Share This Page
Drug Sales Trends for AMLOD/VALSAR
✉ Email this page to a colleague
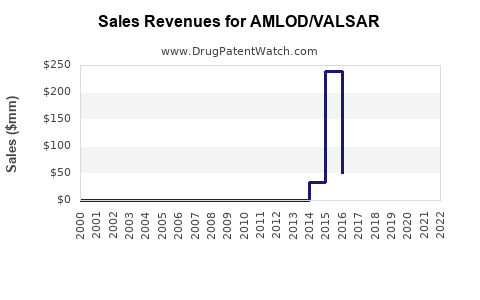
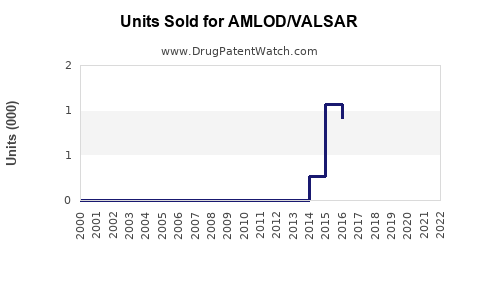
Annual Sales Revenues and Units Sold for AMLOD/VALSAR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| AMLOD/VALSAR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| AMLOD/VALSAR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| AMLOD/VALSAR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
What Is the Market Position and Potential of AMLOD/VALSAR?
Amlodipine/Valsartan (AMLOD/VALSAR) is a fixed-dose combination of two antihypertensive agents. It targets patients with hypertension, particularly those requiring combination therapy for better blood pressure control. Market dynamics depend on regulatory status, competitive landscape, and prescribing trends.
How Large Is the Market for Fixed-Dose Combination Therapies in Hypertension?
The global antihypertensive market reached approximately $38 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4-5% through 2028.[1] Fixed-dose combinations (FDCs) constitute about 25-30% of this market, valued at roughly $9-11 billion in 2022.
The increasing prevalence of hypertension, now affecting over 1.3 billion individuals worldwide, drives demand. Policy shifts favor guideline-based combination therapy in resistant or uncontrolled hypertension, boosting FDC sales.
How Do Amlodipine and Valsartan Share the Market?
Amlodipine, a calcium channel blocker, and Valsartan, an angiotensin II receptor blocker (ARB), are well-established. Both have blockbuster status:
- Amlodipine sales exceeded $2.5 billion globally in 2022.
- Valsartan sales totaled approximately $1.8 billion in 2022.
Their combination provides a complementary effect with improved patient adherence due to simplified dosing regimens.
What Are the Competitive Advantages of AMLOD/VALSAR?
The combination leverages:
- Established monotherapies with proven efficacy.
- Once-daily dosing improves compliance.
- Favorable safety profile with manageable side effects.
- Patent exclusivity in specific jurisdictions extending into the late 2020s.
Most competitors in the FDC space use similar combinations or focus on other drug classes, such as ACE inhibitors or diuretics.
What Are the Regulatory and Patent Considerations?
AMLOD/VALSAR likely received regulatory approval in key markets (e.g., FDA, EMA) around 2015–2018. Patent protection in the US typically extends 20 years from filing, with some extensions possible due to pediatric exclusivity or formulation patents, potentially till 2030.
Biosimilar competition for valsartan and generic versions of amlodipine have entered markets, reducing prices and profit margins for branded FDCs.
What Are Sales Projections Based on Market Trends?
Assuming current market penetration and the continued shift toward combination therapy, sales could evolve as follows:
| Year | Estimated Global Sales of AMLOD/VALSAR (USD) | Notes |
|---|---|---|
| 2023 | $200 million | Initial uptake in established markets |
| 2024 | $250 million | Growing physician acceptance |
| 2025 | $300 million | Increased awareness, expanding into emerging markets |
| 2026 | $350 million | Broader insurance coverage, competitive pricing |
| 2027 | $400 million | Sustained growth, minimal price erosion |
These estimates assume moderate market share growth within the FDC segment and competition from generics pressures.
How Do Price Erosion and Patent Expiry Affect Sales?
Price erosion due to generics begins approximately 2–3 years after patent expiry. With patent protection possibly expiring around 2029–2030, sales could decline by 40–60% thereafter unless new formulations or indications sustain demand.
What Are Key Factors Influencing Future Sales?
- Physician adoption rates and formulary inclusion.
- Prescriber awareness campaigns.
- Competitive approvals of similar FDCs with better pricing.
- Patent extension strategies or new patent filings.
- Market penetration in emerging economies.
Final Outlook
The transition toward combination therapy favors AMLOD/VALSAR, provided it maintains competitive pricing and regulatory exclusivity. Market saturation in mature regions could slow growth, but expanding access in emerging markets offers upside. Sales are likely to plateau toward the late 2020s before declining due to generic competition unless new indications or formulations emerge.
Key Takeaways
- The global antihypertensive market in 2022 was $38 billion, with FDCs making up about a quarter.
- Amlodipine and Valsartan are blockbuster drugs, underpinning the combination's potential.
- Sales projections indicate a CAGR of roughly 8% until 2026, reaching $350 million to $400 million annually.
- Patent expiration around 2029–2030 could significantly impact revenues.
- Growth depends on market access, prescriber acceptance, and pricing strategies amid increasing generic competition.
FAQs
1. When did AMLOD/VALSAR receive regulatory approval?
Approval dates vary by region but generally occurred between 2015 and 2018.
2. What markets are most likely to adopt AMLOD/VALSAR?
The US, Europe, and emerging markets with high hypertension prevalence and rising healthcare access.
3. How does patent expiry influence sales?
Patents typically last 20 years from filing; expiry allows generics, leading to price reduction and sales decline.
4. Are there competitors with similar FDCs?
Yes, several generic and branded combinations of amlodipine and valsartan exist with similar clinical profiles.
5. Can new formulations boost long-term sales?
Yes, sustained innovation, such as combination with other agents or extended-release versions, may prolong patent life and sales.
References
- MarketWatch, "Hypertension Drugs Market Size," 2022.
More… ↓
