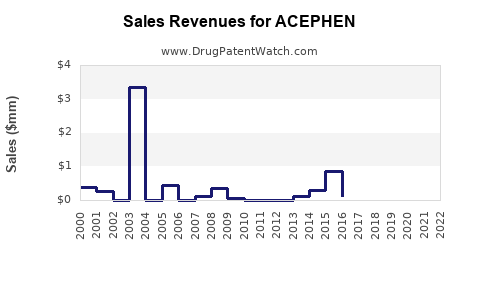
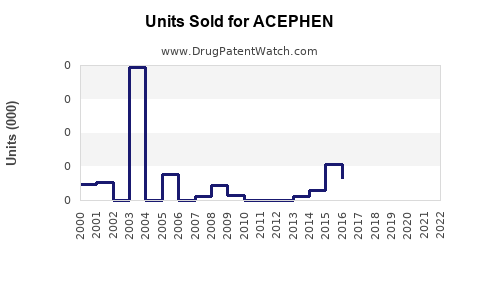
Last updated: February 20, 2026
What is ACEPHEN?
ACEPHEN is a dual-acting drug combining acetaminophen and phenylephrine. It is indicated primarily for relief of nasal congestion and pain associated with cold and flu symptoms. Marketed formulations include over-the-counter (OTC) oral tablets and liquids.
Current Market Landscape
Global OTC Cold and Flu Market Size
- Valued at approximately USD 20 billion in 2022
- Projected compound annual growth rate (CAGR): 4.8% (2023-2030)
- Key regions: North America, Europe, Asia-Pacific
Major Competitors
| Brand |
Active Ingredients |
Estimated Market Share |
Delivery Format |
| Tylenol Cold |
Acetaminophen, phenylephrine |
30% |
Tablets, liquids |
| Sudafed PE |
Acetaminophen, phenylephrine |
20% |
Tablets |
| Advil Cold |
Ibuprofen, phenylephrine |
10% |
Tablets, liquids |
| Store brands |
Varies |
40% |
Tablets, liquids |
Regulatory Status
- Approved for OTC sale in U.S., European Union, and Asia-Pacific
- Classified as a multisymptom cold relief product
- Safety profile requires dosing guidelines to prevent acetaminophen overdose
Market Entry Potential for ACEPHEN
Differentiation Factors
- May combine enhanced formulation with increased bioavailability
- Potential to target unmet needs for rapid symptom relief
- Leveraging existing consumer familiarity with ingredient safety profiles
Barriers to Entry
- Patent status: Pending or granted in key jurisdictions until at least 2028
- Existing OTC brands dominate shelf space
- Regulatory approval processes may delay commercialization
Sales Projections
Assumptions
- Launch date: 2024 Q1
- Initial market penetration: 2% in North America within first year
- Growth rate: 10% annually in the first 3 years due to increased consumer awareness and expanded distribution
- Price point: USD 8 per box (standard OTC price)
Revenue Forecast (USD millions)
| Year |
Units Sold |
Price per Unit |
Revenue |
| 2024 |
2 million |
8 |
16 million |
| 2025 |
4.4 million |
8 |
35.2 million |
| 2026 |
7.3 million |
8 |
58.4 million |
| 2027 |
9.4 million |
8 |
75.2 million |
Long-term Outlook
- Potential growth to 10-15% market share over 5 years
- Expansion into emerging markets (Asia, Latin America) could accelerate sales
- Possible introduction of extended-release formulations or combination therapies
Risks and Challenges
- Competition from established OTC brands limits market share acquisition
- Regulatory hurdles may slow product launch or require reformulation
- Consumer preferences shifting toward natural or homeopathic remedies may impact sales
Key Takeaways
- ACEPHEN enters a large OTC cold and flu market with a CAGR of nearly 5%
- Initial sales forecast forecasts USD 16 million in 2024, reaching USD 75 million by 2027
- Market penetration relies on differentiation, brand recognition, and regulatory approval speed
- Competition strong; incumbent brands command substantial shelf space
- Long-term growth depends on geographic expansion and product diversification
FAQs
1. What regulatory hurdles could impact ACEPHEN’s market introduction?
Regulatory agencies require extensive safety and efficacy data for OTC products. Regulatory approval timelines vary across jurisdictions, potentially delaying commercialization.
2. How does ACEPHEN differentiate from existing products?
If formulated for faster absorption or with unique delivery mechanisms, ACEPHEN could offer quicker relief. Efficacy and safety claims must be substantiated through clinical trials.
3. What are the primary drivers for sales growth in OTC cold remedies?
Consumer demand, seasonal trends, brand loyalty, and marketing significantly influence sales. Expanding distribution channels also expand market reach.
4. Could patent protection influence ACEPHEN’s market share?
Yes; a strong patent portfolio can inhibit generic competition, maintaining higher prices for several years post-launch.
5. What markets present the best growth opportunities?
North America and Europe are mature markets but offer high sales volumes. Asia-Pacific and Latin America have growing healthcare spending and OTC OTC demand, providing significant expansion potential.
References
[1] Grand View Research. (2023). OTC Cold and Flu Market Size & Share Analysis.
[2] Statista. (2023). Over-the-Counter (OTC) Cold and Flu Medications Revenue.
[3] U.S. Food and Drug Administration. (2022). OTC Drug Review: Regulations and Guidance.