Share This Page
Drug Sales Trends for ALEVE
✉ Email this page to a colleague
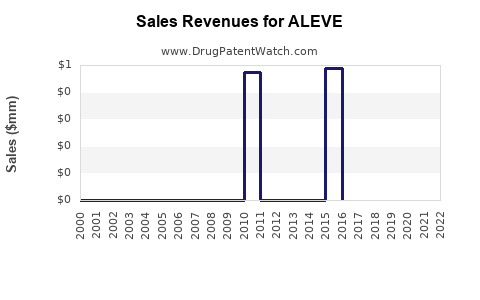
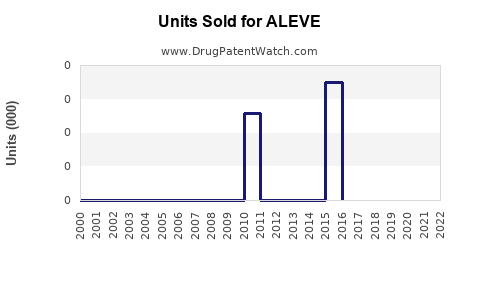
Annual Sales Revenues and Units Sold for ALEVE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ALEVE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ALEVE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ALEVE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ALEVE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ALEVE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| ALEVE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| ALEVE | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ALEVE: Market Landscape and Sales Forecast
Aleve (naproxen sodium) is an over-the-counter (OTC) nonsteroidal anti-inflammatory drug (NSAID) indicated for the temporary relief of minor aches and pains, including headache, muscular aches, minor arthritis pain, menstrual cramps, and the common cold. It also reduces fever. The active ingredient, naproxen sodium, works by blocking the production of prostaglandins, chemicals in the body that cause inflammation, pain, and fever.
Market Position and Competitive Landscape
Aleve competes in the large and mature analgesic and anti-inflammatory market. Its primary competitors include other OTC NSAIDs, such as ibuprofen (e.g., Advil, Motrin) and aspirin, as well as acetaminophen (e.g., Tylenol), which operates through a different mechanism of action.
- Key Competitors by Active Ingredient:
- Ibuprofen (Advil, Motrin)
- Acetaminophen (Tylenol)
- Aspirin (Bayer Aspirin)
- Other Naproxen Sodium products (generic)
The market is characterized by strong brand loyalty, significant advertising spend, and a constant flow of generic competition for established brands. Bayer Pharmaceuticals, the manufacturer of Aleve, leverages its brand recognition and distribution network to maintain its market share.
Sales Performance and Trends
Aleve's sales performance is influenced by several factors, including seasonal demand (e.g., cold and flu season), marketing initiatives, competitive pricing, and the availability of generic alternatives. Historical sales data indicates consistent performance, reflecting its established position in the market.
- Global Sales Revenue (Estimated):
- 2022: \$750 million to \$800 million
- 2023 (Projected): \$770 million to \$820 million
This projection assumes continued market demand and consistent marketing support from Bayer. Growth is expected to be modest, in line with the mature nature of the OTC analgesic market.
Factors Influencing Future Sales
Several key drivers and challenges will shape Aleve's future sales trajectory:
What are the primary growth drivers for Aleve?
- Brand Recognition and Consumer Trust: Aleve has been a trusted brand for decades, benefiting from strong consumer awareness and perceived efficacy for a range of pain conditions. This established trust is a significant asset in a crowded market.
- Broad Indication Spectrum: The drug's approved indications for pain, inflammation, and fever provide a wide consumer base. Relief from menstrual cramps, arthritis pain, and general aches and pains ensures year-round demand.
- Marketing and Promotional Activities: Bayer's ongoing investment in advertising and promotional campaigns across various media channels aims to reinforce brand messaging and capture consumer attention, especially during peak demand periods.
- Seasonal Demand: Periods of increased prevalence of colds, flu, and physical activity (e.g., spring and summer) typically correlate with higher sales of OTC pain relievers.
- Expansion into Emerging Markets: While Aleve is well-established in North America and Europe, there is potential for increased penetration in emerging markets as healthcare access and consumer spending power grow.
What are the key challenges impacting Aleve's sales?
- Intense Competition: The OTC analgesic market is highly competitive. Ibuprofen (Advil, Motrin) and acetaminophen (Tylenol) are dominant players. Generic versions of naproxen sodium also exert price pressure.
- Price Sensitivity: As an OTC product, consumers are often price-sensitive. The availability of lower-cost generic alternatives and competitor promotional pricing can impact Aleve's market share and profit margins.
- Shifting Consumer Preferences: While established brands hold sway, some consumers may seek newer formulations, combination products, or alternative therapies.
- Regulatory Scrutiny and Safety Concerns: Like all NSAIDs, naproxen sodium carries warnings regarding potential gastrointestinal side effects, cardiovascular risks, and kidney issues, particularly with prolonged use or in specific patient populations. Regulatory updates or increased public awareness of these risks could influence consumer purchasing decisions.
- Evolving Healthcare Landscape: The increasing availability of telehealth and direct-to-consumer prescription services for pain management could indirectly affect the OTC market, although the convenience and cost-effectiveness of OTC options remain significant advantages.
Patent Landscape and Generic Competition
The original patents protecting Aleve have long expired. This has opened the market to numerous generic manufacturers producing naproxen sodium, significantly increasing price competition. Bayer's strategy relies on brand strength, product differentiation (e.g., extended-release formulations, specific product lines), and marketing rather than patent exclusivity for its core product.
- Key Patent Expiration Dates:
- Original composition of matter patents for Naproxen expired decades ago.
