Share This Page
Drug Sales Trends for AMITIZA
✉ Email this page to a colleague
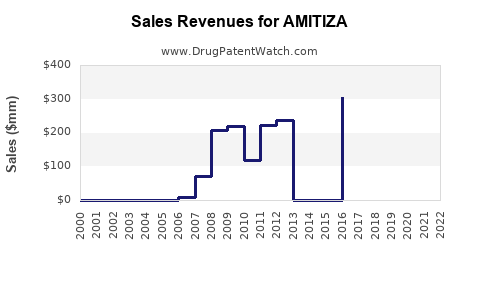
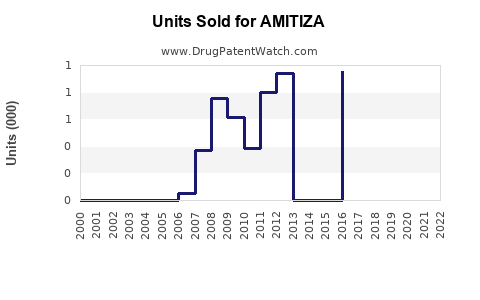
Annual Sales Revenues and Units Sold for AMITIZA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| AMITIZA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| AMITIZA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| AMITIZA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| AMITIZA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| AMITIZA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Amitiza: Patent Landscape and Market Projection
Amitiza (lubiprostone) is a prescription medication used to treat chronic idiopathic constipation (CIC), irritable bowel syndrome with constipation (IBS-C), and opioid-induced constipation (OIC). Its mechanism of action involves activating specific chloride channels in the intestinal epithelium, increasing fluid secretion and facilitating stool passage.
What is the Current Patent Status of Amitiza?
The primary patents covering amitiza have expired. The last of the core composition of matter patents expired in 2017 [1]. However, various formulation patents and method of use patents may still provide some degree of market exclusivity for specific applications or delivery methods.
- US Patent 6,020,373: This foundational patent for lubiprostone expired in 2017.
- Formulation Patents: Additional patents related to specific formulations of lubiprostone may have later expiration dates. For instance, some patents related to improved delivery systems or dosage forms could extend exclusivity for niche applications. However, these are generally less robust than composition of matter patents.
- Method of Use Patents: Patents covering the use of lubiprostone for specific indications, such as IBS-C or OIC, may have also expired or have limited remaining term. The initial FDA approval for CIC was in 2006, followed by IBS-C in 2008 and OIC in 2009.
The absence of strong patent protection for the original composition of matter allows for generic competition, significantly impacting the market dynamics for amitiza.
Who are the Key Manufacturers and Competitors?
Takeda Pharmaceuticals, the current marketer of amitiza in the United States, faces competition from generic versions of lubiprostone. The primary competition comes from:
- Generic Lubiprostone Manufacturers: Multiple pharmaceutical companies have launched generic versions of lubiprostone following patent expirations. These include manufacturers such as:
- Alembic Pharmaceuticals
- Aprazer Pharmaceuticals
- Breckenridge Pharmaceutical
- Impax Laboratories (now part of Amneal Pharmaceuticals)
- Par Pharmaceutical
- Sun Pharmaceutical
- Teva Pharmaceuticals
- Vellore Pharmaceutical
- Zydus Pharmaceuticals
These generic competitors offer lower-priced alternatives, directly impacting amitiza's market share and revenue.
What are the Sales Performance and Market Size of Amitiza?
Amitiza's sales have been affected by the availability of generic alternatives and the introduction of newer therapeutic agents for constipation. Takeda reported net sales for amitiza as follows:
- 2023: $411 million [2]
- 2022: $417 million [3]
- 2021: $404 million [4]
The sales figures indicate a relatively stable market, with slight fluctuations. However, this stability is in the context of a mature product facing significant genericization. The overall market for CIC, IBS-C, and OIC is substantial, driven by the high prevalence of these conditions.
Market Size of Related Conditions:
- Chronic Idiopathic Constipation (CIC): Affects approximately 15-20% of the adult population in developed countries.
- Irritable Bowel Syndrome with Constipation (IBS-C): Affects 5-10% of the adult population globally, with a significant portion experiencing the constipation-predominant subtype.
- Opioid-Induced Constipation (OIC): Occurs in 40-80% of patients taking chronic opioid therapy.
These large patient populations represent a considerable market opportunity, although competition for treatment is increasing.
What is the Projected Market Growth for Amitiza?
The market for amitiza is projected to experience low single-digit growth or decline in the coming years. This projection is primarily driven by:
- Generic Erosion: The continued presence and market penetration of generic lubiprostone will exert downward pressure on amitiza's sales volume and average selling price.
- Competition from Newer Therapies: The development and approval of novel treatments for constipation offer alternative options to patients and prescribers. These include:
- Guanylate cyclase-C (GC-C) agonists: Linzess (linaclotide) and Trulance (plecanatide) are established competitors with distinct mechanisms of action.
- Serotonin 5-HT4 receptor agonists: Prucalopride (available generically and under brand names like Motegrity) offers another therapeutic pathway.
- Dopamine D2 receptor antagonists: Valdoxan (agomelatine) has been explored in some regions for gastrointestinal motility disorders.
- Experimental compounds: Ongoing research aims to develop drugs with improved efficacy, tolerability, or novel mechanisms.
While the prevalence of constipation-related conditions remains high, the market share for older, off-patent drugs like amitiza is likely to diminish as newer, often more targeted or perceived as superior, therapies gain traction.
What are the Key Regulatory Considerations?
Amitiza is approved by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- FDA Approval:
- CIC: Approved in 2006.
- IBS-C: Approved in 2008.
- OIC: Approved in 2009.
- Labeling and Indication Expansion: Any new indications or significant label changes require rigorous clinical trials and regulatory review. The current indications for amitiza are well-established.
