Last updated: February 13, 2026
What Is the Current Market Position of ADVAIR DISKU?
ADVAIR DISKU, an inhaled corticosteroid and long-acting beta-agonist combination therapy, is marketed by GlaxoSmithKline. It targets asthma and chronic obstructive pulmonary disease (COPD). The medication's formulation includes fluticasone furoate and vilanterol.
As of 2022, ADVAIR DISKU has maintained a significant presence within the respiratory drug market, especially in markets with high prevalence of asthma and COPD, such as North America and Europe. It has secured a considerable share due to its once-daily dosing convenience and established efficacy.
How Does ADVAIR DISKU Fit Into the Global Respiratory Market?
The global respiratory disease market was valued at approximately USD 24 billion in 2022, with asthma drugs accounting for around USD 9 billion and COPD drugs around USD 15 billion (source: IQVIA, 2022). ADVAIR DISKU is positioned within the inhaled corticosteroid/long-acting beta-agonist (ICS/LABA) segment, representing roughly 20-25% of the total asthma/COPD treatment market, depending on regional preferences.
Key competitors include:
- Symbicort (AstraZeneca/Boehringer Ingelheim)
- DULERA (Merck)
- LABA/LAMA fixed-dose combinations
ADVAIR DISKU's registration in multiple countries, including the United States, European Union member states, and select Asian markets, ensures its continued relevance.
What Are the Sales Trends for ADVAIR DISKU?
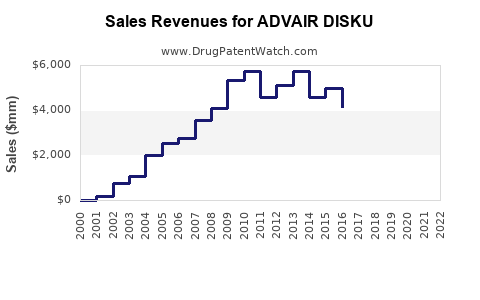
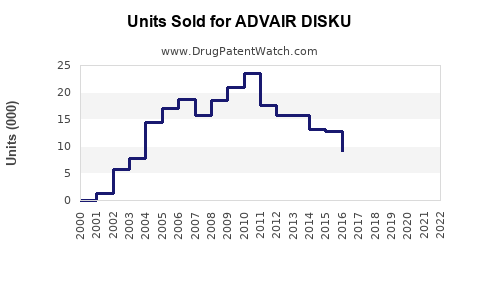
GSK reports that ADVAIR DISKU generated approximately USD 2.5 billion globally in 2022, representing a decline of 3% from USD 2.58 billion in 2021. The decline primarily reflects increased competition and patent expirations in certain markets.
Regional sales breakdown (2022):
| Region |
Sales (USD millions) |
Market Share |
Notes |
| North America |
1,200 |
~48% |
Largest market, high adoption rates |
| Europe |
850 |
~34% |
Mature market, growing generic competition |
| Asia-Pacific |
350 |
~14% |
Rapid growth, expanding access |
| Rest of World |
100 |
~4% |
Emerging markets, increasing awareness |
What Future Sales Projections Are Possible for ADVAIR DISKU?
Sales projections depend on factors including patent status, regulatory environment, competitive landscape, and healthcare policy shifts.
Short-term Outlook (2023-2025)
- Slight decline expected due to patent expiration in key markets (US patent expired in 2022 for certain formulations).
- Competition from generic equivalents and biosimilars may pressure pricing.
- Growth in Asia-Pacific markets can offset declines in mature markets.
Long-term Outlook (2025 and beyond)
- Stable or modest growth forecasted if GSK successfully introduces next-generation formulations.
- Market share could increase if GSK expands into emerging markets and gains regulatory approval for new indications.
- Market entry of new competitors with improved delivery mechanisms may challenge existing positions.
Assumed Sales Trajectory
| Year |
Estimated Global Sales (USD millions) |
Assumptions |
| 2023 |
USD 2.2 billion |
Patent expiries, intensified competition |
| 2024 |
USD 2.1 billion |
Continued competition, broader adoption in Asia |
| 2025 |
USD 2.3 billion |
Market expansion, new formulations or indications |
What Are the Market Risks and Opportunities?
Risks:
- Patent expiry leading to generic erosion.
- Regulatory hurdles in developing markets.
- Competitive products with improved efficacy or delivery systems.
Opportunities:
- Growth in COPD prevalence globally.
- Expansion into new regions, particularly in Asia and Latin America.
- Development of combination therapies with new molecules or delivery devices.
Key Takeaways
- ADVAIR DISKU is a leading ICS/LABA used for asthma and COPD, with approximately USD 2.5 billion in global sales in 2022.
- Its market share is concentrated in North America and Europe, with emerging markets showing growth potential.
- Sales are forecasted to decline modestly in the short term due to patent expirations and competition but may stabilize or grow with expanded market access.
- The medication's future depends on regulatory strategies, competitive innovations, and evolving healthcare policies.
FAQs
Q1: When did the patent expiration for ADVAIR DISKU occur?
A1: The US patent for certain ADVAIR DISKU formulations expired in 2022; international patent timelines vary.
Q2: What competitive advantages does ADVAIR DISKU hold?
A2: Once-daily dosing, established efficacy, and robust clinical data contribute to its competitive edge.
Q3: How does ADVAIR DISKU compare pricing-wise with competitors?
A3: Pricing varies regionally; generic pressures have led to reductions in some markets.
Q4: Are there new formulations or delivery mechanisms under development for ADVAIR DISKU?
A4: GSK continues to explore improved device designs and combination therapies, but specific pipeline updates are typically proprietary.
Q5: What factors most significantly influence the future sales of respiratory drugs like ADVAIR DISKU?
A5: Patent statuses, regulatory approvals, competitive innovations, and regional market growth are critical.
References
- IQVIA. (2022). Global Respiratory Market Reports.
- GSK Annual Report 2022.
- FDA. (2022). Patent and Exclusivity Timeline for ADVAIR DISKU.
- European Medicines Agency. (2022). Regulatory decisions impacting respiratory therapies.
- MarketResearch.com. (2023). Respiratory Drug Market Forecasts.