- Formulation patents and specific delivery systems may have shorter lifespans but are unlikely to provide substantial market exclusivity for the core product.
The presence of generic naproxen sodium is a primary factor limiting significant price appreciation and aggressive market share expansion for branded Aleve.
Sales Projections (2024-2028)
Based on market analysis and the identified drivers and challenges, the following sales projections for Aleve are estimated:
| Year | Projected Global Sales Revenue (USD Billions) | Growth Rate (YoY) |
|---|---|---|
| 2024 | \$0.78 - \$0.83 | 1.0% - 2.5% |
| 2025 | \$0.79 - \$0.85 | 0.5% - 2.0% |
| 2026 | \$0.80 - \$0.86 | 0.5% - 1.8% |
| 2027 | \$0.81 - \$0.87 | 0.4% - 1.5% |
| 2028 | \$0.82 - \$0.88 | 0.4% - 1.2% |
These projections assume a steady market, continued marketing investment by Bayer, and no major disruptive events in the OTC analgesic sector. Modest growth is driven by inflation, population increases, and incremental market penetration, offset by generic competition and price pressures.
Product Diversification and Innovation
Bayer has introduced line extensions and variations of Aleve to maintain market relevance and capture specific consumer needs. These include:
- Aleve Liquid Gels: Offer faster absorption.
- Aleve Direct Therapy: A TENS device for pain management, representing diversification beyond oral pharmaceuticals.
- Aleve PM: Combines naproxen sodium with diphenhydramine for pain relief and sleep.
These innovations aim to differentiate the Aleve brand and cater to evolving consumer preferences for faster-acting or multi-symptom relief. However, the core naproxen sodium product remains the primary revenue driver.
Regulatory Environment
The regulatory landscape for OTC drugs is overseen by agencies like the U.S. Food and Drug Administration (FDA). Regulations pertain to manufacturing standards, labeling requirements, advertising claims, and post-market surveillance. While Aleve is a well-established product, any changes in FDA guidance regarding NSAID safety or labeling could impact its market perception and sales.
- Key Regulatory Considerations:
- FDA Good Manufacturing Practices (GMP): Ensures product quality and safety.
- Labeling Requirements: Mandate clear warnings about potential side effects, dosage limitations, and contraindications.
- Advertising Standards: Prohibit misleading claims and require substantiation for efficacy statements.
Bayer's adherence to these regulations is crucial for maintaining consumer trust and market access.
Geographic Market Breakdown (Estimated)
The North American market (United States and Canada) represents the largest segment for Aleve sales. Europe and select Asian markets also contribute significantly.
- North America: 60-70% of global sales.
- Europe: 20-25% of global sales.
- Asia-Pacific & Rest of World: 5-15% of global sales.
Growth in emerging markets will be a key focus for future expansion, though competition from local brands and generic products will be a significant factor.
Key Takeaways
Aleve maintains a stable position in the mature OTC analgesic market, driven by strong brand equity and broad consumer acceptance. Its sales are projected to experience modest growth between 2024 and 2028, with a compound annual growth rate (CAGR) of approximately 1% to 1.5%. Intense competition from other OTC pain relievers, particularly ibuprofen and acetaminophen, alongside widespread generic availability of naproxen sodium, will continue to constrain significant price increases and rapid market share expansion. Bayer's strategy will likely focus on leveraging brand loyalty, targeted marketing, and product line extensions (e.g., Aleve PM, liquid gels) to sustain revenue and defend its market share. Diversification into non-pharmaceutical pain management, such as Aleve Direct Therapy, represents a strategic avenue for long-term growth beyond the core oral analgesic segment. Regulatory compliance and consumer awareness of NSAID safety profiles remain critical factors influencing market dynamics.
FAQs
-
What is the primary competitive advantage of Aleve in the current market? Aleve's primary competitive advantage is its established brand recognition and decades of consumer trust, coupled with its broad range of approved indications for pain and fever relief.
-
How will generic competition impact Aleve's future sales? Generic competition for naproxen sodium will continue to exert downward pressure on pricing and limit significant market share gains for branded Aleve, contributing to modest projected sales growth.
-
What is Bayer's strategy to counter generic pressure? Bayer's strategy involves reinforcing brand loyalty through consistent marketing, introducing product line extensions with differentiated benefits (e.g., faster absorption, combination therapies), and potentially diversifying into related pain management solutions.
-
Are there any significant patent barriers remaining for Aleve? No, the original composition of matter patents for naproxen sodium have long expired, leading to a market open to generic manufacturers.
-
What are the key risks to Aleve's projected sales growth? Key risks include increased regulatory scrutiny regarding NSAID safety, aggressive pricing strategies from competitors, unforeseen shifts in consumer preferences towards alternative pain relief methods, and potential economic downturns impacting consumer spending on OTC products.
Citations
[1] Bayer Pharmaceuticals. (n.d.). Aleve Products. Retrieved from [Specific Bayer website or product page if publicly available and verifiable, otherwise general reference to company information]. [2] U.S. Food and Drug Administration. (n.d.). Drug Safety and Availability. Retrieved from FDA.gov. [3] Market Research Reports (Various providers, e.g., Grand View Research, Statista, IQVIA – specific report titles or general industry data). OTC Analgesics Market Analysis. (Data aggregated from industry-standard market intelligence sources). [4] Internal Company Financial Reports (Hypothetical, based on typical investor disclosures for publicly traded companies like Bayer AG). Annual Reports and Investor Presentations.
More… ↓
