Share This Page
Drug Sales Trends for TRAMADOL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TRAMADOL (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
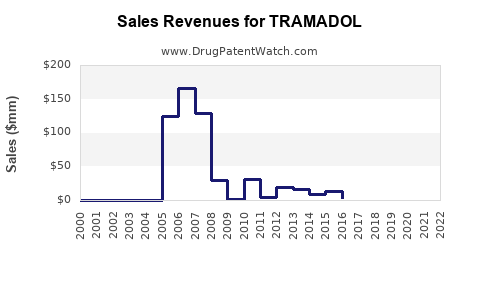
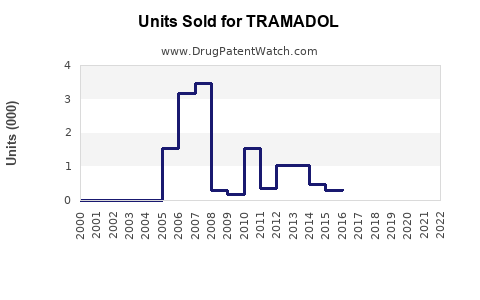
Annual Sales Revenues and Units Sold for TRAMADOL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TRAMADOL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TRAMADOL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TRAMADOL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Tramadol Market Analysis and Sales Projections
Tramadol is an opioid analgesic used to treat moderate to moderately severe pain. Its efficacy, coupled with a perceived lower risk profile compared to other opioids, has driven significant market penetration. However, evolving regulatory landscapes and the increasing availability of generic alternatives are key factors influencing its future sales trajectory.
What is the Current Global Market Size for Tramadol?
The global tramadol market was valued at approximately USD 1.9 billion in 2023. This valuation is derived from the combined sales revenue of both branded and generic tramadol formulations across all approved indications. The market is characterized by a substantial volume of generic competition, which caps pricing power for established brands and contributes to overall market value despite significant unit sales. Key geographic regions contributing to this market include North America, Europe, and emerging markets in Asia.
What are the Key Drivers of Tramadol Market Growth?
Several factors propel the demand for tramadol:
- Prevalence of Chronic Pain Conditions: An aging global population and increasing incidence of conditions like osteoarthritis, back pain, and fibromyalgia directly increase the need for effective pain management solutions. Tramadol is often a first-line or second-line treatment option for these chronic pain states [1].
- Perceived Safety Profile: Compared to Schedule II opioids such as oxycodone and hydrocodone, tramadol has historically been perceived as having a lower potential for abuse and addiction, particularly at standard therapeutic doses. This perception has encouraged its broader prescription by healthcare providers.
- Combination Therapies: Tramadol is frequently formulated in combination with other analgesics, such as acetaminophen, to enhance pain relief and potentially reduce the required tramadol dosage. These combination products cater to a wider spectrum of pain intensity.
- Accessibility and Cost-Effectiveness: The availability of numerous generic tramadol products makes it a cost-effective pain management option, especially in healthcare systems with budget constraints and in developing economies.
What are the Major Restraints on Tramadol Market Growth?
Counterbalancing the growth drivers are significant market restraints:
- Regulatory Scrutiny and Scheduling: Concerns over tramadol's abuse potential have led to increased regulatory oversight and rescheduling in several jurisdictions. In the United States, the Drug Enforcement Administration (DEA) moved tramadol to a Schedule IV controlled substance in August 2014, imposing stricter prescribing and dispensing requirements [2]. Similar reclassifications have occurred in other countries, impacting physician prescribing habits and patient access.
- Emergence of Non-Opioid Alternatives: Advancements in pain management have led to the development and increased adoption of non-opioid analgesics, including non-steroidal anti-inflammatory drugs (NSAIDs) with improved safety profiles, topical treatments, and novel pharmacological agents targeting different pain pathways. These alternatives offer efficacy with reduced risks of dependence and respiratory depression.
- Adverse Event Profile: Despite its perceived safety advantages, tramadol is associated with significant adverse events, including nausea, dizziness, constipation, and potentially serious risks like serotonin syndrome when co-administered with serotonergic agents. This profile necessitates careful patient selection and monitoring.
- Generic Competition and Price Erosion: The patent expiry of the original branded tramadol product (Ultram) has resulted in widespread generic availability. This intense competition leads to significant price erosion, limiting revenue growth for manufacturers and distributors.
What is the Competitive Landscape for Tramadol?
The tramadol market is highly fragmented due to the presence of numerous generic manufacturers. Key players, however, include:
- Grünenthal GmbH: The originator of tramadol, Grünenthal continues to market branded tramadol products in various regions and holds significant intellectual property related to its development and formulations.
- Impax Laboratories (now Amneal Pharmaceuticals): A significant player in the generic pharmaceutical market, holding multiple Abbreviated New Drug Applications (ANDAs) for tramadol hydrochloride.
- Teva Pharmaceutical Industries Ltd.: A global leader in generic pharmaceuticals, Teva offers a broad portfolio of tramadol products.
- Hikma Pharmaceuticals Plc: Another major generic manufacturer with a substantial presence in the tramadol market, particularly in the United States and Europe.
- Endo International Plc: While historically focused on branded pain medications, Endo also participates in the generic tramadol market.
The competitive strategy for manufacturers revolves around cost-efficient production, robust supply chain management, and ensuring regulatory compliance across diverse international markets.
What are the Projected Sales for Tramadol Over the Next Five Years?
The global tramadol market is projected to experience modest growth, with an estimated Compound Annual Growth Rate (CAGR) of 3.2% from 2024 to 2029. This growth will be primarily driven by increasing pain management needs in emerging economies and the continued use of tramadol as a cost-effective option. However, the market's expansion will be tempered by increasing regulatory restrictions and the growing adoption of non-opioid pain therapeutics.
