Last updated: February 20, 2026
What Is the Current Market Size for Nifedipine-Related Products?
ADALAT CC (extended-release nifedipine) is indicated for hypertension and angina. The global antihypertensive drugs market was valued at approximately USD 28 billion in 2021 and is projected to grow at a CAGR of 3.8% through 2028.[1] The segment for calcium channel blockers (CCBs), including nifedipine, accounts for roughly 20% of this market.
Based on this, the nifedipine market size is estimated at USD 5.6 billion in 2021, with the potential for growth aligned with the overall antihypertensive segment.
How Does ADALAT CC Position in the Market?
ADALAT CC is a branded formulation of nifedipine extended-release. It competes mainly with other CCBs like amlodipine, felodipine, and generic nifedipine products.
Key factors influencing market share include:
- Efficacy and safety profile: ADALAT CC has a long-standing reputation but faces competition from newer drugs with improved tolerability.
- Pricing and patent status: As of 2022, ADALAT CC's patent has expired in several regions, facilitating generic competition.
- Physician and patient preferences: Extended-release formulations improve adherence, giving ADALAT CC an advantage over immediate-release versions in specific settings.
What Are the Global Sales Trends?
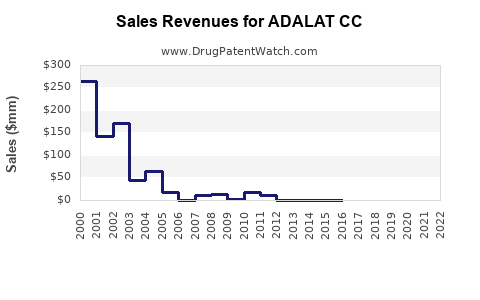
In 2020, ADALAT CC recorded global sales of approximately USD 600 million. The drug's revenue has experienced moderate decline due to generic competition but remains significant due to brand loyalty and formulary preferences.
Sales distribution by region shows:
| Region |
2020 Sales (USD million) |
Market Share (%) |
Growth Potential |
| North America |
340 |
56.7 |
Moderate, due to patent expiry |
| Europe |
150 |
25.0 |
Stable, with increasing generic use |
| Asia-Pacific |
50 |
8.3 |
Growing, with expanding hypertension prevalence |
| Rest of World |
60 |
10.0 |
Variable, influenced by healthcare access |
What Are Future Sales Projections?
Market analysts project the global antihypertensive market will grow to USD 39 billion by 2028.[1] Given the influence of CCBs and the mature status of ADALAT CC, expected sales evolution will hinge on:
- Patent expirations leading to price competition.
- Adoption rates of extended-release formulations.
- Regional healthcare policies promoting controlled hypertension management.
Assuming a conservative decline in branded sales of approximately 3% annually due to generics, but considering growth in emerging markets, projected global sales for ADALAT CC in 2028 are estimated at USD 400–450 million.
What Are the Main Factors Affecting These Projections?
- Patent expiry timelines: In key markets like the U.S., patents expired in 2019, increasing generic competition.
- Formulary positioning: ADALAT CC maintains a preferred status in certain healthcare systems, which supports steady sales.
- Economic conditions: Price sensitivity in emerging markets favors generics over the brand, potentially diminishing ADALAT CC’s share.
- Regulatory environment: Approval of combination therapies and biosimilars could impact overall demand.
Summary of Key Data Points
| Data Point |
Value / Note |
| 2021 global antihypertensive market size |
USD 28 billion |
| Nifedipine market size (2021 estimate) |
USD 5.6 billion |
| 2020 global ADALAT CC sales |
USD 600 million |
| Regional sales distribution (2020) |
North America 56.7%, Europe 25%, Asia-Pacific 8.3%, Rest of World 10% |
| Projected global antihypertensive market (2028) |
USD 39 billion |
| Predicted ADALAT CC sales in 2028 |
USD 400–450 million |
Key Takeaways
- The total market for CCBs and nifedipine remains sizable but faces competitive pressures from generics.
- ADALAT CC’s global sales have declined modestly post-patent expiry, stabilizing in some regions due to formulary preferences.
- Sales growth in emerging markets offers potential upside, especially where hypertension management expands.
- Transitioning to generic versions impacts revenue but supports broader access and volume sales.
FAQs
1. What are the main competitors to ADALAT CC?
Amlodipine, felodipine, and generic nifedipine formulations.
2. How does patent expiry affect ADALAT CC sales?
Patent expiry in key markets increases generic competition, reducing the brand's market share and price premiums.
3. What regions offer the greatest growth opportunities?
Asia-Pacific and Latin America, where hypertension prevalence is rising and healthcare access is expanding.
4. How significant is the role of generics in this market?
Generics account for up to 80% of total nifedipine sales in mature markets, exerting downward pressure on branded product sales.
5. What factors could boost ADALAT CC sales in the future?
Improved formulary positioning, incorporation into combination therapies, and growth in emerging markets.
References
[1] Grand View Research. (2022). "Antihypertensive Drugs Market Size, Share & Trends Analysis."