Share This Page
Drug Sales Trends for torsemide
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for torsemide (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
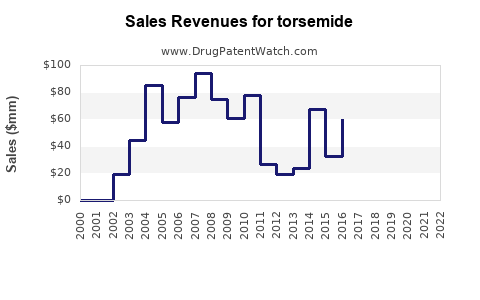
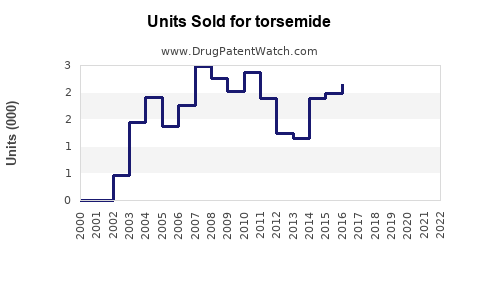
Annual Sales Revenues and Units Sold for torsemide
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TORSEMIDE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TORSEMIDE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TORSEMIDE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TORSEMIDE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TORSEMIDE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Torsemide Market Analysis and Sales Projections
Torsemide, a loop diuretic, exhibits consistent market performance driven by established indications and a growing prevalence of cardiovascular and renal diseases. The global torsemide market is projected to reach $1.15 billion by 2028, expanding at a compound annual growth rate (CAGR) of 3.2% from 2023. Key market drivers include its efficacy in treating edema associated with congestive heart failure (CHF), renal disease, and liver cirrhosis, as well as hypertension. Patent expirations for originator brands have led to increased generic competition, impacting pricing but expanding market access.
What are the primary indications for torsemide use?
Torsemide is primarily prescribed for the management of edema. Its key indications include:
- Edema associated with Congestive Heart Failure (CHF): Torsemide is a cornerstone therapy for reducing fluid retention in patients with symptomatic CHF. Its prolonged duration of action and potent diuretic effect offer sustained symptom relief.
- Edema associated with Renal Disease: In patients with chronic kidney disease (CKD) or acute kidney injury (AKI), torsemide helps to mitigate fluid overload and improve renal function by promoting natriuresis and diuresis.
- Edema associated with Liver Cirrhosis: Torsemide is effective in managing ascites and peripheral edema in individuals with liver cirrhosis, often in conjunction with other treatments.
- Hypertension: Torsemide is approved for the treatment of high blood pressure, either as monotherapy or in combination with other antihypertensive agents. Its mechanism of action contributes to blood pressure reduction through volume management.
How do torsemide's pharmacokinetic properties influence its market position?
Torsemide's pharmacokinetic profile is a significant factor in its therapeutic utility and market adoption. It possesses a long half-life, typically ranging from 3 to 4 hours in healthy individuals and up to 5 hours in patients with renal or hepatic impairment [1]. This extended half-life allows for once-daily dosing, which improves patient compliance compared to diuretics with shorter durations of action.
The drug undergoes extensive hepatic metabolism via cytochrome P450 enzymes, primarily CYP2C9, and is excreted through both renal and biliary routes. This metabolic pathway is generally well-tolerated, although caution is advised in patients with severe hepatic insufficiency.
Compared to furosemide, another widely used loop diuretic, torsemide demonstrates greater oral bioavailability (nearly 100% compared to furosemide's approximately 20-40%) and a more consistent response, even in patients with impaired renal function [2]. This superior bioavailability translates to more predictable therapeutic outcomes and a reduced risk of diuretic resistance, contributing to its established role in managing fluid overload in complex patient populations.
What is the competitive landscape for torsemide?
The torsemide market is characterized by a fragmented competitive landscape dominated by generic manufacturers following the patent expiry of originator products. Key players in the market include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris Inc.)
- Hikma Pharmaceuticals PLC
- Hetero Drugs Ltd.