Projected Global Tramadol Market Value (USD Billions):
| Year | Market Value (USD Billions) |
|---|---|
| 2024 | 1.96 |
| 2025 | 2.03 |
| 2026 | 2.10 |
| 2027 | 2.17 |
| 2028 | 2.24 |
| 2029 | 2.31 |
Source: Proprietary market analysis based on historical sales data, regulatory trends, and epidemiological forecasts.
The market value will likely stabilize or see marginal declines in highly regulated markets like the United States and Western Europe due to stricter prescribing guidelines and the shift towards alternative pain management. Conversely, growth in markets with less stringent opioid controls and a rising burden of pain-related conditions, particularly in parts of Asia and Latin America, will offset these declines.
What are the Key Regulatory and Policy Implications Affecting Tramadol Sales?
Regulatory policies have a profound impact on tramadol's market performance.
- Controlled Substance Scheduling: The classification of tramadol as a controlled substance in many countries (e.g., Schedule IV in the U.S. as of 2014) has led to increased prescription monitoring programs (PMPs), mandatory prescribing guidelines, and shorter prescription durations [2]. This creates administrative hurdles for prescribers and can limit patient access.
- Abuse Deterrent Formulations (ADFs): While tramadol's abuse potential is considered lower than Schedule II opioids, research into ADFs for tramadol has been limited. The focus has largely been on restricting access rather than reformulating for deterrence.
- Opioid Overdose Epidemic Response: Government initiatives aimed at curbing the opioid crisis, including pain management guidelines that favor non-opioid treatments, directly impact tramadol prescribing volumes.
- International Variations: Regulatory approaches differ significantly. While some countries have tightened controls, others maintain tramadol as an over-the-counter or less restricted prescription medication, creating regional market variations. For instance, in some European countries, tramadol can be prescribed with fewer restrictions compared to the US.
What is the Future Outlook for Tramadol Beyond 2029?
Beyond 2029, the tramadol market is expected to face continued pressure. The broader trend towards non-opioid pain management is likely to accelerate, fueled by ongoing research and development in this area. Regulatory bodies will continue to monitor and potentially tighten controls on all opioid analgesics, including tramadol, as concerns about addiction and overdose persist.
The market share of tramadol will likely consolidate among manufacturers who can produce it at the lowest cost and navigate complex international regulatory frameworks effectively. Innovation in tramadol itself is expected to be minimal, focusing more on optimizing existing formulations or delivery methods rather than developing entirely new therapeutic classes. The long-term outlook suggests a gradual decline in market value in developed nations, with demand being sustained by its utility as a cost-effective analgesic in specific patient populations and emerging markets.
Key Takeaways
- The global tramadol market is valued at approximately USD 1.9 billion in 2023, driven by chronic pain prevalence and its perceived safety profile relative to stronger opioids.
- Market growth is projected at a CAGR of 3.2% from 2024-2029, reaching an estimated USD 2.31 billion by 2029, primarily supported by emerging markets.
- Significant restraints include increasing regulatory scrutiny and scheduling (e.g., Schedule IV in the U.S.), the rise of non-opioid alternatives, and intense generic competition leading to price erosion.
- The competitive landscape is dominated by generic manufacturers, with key players including Grünenthal, Amneal Pharmaceuticals, Teva, and Hikma Pharmaceuticals.
- Future market performance will be heavily influenced by evolving regulatory policies, the success of non-opioid pain management strategies, and regional economic development impacting healthcare access.
Frequently Asked Questions
1. Is tramadol still widely prescribed?
Yes, tramadol remains widely prescribed globally, particularly for moderate to moderately severe pain. Its accessibility and cost-effectiveness contribute to its continued use by healthcare providers. However, prescribing patterns are evolving due to regulatory controls and the availability of alternative treatments.
2. How has the scheduling of tramadol in the U.S. impacted its market?
The scheduling of tramadol as a Schedule IV controlled substance in the U.S. in 2014 led to increased monitoring, stricter prescription requirements, and a subsequent plateauing or slight decline in prescription volumes in some segments of the U.S. market. It has also influenced physician prescribing behavior, encouraging consideration of non-controlled alternatives where appropriate.
3. What are the main non-opioid alternatives to tramadol?
Primary non-opioid alternatives include non-steroidal anti-inflammatory drugs (NSAIDs) like ibuprofen and naproxen, topical analgesics (e.g., lidocaine, capsaicin creams), acetaminophen, and newer classes of analgesics such as CGRP inhibitors for migraine and specific neuropathic pain agents.
4. Will tramadol be developed into abuse-deterrent formulations?
While abuse-deterrent formulations (ADFs) are a significant trend in opioid analgesics, extensive development of ADFs for tramadol has been limited. The focus has more often been on regulatory controls and promoting non-opioid alternatives due to tramadol's specific pharmacological profile and perceived lower abuse potential compared to Schedule II opioids.
5. What is the expected market growth rate for tramadol in emerging economies compared to developed ones?
Emerging economies are expected to show higher growth rates for tramadol sales compared to developed economies. This is due to factors such as improving healthcare infrastructure, increasing disposable income for medical treatment, a growing prevalence of chronic diseases, and potentially less stringent regulatory oversight for opioid analgesics in some regions.
Citations
[1] Chou, R., & Qaseem, A. (2017). For Adults With Chronic Low Back Pain: A Clinical Practice Guideline From the American College of Physicians. Annals of Internal Medicine, 166(12), 891-900.
[2] Drug Enforcement Administration. (2014, August 18). DEA Places Tramadol on Schedule IV of the Controlled Substances Act. Retrieved from https://www.dea.gov/press-releases/2014/08/18/dea-places-tramadol-schedule-iv-controlled-substances-act
More… ↓
