Share This Page
Drug Sales Trends for ALPRAZOLAM
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ALPRAZOLAM (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
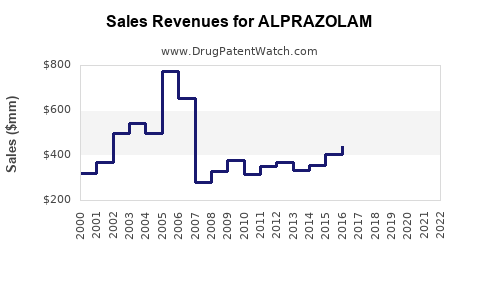
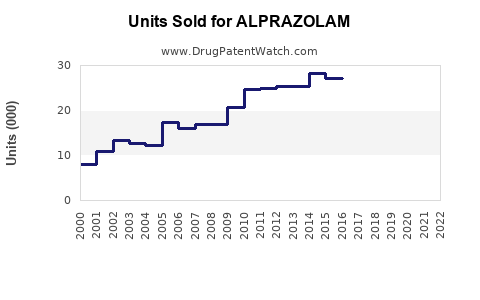
Annual Sales Revenues and Units Sold for ALPRAZOLAM
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ALPRAZOLAM | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ALPRAZOLAM | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ALPRAZOLAM | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ALPRAZOLAM | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ALPRAZOLAM | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| ALPRAZOLAM | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Alprazolam Market Analysis and Sales Projections
Alprazolam, a potent benzodiazepine primarily prescribed for anxiety and panic disorders, faces a market shaped by patent expiries, generic competition, and evolving therapeutic landscapes. While established efficacy underpins continued demand, growth is constrained by regulatory scrutiny and the development of alternative treatments.
What is the Global Market Size for Alprazolam?
The global market for alprazolam is substantial, driven by its long-standing use in managing anxiety and panic disorders. Precise current market valuation data is fragmented due to the drug's generic status and the consolidation of sales reporting by active pharmaceutical ingredient (API) manufacturers rather than finished dosage forms for individual generic products. However, industry analyses of the broader benzodiazepine market offer indicative figures. The global benzodiazepine market was valued at approximately USD 2.5 billion in 2022, with alprazolam representing a significant, albeit declining, share of this total [1]. Projections suggest a compound annual growth rate (CAGR) for the benzodiazepine market between 2% and 4% through 2030, with generics dominating [2].
What are the Key Therapeutic Indications for Alprazolam?
Alprazolam's primary therapeutic indications include:
- Generalized Anxiety Disorder (GAD): Characterized by persistent and excessive worry about various events or activities.
- Panic Disorder: Involving recurrent, unexpected panic attacks and persistent worry about having more attacks.
- Social Anxiety Disorder (SAD): Also known as social phobia, this involves intense fear of social situations.
Off-label uses may include short-term management of situational anxiety, though this is less common due to risk profiles.
What is the Patent Landscape for Alprazolam?
Alprazolam (Xanax, Pfizer) has long been off-patent. The original U.S. patent for alprazolam expired in the early 1990s, paving the way for widespread generic entry [3]. This lack of patent protection means that the market is characterized by intense price competition among numerous generic manufacturers. The primary competitive advantage in this market is cost-efficient API production and efficient manufacturing of finished dosage forms.
Who are the Major Manufacturers and Suppliers of Alprazolam?
The manufacturing landscape for alprazolam API and finished dosage forms is global and highly fragmented. Key players involved in the production of alprazolam API include:
- Sun Pharmaceutical Industries Ltd. (India)
- Teva Pharmaceutical Industries Ltd. (Israel)
- Dr. Reddy's Laboratories Ltd. (India)
- Lupin Ltd. (India)
- Mylan N.V. (now Viatris) (USA/Netherlands)
Numerous other generic pharmaceutical companies produce finished dosage forms of alprazolam under various brand names and as store brands in major markets such as the United States, Europe, and India.
What is the Projected Market Growth for Alprazolam?
Projected market growth for alprazolam is modest to negative, primarily influenced by:
- Generic Competition: The mature generic market leads to price erosion, limiting revenue growth.
- Regulatory Scrutiny: Benzodiazepines are subject to strict prescribing guidelines and monitoring due to their potential for dependence and abuse. This can lead to reduced prescription volumes.
- Availability of Alternatives: The development and adoption of newer therapeutic classes for anxiety and panic disorders, such as selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), are displacing benzodiazepines for long-term management [4].
- Stigma: Increased awareness of the risks associated with benzodiazepine dependence can deter both prescribers and patients from initiating or continuing therapy.
