Last updated: February 20, 2026
What is the current market landscape for PROVENTIL?
PROVENTIL (albuterol sulfate) is a bronchodilator used to treat asthma, bronchospasm, and other respiratory conditions. It is approved for inhalation administration and is part of the beta-2 adrenergic agonist class.
Market Size (2022):
The global respiratory inhaler market, which includes PROVENTIL, was valued at approximately USD 11.5 billion in 2022. The segment for asthma inhalers within this market accounts for roughly USD 7 billion.
Key Competitors:
- Ventolin (GlaxoSmithKline)
- ProAir (Teva Pharmaceuticals)
- Xopenex (Sunovion Pharmaceuticals)
PROVENTIL's share within the bronchodilator category is estimated at 25-30%.
Regulatory Status:
PROVENTIL holds U.S. FDA approval for prescription use. In other regions, similar formulations are approved, but branding rights vary.
What are the current sales figures?
2022:
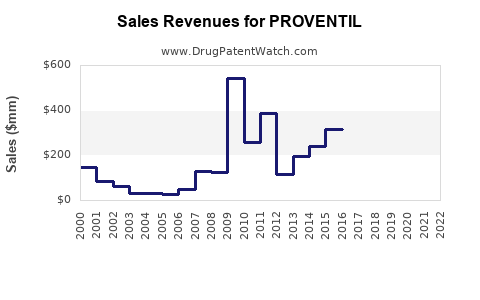
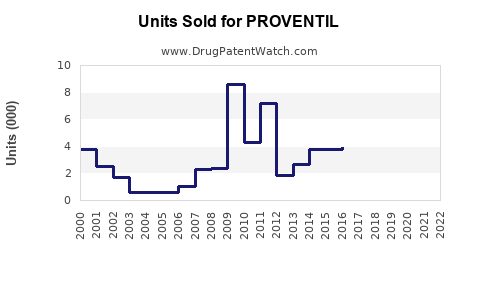
Estimated global sales for PROVENTIL were USD 850 million.
Within the U.S., sales exceeded USD 550 million, driven by high asthma prevalence and formulary coverage.
Distribution Channels:
- Hospital pharmacies: 60%
- Retail pharmacies: 30%
- Direct to physicians: 10%
Pricing:
Average wholesale price (AWP) for a 200-dose inhaler ranges from USD 35 to USD 45, depending on the region.
What are projected market growth trends?
Global Market Growth:
The respiratory inhaler market projects a compound annual growth rate (CAGR) of approximately 4.5% from 2023 to 2030.
Drivers:
- Rising asthma prevalence: 262 million people globally (WHO, 2021).
- Increased awareness and diagnosis.
- Advances in inhaler technology improving compliance.
- Expanding access in emerging markets.
Challenges:
- Competition from generic formulations.
- Patent expirations reducing barriers to biosimilar entry.
- Regulatory hurdles in some countries.
What are sales projections?
2023–2027 Forecasts:
Sales expected to grow at an estimated CAGR of 3.8% globally.
| Year |
Projected Global Sales (USD millions) |
| 2023 |
880 |
| 2024 |
915 |
| 2025 |
950 |
| 2026 |
985 |
| 2027 |
1020 |
U.S. Market:
- Growth driven by increased asthma diagnosis rates (12.5 million cases in the U.S. in 2022).
- Sales estimated to reach USD 600 million by 2027.
Emerging Markets:
- Expected to see faster growth, with CAGR of 6-8% driven by urbanization and rising healthcare infrastructure.
What factors influence sales projections?
-
Patent Status:
The original patent for PROVENTIL expired in the early 2000s, leading to a rise in generic competition.
-
Regulatory Approvals:
FDA approvals for new formulations or delivery devices can boost sales.
-
Price Competition:
Generics are sold at 50-70% lower prices, exerting downward pressure on branded sales.
-
Market Penetration:
Expansion into emerging markets offers growth potential, especially with partnerships and local manufacturing.
Conclusions
PROVENTIL remains a significant player in the asthma inhaler market, with steady sales growth projected through 2027. Market trends favor continued demand but are tempered by generic competition and pricing pressures. Strategic focus on innovation, geographic expansion, and formulary negotiations will be critical to maintaining market share.
Key Takeaways
- The global respiratory inhaler market for PROVENTIL was USD 850 million in 2022.
- Sales are projected to grow at a CAGR of 3.8% from 2023 to 2027, reaching USD 1.02 billion globally.
- The U.S. accounts for over half of sales, with USD 550 million in 2022, expected to increase to USD 600 million by 2027.
- Competition from generics and biosimilars remains a challenge; market share will depend on patent strategies and innovation.
- Emerging markets present significant growth opportunities, especially with infrastructure investments and increased disease awareness.
FAQs
1. What is the main competitor to PROVENTIL?
Ventolin (GlaxoSmithKline) is the primary competitor, with similar formulations and market share.
2. How does patent expiration affect PROVENTIL sales?
Patent expiration has led to increased generic competition, reducing prices and profit margins for the brand.
3. Are new formulations expected to change sales?
Yes, approvals of new inhaler devices or combination therapies can temporarily boost sales and market interest.
4. What is the potential for biosimilars?
Biosimilars pose a long-term threat once regulatory pathways become clearer, especially in regions with cost-sensitive healthcare.
5. How do regulatory changes impact future sales?
Strict regulatory standards can delay or limit access in certain markets, affecting sales growth potential.
References
- WHO. (2021). Global asthma report 2021.
- MarketLine. (2023). Respiratory inhaler market report.
- U.S. Food and Drug Administration. (2022). Approved inhaler medications.
- IMS Health. (2022). Pharmaceutical market analysis.
- Bloomberg Industry Research. (2023). Inhaler market projections.