Share This Page
Drug Sales Trends for ADVAIR DISKUS
✉ Email this page to a colleague
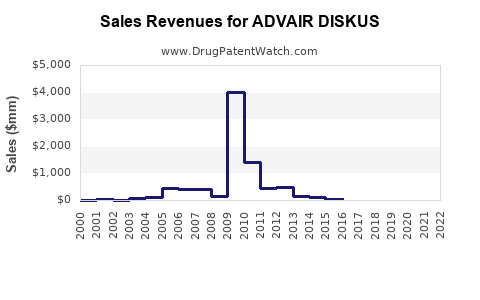
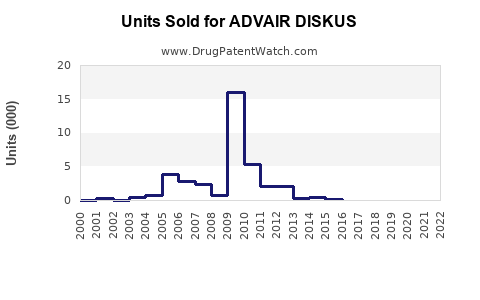
Annual Sales Revenues and Units Sold for ADVAIR DISKUS
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ADVAIR DISKUS | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ADVAIR DISKUS | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ADVAIR DISKUS | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Advair Diskus
Advair Diskus, a combination inhaler containing fluticasone propionate and salmeterol xinafoate, is a leading treatment for asthma and chronic obstructive pulmonary disease (COPD). Its market position is driven by its established efficacy, regulatory approvals, and clinical guidelines. The drug generated approximately $4.1 billion globally in sales in 2022.
Market Size and Growth Dynamics
Global Respiratory Disease Market
The global respiratory disease treatment market was valued at $35 billion in 2022 and is projected to reach $50 billion by 2030, growing at a compound annual growth rate (CAGR) of around 4.8%. Growth drivers include increased prevalence of asthma and COPD, aging populations, and advancements in inhaler technologies.
Competitive Landscape
Key competitors include:
- Symbicort (AstraZeneca/Bristol-Myers Squibb)
- Advair's successor brands and generics
- Breo Ellipta (GSK)
- Dulera (AstraZeneca)
Advair's market share declined after patent expiry in multiple territories but remains significant, especially in the U.S. due to existing formulary and physician familiarity.
Sales Projections
Historical Sales Trends
| Year | Global Sales (USD billions) |
|---|---|
| 2018 | 5.2 |
| 2019 | 4.9 |
| 2020 | 4.5 |
| 2021 | 4.3 |
| 2022 | 4.1 |
Sales declined post-patent expiry but stabilized through continued prescriptions and generic competition.
Short to Mid-term Forecast (2023-2027)
Assuming increased adoption in emerging markets and product lifecycle management, sales are projected to grow modestly post-2023, averaging around 3% annually:
| Year | Estimated Global Sales (USD billions) |
|---|---|
| 2023 | 4.2 |
| 2024 | 4.3 |
| 2025 | 4.4 |
| 2026 | 4.5 |
| 2027 | 4.6 |
Key Factors Impacting Sales
- Patent expirations: Generic versions entered several markets, reducing prices.
- Regulatory approvals: New indications or formulations may expand use.
- Market penetration: Growth in emerging regions can offset declines in mature markets.
- Pricing strategies: Discounting and value-based pricing influence volume.
Opportunities and Risks
Opportunities
- Expansion in Asia-Pacific and Latin America due to rising asthma and COPD prevalence.
- Introduction of new formulations or fixed-dose combinations to improve adherence.
- Digital health integration for adherence monitoring.
Risks
- Loss of market share to generics and alternative therapies.
- Regulatory delays or restrictions.
- Market saturation in developed countries.
Regulatory and Policy Influence
- US FDA approval renewed for additional indications in 2021.
- European Medicines Agency monitors inhaler safety; recent updates include concerns over rare side effects.
- Pricing reforms, particularly in Europe and parts of Asia, threaten profit margins.
Conclusion
Advair Diskus remains a significant product within the respiratory therapy market. While patent expiries have diminished its sales growth potential, new market penetration, technological improvements, and formulary management sustain its revenue streams in the medium term. Future sales depend heavily on regulatory approvals, competitive dynamics, and regional market development.
Key Takeaways
- Global sales peaked at $5.2 billion in 2018, declined thereafter, and stabilized around $4.1 billion in 2022.
- The respiratory treatment market is projected to grow to $50 billion by 2030, supporting moderate sales growth for established products like Advair.
- Market share erosion due to generics requires strategic management, including innovation and market expansion.
- Emerging markets present growth opportunities, offsetting declines in mature regions.
- Regulatory developments could influence sales trajectory and market accessibility.
FAQs
1. How has patent expiry affected Advair Diskus sales?
Patent expiry in key markets led to generic competition, reducing prices and overall sales. This contributed to the decline from peak revenues of $5.2 billion in 2018.
2. What markets are emerging as growth opportunities?
The Asia-Pacific and Latin American regions show rising prevalence of respiratory conditions, increased healthcare infrastructure, and lower generic penetration, creating growth opportunities.
3. Are there significant upcoming regulatory changes that will impact Advair?
Regulatory agencies are monitoring safety updates and may influence prescribing practices through safety communications or restrictions, impacting sales indirectly.
4. Can new formulations or indications revitalize sales?
Yes, FDA approvals for additional indications or innovative inhaler formulations could expand market share and improve adherence, revitalizing revenue streams.
5. What is the competitive outlook for Advair?
The competitive landscape includes newer inhalers like Breo Ellipta, which are gaining market share due to favorable safety profiles and dosing convenience. Generics pose a persistent threat, though.
References
[1] MarketWatch. (2022). Respiratory drugs market analysis.
[2] IQVIA. (2022). Global pharmaceutical market data.
[3] European Medicines Agency. (2022). Inhaler safety updates.
[4] U.S. Food and Drug Administration. (2021). Advair Diskus approval renewal.
More… ↓
