Last updated: February 14, 2026
Nystatin is an antifungal medication primarily used to treat fungal infections of the skin, mouth, esophagus, and intestines. It has been available since the 1950s and is marketed in various formulations, including topical creams, powders, ointments, and oral suspensions.
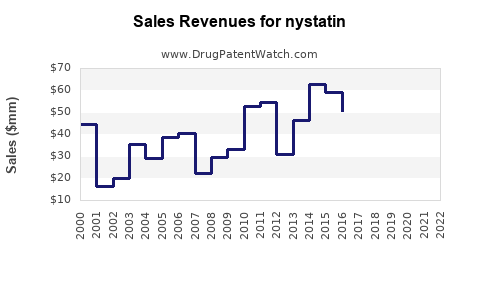
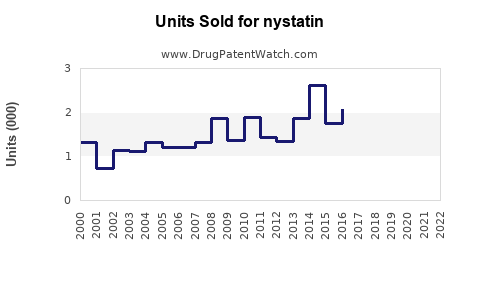
Global Market Size (2022)
Estimated at $150 million, driven predominantly by North America, Europe, and parts of Asia-Pacific. The antifungal drugs market overall was valued at approximately $13 billion in 2022, with Nystatin constituting about 1%-2% of this figure.
Market Dynamics
The market remains relatively stable, supported by the following factors:
- Rising fungal infection cases: Increased incidence of immunocompromised conditions such as HIV/AIDS, cancer, and organ transplantation boosts demand.
- Shift towards topical administration: Fewer side effects compared to systemic antifungals increase preference.
- Off-label use and pediatric applications: Its safety profile in children sustains demand in pediatric populations.
Competitive Landscape
Main competitors include:
- Clotrimazole
- Miconazole
- Amphotericin B (systemic)
- Fluconazole (systemic)
Nystatin's niche in topical and oral mucosal applications faces competition but maintains a foothold due to its minimal systemic absorption and toxicity profile.
Regulatory Status
Approved by the U.S. FDA in the 1950s, classified as over-the-counter (OTC) or prescription depending on formulation. Regulatory pathways for various formulations are straightforward, aiding market access.
Sales Projections and Future Trends (2023–2028)
Assumptions
- CAGR (Compound Annual Growth Rate): 3-4%
- Market growth driven by increasing infection rates and pediatric use
- No major patent expirations or generic disruptions anticipated soon
Projected Market Size (2028)
Estimated to reach approximately $180 million to $210 million globally if CAGR aligns with 3-4%. Growth remains moderate due to its established status and competition from newer antifungals.
| Year |
Market Size (USD millions) |
CAGR |
Notes |
| 2022 |
150 |
— |
Baseline |
| 2023 |
155–156 |
3.3% |
Slight increase due to rising infections |
| 2024 |
160–163 |
3–4% |
Expansion in emerging markets |
| 2025 |
165–170 |
3–4% |
Dental and oral candidiasis growth |
| 2026 |
170–176 |
3–4% |
Pediatric use stabilizes |
| 2027 |
175–180 |
3–4% |
Healthcare infrastructure improvements |
| 2028 |
180–210 |
3–4% |
Potential impact from generics or formulary shifts |
Market Drivers and Barriers
Drivers:
- Increase in HIV/AIDS and cancer patients
- Growing preference for topical antifungals due to safety
- Expansion into developing markets with rising healthcare investments
Barriers:
- Competition from systemic antifungals with broader spectrum
- OTC formulation limitations in some regions
- Emergence of antifungal resistance, though less relevant for Nystatin due to topical use and minimal systemic absorption
Regulatory and Commercial Considerations
Nystatin remains largely an off-patent drug with many generic versions, limiting pricing power but ensuring widespread availability. The OTC availability in many countries supports high accessibility, although prescription-only status persists for certain formulations.
Market entry and expansion require adherence to regional regulatory pathways for new formulations or indications. The product's safety profile helps with market acceptance, especially in pediatric and superficial infections.
Conclusion
Nystatin is a mature antifungal with a stable global market. Growth is expected to be moderate, supported by demographic trends and increasing fungal infections. Competition from systemic antifungals and generics remains a key factor influencing sales.
Key Takeaways
- The global Nystatin market was valued at approximately $150 million in 2022.
- Growth projections from 2023 to 2028 indicate a CAGR of 3-4%, reaching up to $210 million.
- Market drivers include rising fungal infections and preference for topical formulations, with barriers posed by competition and antifungal resistance.
- The drug’s established safety profile and broad availability ensure continued relevance, especially in pediatric and superficial infections.
FAQs
-
What formulations of Nystatin are most commonly used?
Topical creams, ointments, powders, and oral suspensions are most common. Oral formulations are frequently used for mucosal and esophageal candidiasis.
-
Are there new formulations or indications for Nystatin in development?
While limited, research explores new delivery systems (e.g., lozenges, patches) and potential off-label uses, but none have reached widespread commercial availability as of 2023.
-
How does Nystatin compare to newer antifungal agents?
It has a more favorable safety profile for superficial and mucosal infections but lacks systemic activity against invasive fungi, where azoles or amphotericin B are preferred.
-
What regulatory challenges exist for expanding Nystatin's use?
Approval for new indications requires clinical trials demonstrating safety and efficacy. Patent protections are minimal or expired, limiting innovation incentive.
-
What is the outlook for generic competition impacting Nystatin sales?
Generic versions dominate the market, exerting downward pressure on prices but ensuring access. Brand-specific differentiation is limited.
References
- MarketsandMarkets. "Antifungal Drugs Market." 2022.
- IBISWorld. "Antifungal Drug Industry in the US." 2022.
- U.S. Food and Drug Administration. Nystatin Monograph. 2023.