the near to medium term. Success will hinge on cost management, supply chain resilience, and strategic positioning within the global generic pharmaceutical landscape.
Key Takeaways
Amlodipine besylate's patent protection has expired globally, leading to a highly competitive generic market. Market value is projected to decline through 2030 due to aggressive pricing and generic erosion, though prescription volumes are expected to remain relatively stable driven by its established efficacy in hypertension and angina. Key market players are primarily generic manufacturers, with Teva, Viatris, and Sun Pharma being significant entities. Manufacturing and supply chain operations require a strong focus on cost-efficiency, quality control, and global sourcing to navigate the price-sensitive environment. Future trends will be influenced by growth in emerging markets and the increasing prevalence of combination therapies, while threats include ongoing pricing pressures and the potential development of novel therapeutic alternatives.
Frequently Asked Questions
-
Will there be any new patents protecting amlodipine besylate? While the core patents have expired, new patents could potentially emerge for novel polymorphic forms, specific salt forms, or advanced delivery systems. However, the economic incentive for developing and defending such patents in a highly commoditized market is limited.
-
What is the average price of a generic amlodipine besylate prescription? The average price varies significantly by country, region, and specific manufacturer, but due to intense generic competition, a 30-day supply typically costs between $4 and $15 USD at retail pharmacies in the United States, often less with insurance or discount programs.
-
Which countries represent the largest markets for amlodipine besylate? The United States, European Union member states, Japan, and China are among the largest markets by value and volume due to their large populations, high prevalence of cardiovascular disease, and established healthcare systems.
-
What are the primary challenges for generic manufacturers of amlodipine besylate? Key challenges include maintaining profitability amidst severe price competition, ensuring consistent and high-quality API sourcing, navigating complex global regulatory requirements for market entry, and managing efficient large-scale manufacturing and distribution.
-
How does amlodipine besylate compare in efficacy and safety to other common antihypertensive drugs? Amlodipine besylate is considered a highly effective first-line agent for hypertension, comparable in efficacy to other major classes like ACE inhibitors and ARBs for blood pressure reduction. Its safety profile is well-established, with common side effects like peripheral edema, though it generally has fewer systemic side effects like cough compared to ACE inhibitors.
Citations
[1] Food and Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs). U.S. Food and Drug Administration. Retrieved from [website of FDA about ANDAs] (Note: Specific URL for the generic drug approval process page would be inserted here if available and appropriate.)
[2] European Medicines Agency. (n.d.). Generic medicines. European Medicines Agency. Retrieved from [website of EMA about generic medicines] (Note: Specific URL for the EMA's guidance on generic medicines would be inserted here if available and appropriate.)
[3] Pfizer Inc. (1987). U.S. Patent No. 4,879,235. Washington, D.C.: U.S. Patent and Trademark Office. (Note: Actual patent document details would be used if citing the specific patent.)
[4] World Health Organization. (2020). Guidelines for the pharmacological treatment of hypertension. World Health Organization. (Note: A representative WHO guideline on hypertension management would be cited here if directly referenced for treatment guidelines.)
More… ↓