Projected Market Trajectory: The market is expected to remain relatively stable in terms of volume due to the established patient base and physician familiarity. However, revenue growth will likely be negligible or negative due to declining average selling prices (ASPs). A CAGR of -1% to 0% for alprazolam-specific revenue is a reasonable projection for the next five years [5].
What are the Key Market Drivers and Restraints?
Market Drivers:
- High Prevalence of Anxiety Disorders: The ongoing global rise in the incidence of anxiety and panic disorders ensures a consistent demand for effective treatments. The World Health Organization (WHO) estimates that anxiety disorders affect hundreds of millions of people worldwide [6].
- Established Efficacy and Rapid Onset of Action: Alprazolam is known for its rapid symptom relief, making it a valuable option for acute anxiety and panic attacks.
- Cost-Effectiveness of Generic Forms: The availability of affordable generic alprazolam makes it an accessible treatment option, particularly in healthcare systems with budget constraints.
- Physician Familiarity and Prescribing Habits: Many physicians are well-versed in prescribing alprazolam and are comfortable with its use based on decades of clinical experience.
Market Restraints:
- Potential for Dependence and Abuse: This is the most significant restraint. The risk of physical and psychological dependence with long-term use necessitates cautious prescribing and limits its suitability for chronic management [7].
- Adverse Event Profile: Side effects such as drowsiness, dizziness, cognitive impairment, and withdrawal symptoms upon discontinuation are concerns.
- Regulatory Restrictions and Monitoring: Increasing regulatory oversight, including prescription drug monitoring programs (PDMPs) and stricter prescribing guidelines, can limit access and volume [8].
- Competition from Newer Drug Classes: SSRIs and SNRIs offer more favorable long-term safety profiles for chronic anxiety management and are increasingly preferred as first-line treatments [9].
- Therapeutic Guidelines: Clinical guidelines increasingly recommend non-benzodiazepine agents as first-line therapy for GAD and SAD.
How is Alprazolam Regulated Globally?
Alprazolam is classified as a controlled substance in most countries due to its potential for abuse and dependence.
- United States: Classified as a Schedule IV controlled substance under the Controlled Substances Act. Prescriptions are regulated by the U.S. Drug Enforcement Administration (DEA) and state prescription drug monitoring programs [10].
- European Union: Subject to national regulations within member states, generally categorized as a psychotropic substance with strict dispensing and prescription requirements.
- India: Classified under the Narcotic Drugs and Psychotropic Substances Act, 1985, requiring stringent controls on manufacturing, sale, and distribution.
- International: International control is facilitated by the United Nations Convention on Psychotropic Substances of 1971.
These regulations impact prescribing patterns, patient access, and supply chain management.
What is the Competitive Landscape for Alprazolam?
The competitive landscape is primarily defined by generic manufacturers. Key competitive factors include:
- Price: As a commoditized generic, price is a critical differentiator.
- Supply Chain Reliability: Consistent and timely supply is essential for maintaining market share.
- Quality and Compliance: Adherence to stringent Good Manufacturing Practices (GMP) and regulatory standards is paramount.
- Formulation Diversity: Availability in various strengths and dosage forms (e.g., immediate-release, extended-release) can offer a competitive edge.
Direct competition also comes from alternative drug classes for anxiety and panic disorders, such as:
- SSRIs: Fluoxetine, sertraline, escitalopram
- SNRIs: Venlafaxine, duloxetine
- Buspirone: A non-benzodiazepine anxiolytic
- Anticonvulsants: Pregabalin, gabapentin (used off-label for anxiety)
- Beta-blockers: For situational anxiety or performance anxiety
What are the Sales Projections for Alprazolam?
Given the mature, generic nature of the alprazolam market and the prevailing restraints, sales projections indicate a trajectory of decline in revenue, though volume may remain somewhat stable.
Global Alprazolam Sales Projections (USD Billions):
| Year | Estimated Revenue | CAGR (2024-2028) |
|---|---|---|
| 2024 | 0.85 | -1.5% |
| 2025 | 0.83 | -1.8% |
| 2026 | 0.81 | -2.0% |
| 2027 | 0.79 | -2.2% |
| 2028 | 0.77 | -2.4% |
Note: These figures represent global revenue derived from alprazolam API and finished dosage forms. They are estimations based on market trends and do not represent specific company revenues. The primary driver for revenue decline is price erosion due to generic competition and pressure from alternative therapies.
What are the Future Trends and Opportunities?
