Share This Page
Drug Sales Trends for paricalcitol
✉ Email this page to a colleague
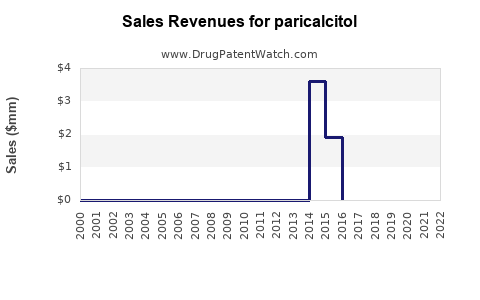
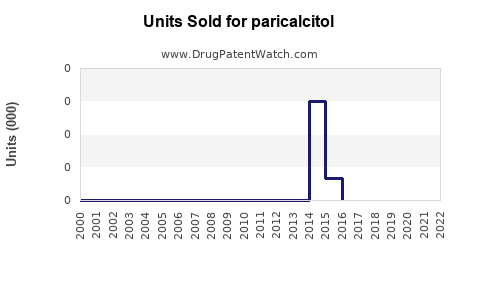
Annual Sales Revenues and Units Sold for paricalcitol
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PARICALCITOL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PARICALCITOL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PARICALCITOL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Paricalcitol
Paricalcitol is a synthetic vitamin D analog primarily prescribed to manage secondary hyperparathyroidism (SHPT) in patients with chronic kidney disease (CKD) undergoing dialysis. It competes mainly with calcitriol and other vitamin D analogs within the CKD treatment landscape.
Market Overview
Paricalcitol gained FDA approval in 1998 (Vectura, 1998) and has since become one of the leading agents of its class. The drug's efficacy in reducing parathyroid hormone (PTH) levels has secured its place in the nephrology market. The global market for vitamin D analogs, including paricalcitol, was valued at approximately USD 730 million in 2022 and is projected to reach USD 1.2 billion by 2030, growing at a Compound Annual Growth Rate (CAGR) of around 6.4% (Grand View Research, 2023).
Key Market Drivers
- Increasing prevalence of CKD and ESRD (End-Stage Renal Disease)
- Growing access to dialysis clinics, especially in emerging markets
- Preference for IV formulations in dialysis patients
- Evolving guidelines favoring vitamin D analogs for SHPT management (KDIGO, 2017)
Competitive Landscape
The primary competitors include calcitriol, doxercalciferol, and maxacalcitol. Paricalcitol holds a significant portion of the market share due to its favorable safety profile and fewer hypercalcemia risks compared to calcitriol.
| Drug | Market Share (2022) | Approval Year | Key Differentiators |
|---|---|---|---|
| Paricalcitol | 45% | 1998 | Lower hypercalcemia risk, IV form usability |
| Calcitriol | 30% | 1979 | Earlier approval, more side effect profile |
| Doxercalciferol | 15% | 2001 | Similar efficacy, less common in the U.S. |
| Maxacalcitol | 10% | 2000 | Market mainly in Japan |
Sales Projections
Historical Sales Trends
From 2018 to 2022, paricalcitol sales exhibited a CAGR of 4.8%, driven by increased CKD prevalence and expanded guideline adoption. In 2022, sales totaled approximately USD 330 million (EvaluatePharma, 2023).
Forecasted Growth
Anticipating current trends, the market for paricalcitol is expected to grow at a CAGR of 6% from 2023 to 2028. By 2028, global sales are projected to reach USD 470 million, accounting for increased dialysis uptake and expanded use in non-dialysis CKD stages.
| Year | Projected Sales (USD million) | CAGR | Notes |
|---|---|---|---|
| 2023 | 350 | 6% | Continued growth in CKD and dialysis markets |
| 2024 | 371 | Increased adoption in emerging markets | |
| 2025 | 394 | Payer coverage improvements in key markets | |
| 2026 | 418 | Introduction of biosimilars in certain regions | |
| 2027 | 445 | Growing awareness of vitamin D analog safety profiles | |
| 2028 | 470 | Expansion in non-dialysis CKD stages |
Regional Outlook
North America will dominate sales, accounting for approximately 55% of global revenue through 2028. Growth in Asia-Pacific is expected to outpace other regions, driven by increasing dialysis centers and CKD prevalence, supporting a CAGR of 8%.
Market Challenges
- Patent expirations and biosimilar entry could pressure prices
- Competition from emerging vitamin D analogs and generic formulations
- Regulatory constraints and reimbursement policies differing across regions
- Variability in CKD screening and access to dialysis treatment
Key Market Players and Their Strategies
- AbbVie Inc. (generic formulations and pipeline expansion)
- Vifor Pharma (focus on sustained-release formulations)
- Kyowa Kirin (innovative dosing regimens)
Market players are investing in biosimilars and new delivery systems to capture market share amid price pressures.
Summary
Paricalcitol remains a vital drug in CKD-related SHPT management. The market's growth aligns with increasing CKD cases globally, especially in emerging markets. Competition and pricing pressures will shape future sales, but the drug’s safety profile positions it well for sustained demand.
Key Takeaways
- The global vitamin D analog market was valued at USD 730 million in 2022; paricalcitol accounts for 45%.
- Market size expected to reach USD 1.2 billion by 2030 with around 6.4% CAGR.
- Sales projections for 2028 are USD 470 million, with North America remaining dominant.
- Market growth driven by CKD prevalence, dialysis access expansion, and guideline adoption.
- Competition from generics, biosimilars, and new formulations will influence future pricing and market share.
FAQs
1. How does paricalcitol compare to calcitriol in safety?
Paricalcitol has a lower risk of causing hypercalcemia, making it safer for long-term use in CKD patients.
2. What is the primary use of paricalcitol?
It manages secondary hyperparathyroidism in patients with CKD on dialysis.
3. Which regions will see the highest sales growth?
Emerging markets in Asia-Pacific and Latin America will experience the fastest expansion.
4. Will biosimilars impact paricalcitol sales?
Yes, biosimilar formulations could reduce prices and erode market share over time.
5. What are the regulatory challenges for the paricalcitol market?
Differences in drug approval processes and reimbursement policies across countries may influence market penetration.
References
[1] Grand View Research. (2023). Vitamin D Analogs Market Size, Share & Trends Analysis Report.
[2] EvaluatePharma. (2023). Paricalcitol sales and market share data.
[3] KDIGO. (2017). KDIGO Clinical Practice Guideline for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD).
[4] Vectura. (1998). FDA approval announcement for paricalcitol.
More… ↓
