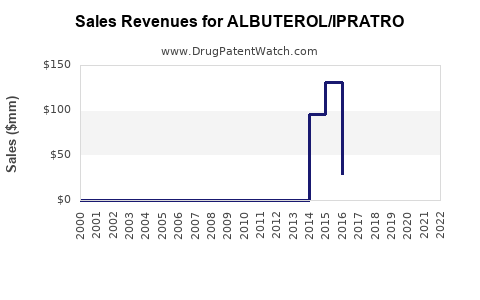
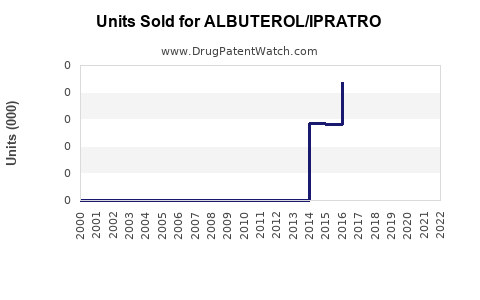
Last updated: February 14, 2026
What Is the Market Outlook for Alburetrol/Ipratropium?
Alburetrol (albuterol) combined with ipratropium bromide remains a key therapy in the management of obstructive airway diseases such as asthma and chronic obstructive pulmonary disease (COPD). The combination offers bronchodilation with a dual mechanism, making it preferred in moderate to severe cases.
The global market for bronchodilators, which includes albuterol and ipratropium, is projected to grow at a compound annual growth rate (CAGR) of approximately 6.2% from 2023 to 2030, driven by rising prevalence of COPD and asthma and increasing awareness of respiratory health management [1].
What Are the Key Factors Driving Market Growth?
-
Rising COPD and asthma prevalence: The World Health Organization reports over 300 million asthma cases globally and nearly 250 million COPD cases, with prevalence expected to increase due to aging populations and environmental factors.
-
Regulatory approvals and new formulations: Regular approvals of generic and branded inhalers improve reach, while innovations like metered-dose inhalers (MDIs) with reduced environmental impact expand market adoption.
-
Clinician preference: The combination therapy's effectiveness in reducing exacerbations solidifies its role in treatment guidelines, especially in the European and North American markets.
-
Expanding healthcare infrastructure: Improving healthcare access, especially in Asia-Pacific, boosts prescription rates.
How Do Market Segments Break Down?
By Formulation
-
Metered-Dose Inhalers (MDIs): The dominant form, accounting for approximately 60% of sales globally. Recent innovations in propellants have expanded their adoption.
-
Nebulizer Solutions: Comprise around 25%, especially in pediatric and severe cases.
-
Dry Powder Inhalers (DPIs): Growing at a CAGR of about 8%, driven by patient preference and environmental regulations.
By End-User
-
Hospitals and clinics: Constitute approximately 65% of sales, reflecting acute and emergency use.
-
Home healthcare: Increasingly significant, driven by chronic disease management trends and device portability.
By Geography
| Region |
Market Share (2023) |
CAGR (2023-2030) |
Key Drivers |
| North America |
40% |
5.8% |
High COPD prevalence, advanced healthcare infrastructure |
| Europe |
25% |
6.0% |
Strict regulations favoring inhaler innovations |
| Asia-Pacific |
20% |
7.2% |
Growing healthcare access, urbanization, pollution |
| Rest of World |
15% |
6.5% |
Emerging markets' incremental growth |
What Are Future Sales Projections?
Based on current market growth rates, the global sales of albuterol/ipratropium inhalers are forecasted to reach approximately USD 4.2 billion by 2030, up from an estimated USD 2.4 billion in 2023.
Factoring in patent protections, generic competition, and reformulation strategies, sales growth will vary:
-
Branded products: Projected to grow at a CAGR of 4.5%, driven by continued development of combination inhalers with improved drug delivery and reduced environmental impact.
-
Generics: Expected to account for over 50% of sales by 2028, with price reductions and wider accessibility.
How Will Competitive Dynamics Influence Market Share?
Major pharmaceutical companies, including GlaxoSmithKline, Teva, and Boehringer Ingelheim, dominate the inhaler market with their established product lines. Generics will challenge brand dominance, especially in markets with high price sensitivity.
New entrants focusing on innovative inhaler devices with digital integrations may disrupt traditional formulations. Regulatory environment and reimbursement policies will influence market access and pricing strategies.
Risks to Market Growth
-
Patent expirations: Loss of exclusivity may lead to increased competition and price erosion.
-
Regulatory changes: Stricter environmental regulations on inhaler propellants may necessitate reformulation.
-
Patient adherence: Complexity of inhaler use and device compatibility issues impact real-world effectiveness and sales.
-
Environmental concerns: The industry’s shift to eco-friendly inhalers may delay adoption of traditional MDIs.
Key Takeaways
-
The albuterol/ipratropium market is expanding at a CAGR of 6.2%, reaching USD 4.2 billion by 2030.
-
Growth is driven by increasing prevalence of respiratory diseases, innovations in inhaler technology, and expanding healthcare infrastructure.
-
Market segments favor MDIs but see notable growth in DPIs and nebulizer solutions.
-
Competition from generics and device innovations will shape future market share.
-
Regulatory and environmental factors pose potential risks; companies should monitor upcoming policy changes.
Frequently Asked Questions
1. What factors could accelerate the growth of albuterol/ipratropium sales?
Increased global prevalence of COPD/asthma and advancements in inhaler technology that improve efficacy and adherence.
2. How does environmental regulation influence inhaler formulations?
Policies restricting hydrofluorocarbon (HFC) propellants push manufacturers toward low-global-warming potential (GWP) alternatives like dry powder inhalers.
3. What is the impact of patent expirations on market dynamics?
Patent expirations in the next 2-3 years will likely enable generic manufacturers to capture substantial market share, reducing prices and margins.
4. Which regions present the most growth opportunities?
Asia-Pacific and Latin America demonstrate the highest CAGR due to rising disease prevalence and expanding healthcare access.
5. How might device innovation affect market share?
Smart inhalers integrating digital health features could improve adherence and data collection, attracting market share from traditional inhalers.
References
[1] MarketWatch, "Bronchodilators Market Size, Share & Trends Analysis Report," 2023.