- Post-Marketing Surveillance: Like all pharmaceuticals, amitiza is subject to ongoing post-marketing surveillance to monitor for adverse events and ensure continued safety.
- Generic Drug Approval: Generic versions of amitiza must demonstrate bioequivalence to the reference listed drug, amitiza, and undergo regulatory review for quality and manufacturing standards.
Regulatory hurdles for new drug approvals, particularly for gastrointestinal disorders, remain high, requiring extensive clinical data to demonstrate safety and efficacy.
What are the Commercialization Strategies for Amitiza?
Takeda's commercialization strategy for amitiza, post-genericization, focuses on:
- Brand Loyalty and Physician Relationships: Leveraging established relationships with healthcare providers to maintain prescription volume for the branded product.
- Value-Based Messaging: Emphasizing the long-standing clinical experience and established efficacy profile of amitiza, particularly for patients who have responded well to the drug.
- Patient Support Programs: Offering programs to assist patients with access, adherence, and affordability of the branded medication.
- Targeted Marketing: Focusing on specific patient segments and prescriber groups where amitiza continues to hold a strong therapeutic position.
However, the ability to command premium pricing or significantly expand market share is constrained by the availability of lower-cost generic alternatives.
What is the Competitive Landscape of Constipation Treatments?
The market for constipation treatments is highly competitive and segmented by the underlying cause and mechanism of action.
Key Therapeutic Classes and Representative Drugs:
| Drug Class | Mechanism of Action | Representative Drug(s) | Approval Status (US) | Market Position Notes |
|---|---|---|---|---|
| Chloride Channel Activators | Stimulates chloride secretion, increasing luminal fluid. | Amitiza (lubiprostone) | CIC, IBS-C, OIC | Original compound, now facing generic competition. Established efficacy but newer agents offer different mechanisms. |
| Guanylate Cyclase-C (GC-C) Agonists | Activates GC-C, increasing cGMP, chloride/bicarbonate secretion, and fluid. | Linzess (linaclotide) | CIC, IBS-C | Significant market share, often considered a first-line option for IBS-C. Different efficacy and tolerability profile than lubiprostone. |
| Trulance (plecanatide) | CIC, IBS-C | Similar to linzess but with potential differences in efficacy and side effect profiles. | ||
| Serotonin 5-HT4 Receptor Agonists | Stimulates motilin release, increasing colonic transit. | Motegrity (prucalopride) | CIC | Approved for chronic idiopathic constipation. Offers a different mechanism focused on motility enhancement. Available as generic as well. |
| Osmotic Laxatives | Draws water into the colon. | Miralax (polyethylene glycol) | OTC/Rx | Widely used, generally safe, and effective for many. Over-the-counter availability limits branded prescription drug market capture. |
| Stimulant Laxatives | Stimulates nerve endings in the colon. | Senna, Bisacodyl | OTC | Long history of use, often for short-term relief. Not typically first-line for chronic conditions due to potential for dependence and side effects. |
| Dopamine D2 Receptor Antagonists | Reduces visceral hypersensitivity and improves motility. | Prucalopride (as a prokinetic agent) | Varies by region | Primarily used in Europe for prokinetic effects. Limited use in the US for constipation specifically compared to other agents. |
The competitive landscape necessitates ongoing evaluation of treatment algorithms and patient profiles to determine the most appropriate therapeutic option.
What are the Key Takeaways?
- Patent Expiration: The core patents for amitiza have expired, allowing for widespread generic competition.
- Market Stability with Decline Risk: Amitiza (branded) has shown sales stability in recent years, but this is under pressure from generic lubiprostone and newer therapies. Future growth is projected to be minimal or negative.
- Strong Generic Presence: Multiple manufacturers offer generic lubiprostone, significantly impacting the pricing and market share of the branded product.
- Competitive Market: The constipation treatment market is crowded, with newer agents like GC-C agonists and 5-HT4 agonists offering alternative mechanisms and often gaining market preference.
- Established Efficacy: Amitiza remains a treatment option with a known efficacy profile, particularly for patients who have previously responded well.
Frequently Asked Questions
1. What is the primary reason for the declining market share potential of branded amitiza?
The primary reason is the expiration of its key composition of matter patents, which has led to the introduction of numerous lower-cost generic alternatives.
2. How does amitiza compare to newer GC-C agonists like Linzess and Trulance in terms of mechanism of action?
Amitiza is a chloride channel activator, increasing intestinal fluid secretion. GC-C agonists like Linzess and Trulance also increase fluid secretion but do so by activating the guanylate cyclase-C receptor pathway, leading to increased cyclic guanosine monophosphate (cGMP).
3. Are there any remaining patent protections that could provide significant market exclusivity for amitiza?
While the primary patents have expired, there may be some remaining patent protections for specific formulations or method-of-use patents. However, these are unlikely to provide the same level of broad market exclusivity as composition of matter patents.
4. What are the main indications for which amitiza is currently prescribed?
Amitiza is prescribed for chronic idiopathic constipation (CIC), irritable bowel syndrome with constipation (IBS-C), and opioid-induced constipation (OIC).
5. How does the cost of branded amitiza typically compare to generic lubiprostone?
Branded amitiza is significantly more expensive than generic lubiprostone. The price difference is a major factor driving patient and payer preference towards generic versions.
Citations
[1] U.S. Patent 6,020,373. (1997). Takeda Pharmaceutical Company Limited. [2] Takeda Pharmaceutical Company Limited. (2023). Integrated Report 2023. [3] Takeda Pharmaceutical Company Limited. (2022). Integrated Report 2022. [4] Takeda Pharmaceutical Company Limited. (2021). Integrated Report 2021.
More… ↓