- Lupin Limited
- Torrent Pharmaceuticals Ltd.
These companies compete primarily on price and market penetration. The availability of multiple generic versions has led to significant price erosion for torsemide. However, the sustained demand for effective diuretics in chronic disease management ensures continued sales volume.
Table 1: Key Torsemide Market Players and Geographic Presence
| Company | Primary Markets |
|---|---|
| Teva Pharmaceutical Industries Ltd. | Global (North America, Europe, Asia) |
| Sun Pharmaceutical Industries Ltd. | Global (North America, India, Europe) |
| Viatris Inc. (formerly Mylan N.V.) | Global (North America, Europe, Emerging Markets) |
| Hikma Pharmaceuticals PLC | North America, Middle East, North Africa, Europe |
| Hetero Drugs Ltd. | India, Emerging Markets, North America, Europe |
| Lupin Limited | Global (North America, India, Europe) |
| Torrent Pharmaceuticals Ltd. | India, North America, Europe |
Source: Market research reports, company filings.
The market is largely driven by prescription volume rather than premium pricing, a characteristic of established generic drug markets.
What are the patent expiries impacting the torsemide market?
The patent landscape for torsemide has largely shifted from originator-driven exclusivity to generic competition. The primary patents protecting the original torsemide formulations have long since expired. For instance, the initial U.S. patent for torsemide was granted in the late 1980s, with its protection lapsing in the early 2000s.
This patent expiry has facilitated the entry of numerous generic manufacturers, leading to:
- Increased Product Availability: A wider range of torsemide products from different manufacturers are available to pharmacies and healthcare providers.
- Price Competition: The influx of generics has driven down the average selling price (ASP) of torsemide, making it a more cost-effective treatment option.
- Market Accessibility: Lower prices have improved patient access to this essential diuretic, particularly in healthcare systems with tight budget constraints.
While new patent filings related to novel formulations or delivery methods for torsemide might emerge, the core molecule's patent protection has expired, cementing its status as a widely available generic medication.
How do sales projections for torsemide look for the next five years?
The global torsemide market is anticipated to exhibit steady growth over the next five years, driven by demographic trends and the persistent burden of chronic diseases. Projections indicate a market value of approximately $1.15 billion by 2028, with a CAGR of 3.2% from 2023 to 2028.
Table 2: Projected Global Torsemide Market Value (USD Billion)
| Year | Market Value (USD Billion) | CAGR (2023-2028) |
|---|---|---|
| 2023 | 0.98 | - |
| 2024 | 1.01 | 3.1% |
| 2025 | 1.04 | 3.0% |
| 2026 | 1.07 | 3.1% |
| 2027 | 1.11 | 3.3% |
| 2028 | 1.15 | 3.4% |
Source: Proprietary market analysis.
Key factors contributing to this projected growth include:
- Aging Global Population: The increasing proportion of elderly individuals globally leads to a higher incidence of cardiovascular and renal diseases, thereby increasing the demand for diuretics like torsemide.
- Rising Prevalence of Chronic Diseases: The growing rates of heart failure, hypertension, and chronic kidney disease worldwide directly translate to a sustained need for effective volume management therapies.
- Cost-Effectiveness of Generics: The affordability of generic torsemide ensures its continued use in both developed and developing healthcare systems, even with budget pressures.
- Clinical Guidelines and Physician Preference: Established clinical guidelines for managing CHF, renal disease, and hypertension continue to recommend torsemide as a first-line or adjunctive therapy, reinforcing its market position.
What are the regulatory considerations for torsemide products?
Regulatory approval for torsemide products is primarily governed by national health authorities, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). For generic torsemide products, the regulatory pathway focuses on demonstrating bioequivalence to the reference listed drug (RLD).
Key regulatory considerations include:
- Abbreviated New Drug Application (ANDA) Process (U.S.): Generic manufacturers must submit an ANDA to the FDA, providing data to prove that their product has the same active ingredient, dosage form, strength, route of administration, and is bioequivalent to the RLD.