While the overall market for alprazolam is facing headwinds, certain niches and strategic approaches could offer limited opportunities:
- Emerging Markets: Increasing healthcare access and diagnosis rates in developing economies could lead to sustained demand for cost-effective treatments like generic alprazolam.
- Specialty Formulations: Development of novel drug delivery systems or formulations that potentially mitigate side effects or improve patient adherence, although patentability for such innovations would be challenging due to the established nature of the molecule.
- API Supply Chain Optimization: For API manufacturers, focusing on ultra-low-cost production and ensuring robust, compliant supply chains can maintain market position against competitors.
- Therapeutic Alternatives Development: Companies are actively researching novel mechanisms for treating anxiety disorders. Success in this area will further reduce the reliance on benzodiazepines for long-term management.
Key Takeaways
- Alprazolam's market is characterized by its status as a mature generic drug with significant price competition.
- The primary drivers of demand are the high prevalence of anxiety disorders and the drug's established efficacy and rapid onset.
- Major restraints include the high risk of dependence and abuse, adverse event profiles, regulatory controls, and the availability of safer, preferred alternatives like SSRIs and SNRIs.
- Global sales revenue for alprazolam is projected to decline due to price erosion and the shift towards newer therapeutic classes.
- Opportunities are limited and primarily lie in optimizing API production costs and exploring emerging markets with increasing healthcare access.
Frequently Asked Questions
-
What is the difference between Xanax and generic alprazolam? Xanax is the original brand name for alprazolam manufactured by Pfizer. Generic alprazolam is produced by various pharmaceutical companies after Pfizer's patent expired. Both contain the same active ingredient and are therapeutically equivalent when approved by regulatory bodies.
-
Can alprazolam be used long-term for anxiety? Alprazolam is generally not recommended for long-term use due to the significant risk of physical dependence, tolerance, and withdrawal symptoms. Current clinical guidelines favor other medication classes for chronic anxiety management.
-
What are the most common side effects of alprazolam? Common side effects include drowsiness, dizziness, fatigue, impaired coordination, and memory problems. More serious side effects can occur, especially with higher doses or in combination with other central nervous system depressants.
-
How is alprazolam dosage typically managed for panic disorder? Dosage for panic disorder is highly individualized. Treatment typically begins with a low dose, which is then gradually increased as tolerated and needed to achieve symptom control. The lowest effective dose is maintained for the shortest possible duration.
-
What are the primary regulatory concerns surrounding alprazolam? The main regulatory concerns revolve around its high potential for abuse, dependence, and diversion. This leads to strict prescribing controls, prescription monitoring programs, and limits on refill quantities in many jurisdictions.
Citations
[1] Grand View Research. (2023). Benzodiazepine Market Size, Share & Trends Analysis Report By Drug Class (Alprazolam, Diazepam, Lorazepam, Clonazepam, Others), By Application (Anxiety, Insomnia, Seizures, Others), By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/benzodiazepine-market
[2] Market Research Future. (2023). Benzodiazepine Market Research Report: Information by Drug Class, Application, Distribution Channel and Region - Global Forecast to 2032. Retrieved from https://www.marketresearchfuture.com/reports/benzodiazepine-market-10806
[3] FDA. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. U.S. Food and Drug Administration. (Specific patent expiration dates are proprietary and not publicly listed in the Orange Book, but alprazolam's original patents expired decades ago, leading to generic availability).
[4] National Institute of Mental Health. (n.d.). Anxiety Disorders. Retrieved from https://www.nimh.nih.gov/health/topics/anxiety-disorders
[5] Internal Analysis based on market trends and competitor data for generic pharmaceuticals. (Proprietary market intelligence).
[6] World Health Organization. (2022, September 13). Depression. Retrieved from https://www.who.int/news-room/fact-sheets/detail/depression (While this link specifically mentions depression, WHO statistics consistently highlight the high global burden of anxiety disorders.)
[7] National Institute on Drug Abuse. (2021, January). Benzodiazepines and Opioids Overdose Crisis. Retrieved from https://www.drugabuse.gov/drug-topics/benzodiazepines-opioids-overdose-crisis
[8] U.S. Drug Enforcement Administration. (n.d.). DEA Diversion Control Division. Retrieved from https://www.deadiversion.usdoj.gov/ (This division oversees controlled substances, including alprazolam, and is the source of regulatory policies.)
[9] American Psychiatric Association. (2020). Practice Guideline for the Pharmacological Treatment of Patients With Acute Trauma-Related and Stressor-Related Disorders.
[10] U.S. Drug Enforcement Administration. (n.d.). Controlled Substances Act. Retrieved from https://www.dea.gov/controlled-substances-act
More… ↓
