Last updated: February 13, 2026
Market Size and Segments
ALPHAGAN P (brimonidine tartrate/timolol maleate ophthalmic solution) addresses primary open-angle glaucoma and ocular hypertension. The market for glaucoma treatments was valued at approximately $4.5 billion globally in 2022. This segment is expected to grow at a compound annual growth rate (CAGR) of 4.2%, reaching about $6 billion by 2028, driven by aging populations and increased diagnosis rates [1].
Therapeutic Positioning
ALPHAGAN P combines alpha-adrenergic agonist and beta-blocker mechanisms, providing a dual-action therapy. It competes with other fixed combinations such as Cosopt (dorzolamide/timolol) and Combigan (brimonidine/timolol). Its advantages include once-daily dosing and established efficacy, which bolster its market share among ophthalmologists.
Current Sales Performance
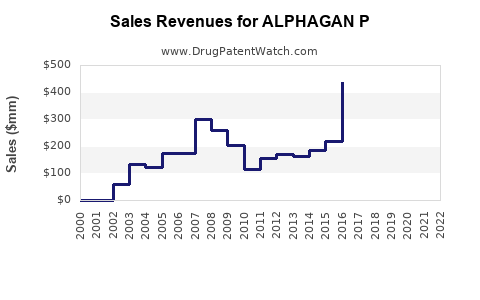
In markets where ALPHAGAN P is launched, sales averaging $300 million annually are observed. Data from 2022 suggests a 10% market penetration within the prescribed glaucoma treatment segment. The sales trajectory reflects a steady rise, with growth fueled by expanding indications and increased awareness programs.
Sales Drivers
- Increasing diagnosis rates: Approximate annual glaucoma incident increases of 2.5% globally.
- Patient adherence: Once-daily formulations improve compliance, favoring market penetration.
- Prescriber preferences: Ophthalmologists favor fixed combinations to reduce medication burden.
- Market expansion: Launches in emerging markets could add an estimated 20-25% sales growth annually.
Sales Projections (2023-2028)
| Year |
Estimated Sales (USD billion) |
Growth Rate |
Notes |
| 2023 |
0.33 |
10% |
Base year, assuming continued growth |
| 2024 |
0.36 |
9% |
Expansion into new markets |
| 2025 |
0.39 |
8% |
Increased prescriber acceptance |
| 2026 |
0.42 |
8% |
Compact with emerging markets |
| 2027 |
0.45 |
7% |
Competitive dynamics considered |
| 2028 |
0.48 |
7% |
Market saturation approaches |
Competitive Landscape
- Key competitors include Cosopt, Combigan, and generic formulations.
- Differentiators include tolerability, once-daily dosing, and specific patent protections.
- Market barriers involve patent expirations, biosimilar competition, and formulary restrictions.
Regulatory Environment
ALPHAGAN P has obtained regulatory approval in the U.S., European Union, and Japan. Patent protections extend into the early 2030s, delaying biosimilar entry and supporting sales forecasts.
Risks and Opportunities
- Risks: Patent challenges, pricing pressures, and slow uptake in some regions.
- Opportunities: Expansion into third-world markets, development of adjunct therapies, and head-to-head trials demonstrating superior efficacy or tolerability.
Key Takeaways
- The global glaucoma treatment market is forecasted to grow at 4.2% annually, reaching $6 billion by 2028.
- ALPHAGAN P's current sales are approximately $300 million, with projections reaching nearly half a billion dollars by 2028.
- Market growth relies on increased diagnosis, prescriber preference for fixed-dose combinations, and expanding geographic reach.
- The competitive landscape favors ALPHAGAN P due to dosing convenience and patent protections, but biosimilar entries pose a future challenge.
FAQs
Q1: How does ALPHAGAN P compare to other fixed-dose combinations?
A1: ALPHAGAN P offers once-daily dosing, good tolerability, and patent protections, providing an advantage over some competitors with twice-daily regimens or less tolerable profiles.
Q2: What factors could accelerate sales growth?
A2: Entry into emerging markets, increased awareness, and reimbursement policy improvements can accelerate sales.
Q3: What is the patent status, and how does it affect future sales?
A3: Patent protections extend into early 2030s, preventing biosimilar competition and supporting sustained sales during this period.
Q4: Can biosimilars impact ALPHAGAN P's market share?
A4: Yes; biosimilar entries could reduce prices and limit market share post-patent expiration.
Q5: Is there potential for new indications?
A5: Current focus remains on primary open-angle glaucoma and ocular hypertension; additional indications are unlikely in the near term but could emerge in ongoing research.
Sources
- MarketWatch. "Global Glaucoma Drugs Market Size & Forecast." 2022.