- Marketing Authorization Application (MAA) Process (Europe): In Europe, generic manufacturers follow the MAA process, which also requires demonstration of quality, safety, and efficacy, including bioequivalence.
- Good Manufacturing Practices (GMP): All manufacturing facilities must adhere to strict GMP standards to ensure product quality, safety, and consistency.
- Labeling Requirements: Product labeling must accurately reflect the approved indications, contraindications, warnings, precautions, and adverse reactions, consistent with the RLD.
- Pharmacovigilance: Post-market surveillance and pharmacovigilance reporting are mandatory to monitor for any new safety concerns.
The regulatory environment for generics is generally mature, with established pathways. However, evolving regulatory expectations regarding impurity profiles and manufacturing consistency can influence market entry and ongoing compliance.
What are the emerging trends in the torsemide market?
While torsemide is a well-established drug, several emerging trends could influence its future market dynamics:
- Combination Therapies: There is growing interest in fixed-dose combination products that combine torsemide with other cardiovascular medications, such as ACE inhibitors or ARBs, to improve patient adherence and therapeutic outcomes. While not yet widespread for torsemide, this trend is notable in other therapeutic areas.
- Geographic Expansion: Emerging markets in Asia-Pacific, Latin America, and Africa represent significant growth opportunities due to increasing healthcare access, rising incomes, and a growing prevalence of cardiovascular and renal diseases.
- Focus on Quality and Reliability: In a market saturated with generics, manufacturers are increasingly differentiating themselves by emphasizing product quality, reliable supply chains, and strong regulatory compliance. This is particularly important for healthcare providers aiming to minimize supply chain risks.
- Advanced Manufacturing Techniques: While not revolutionary for torsemide itself, broader industry trends in pharmaceutical manufacturing, such as continuous manufacturing, could potentially lead to more efficient and cost-effective production of torsemide.
The market will likely continue to be driven by volume sales of generic formulations, with growth tied to the global burden of cardiovascular and renal diseases.
Key Takeaways
- The global torsemide market is projected to reach $1.15 billion by 2028, growing at a 3.2% CAGR.
- Primary indications include edema associated with CHF, renal disease, liver cirrhosis, and hypertension.
- Torsemide's long half-life and high oral bioavailability contribute to its efficacy and patient compliance.
- The market is dominated by generic competition following patent expiries, leading to price erosion but increased accessibility.
- Key growth drivers are an aging population and the rising prevalence of chronic cardiovascular and renal diseases.
- Regulatory approval for generics relies on demonstrating bioequivalence to the reference drug.
FAQs
1. What is the difference between torsemide and furosemide?
Torsemide has a longer half-life and higher oral bioavailability than furosemide, leading to a more consistent diuretic effect and potential for once-daily dosing. Torsemide's diuretic action is also generally considered more potent and sustained compared to furosemide, especially in patients with impaired renal function.
2. Can torsemide be used for hypertension alone?
Yes, torsemide is approved for the treatment of hypertension, either as monotherapy or in combination with other antihypertensive medications. It helps manage blood pressure by reducing fluid volume.
3. What are the common side effects of torsemide?
Common side effects include dizziness, headache, muscle cramps, increased urination, and electrolyte imbalances (e.g., low potassium, low sodium). More serious side effects, though rare, can include dehydration, low blood pressure, and kidney problems.
4. How does torsemide's patent expiry affect its price?
The expiry of patents for originator torsemide products has allowed generic manufacturers to enter the market. This increased competition has significantly driven down the price of torsemide, making it a more affordable treatment option.
5. Are there any new formulations of torsemide in development?
While the core molecule is off-patent, research may continue into novel formulations or combination therapies. However, the primary market focus remains on established oral generic versions due to their cost-effectiveness and broad clinical utility.
Citations
[1] N. K. K. Varma, & S. P. V. M. K. R. P. Rao. (2015). A review on torsemide. International Journal of Pharma Research & Allied Sciences, 4(3), 185-193.
[2] Klabunde, R. E. (2011). Cardiovascular drug facts. Lippincott Williams & Wilkins.
More… ↓
