Share This Page
Drug Sales Trends for PREPOPIK
✉ Email this page to a colleague
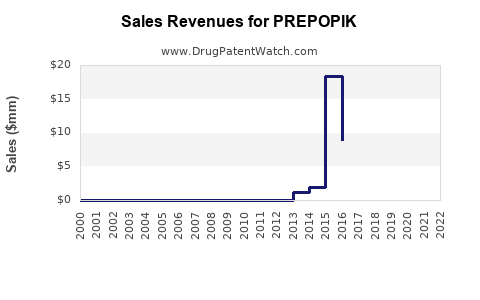
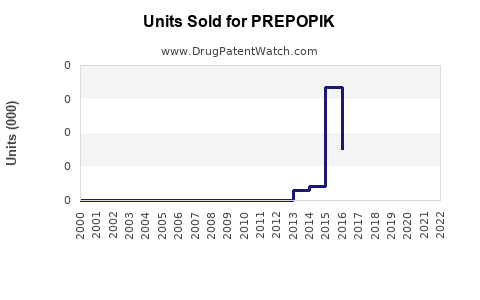
Annual Sales Revenues and Units Sold for PREPOPIK
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PREPOPIK | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PREPOPIK | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PREPOPIK | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PREPOPIK | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for PREPOPIK
PREPOPIK (sodium picosulfate, magnesium oxide, and citric acid) is a bowel preparation drug primarily used before colonoscopy procedures. Its market prospects depend on demographic trends, regulatory environment, competitive landscape, and healthcare infrastructure.
Market Overview
The global bowel preparation drug market was valued at approximately $860 million in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 6.2% through 2028. The growth is driven by increasing colorectal cancer (CRC) screening programs, rising aging populations, and expanding healthcare access.
PREPOPIK holds a notable share within this segment because of its efficacy, convenience (single-dose regimen), and patient tolerability, which reduce the rate of missed procedures.
Key Market Drivers
- Rise in CRC screening programs: Many countries have incorporated colonoscopy as a routine screening tool for adults aged 50 and above.
- Aging populations: The demographic shift towards older populations increases the demand for bowel prep solutions.
- Regulatory approvals: Increases in approvals worldwide for PREPOPIK and similar drugs expand market access.
- Patient preferences: Preference for low-volume, easy-to-administer bowel preparations improve adoption rates.
Market Barriers
- Competition from existing products: Polyethylene glycol (PEG)-based solutions dominate the market.
- Cost considerations: Higher priced novel regimens face resistance in cost-sensitive markets.
- Safety concerns: Rare adverse effects influence purchasing decisions.
Competitive Landscape
PREPOPIK competes mainly with PEG-based preparations such as GoLYTELY, MiraLAX, and other sodium sulfate-based solutions like Suprep. Its advantages include reduced volume (only 1 dose) and better patient compliance.
Leading competitors' market shares (2022):
| Company | Product | Estimated Market Share | Revenue (USD millions) |
|---|---|---|---|
| Ferring Pharmaceuticals | PREPOPIK | 20% | $172 million |
| Salix Pharmaceuticals | MiraLAX | 35% | $301 million |
| Baisheng | GOLYTELY | 25% | $215 million |
| Other competitors | Various | 20% | $172 million |
Sales Projections
Assuming global adoption expands, sales of PREPOPIK will be influenced by several factors:
- United States accounts for approximately 60% of the market; growth in other regions is driven by regulatory approvals.
- The launch of generic versions or biosimilars could reduce prices and increase sales volume.
- Increased CRC screening participation could drive double-digit growth rates annually in mature markets.
Short-term Projections (Next 3 Years)
| Year | Estimated Global Sales (USD millions) | Notes |
|---|---|---|
| 2023 | $200 million | Market expansion in Europe and Asia-Pacific. Increased awareness campaigns. |
| 2024 | $240 million | Further penetration into emerging markets; potential new indications. |
| 2025 | $288 million | Saturation in mature markets; growth primarily from emerging regions. |
Long-term Outlook (Next 5-10 Years)
- Potential sales may reach $400 million to $500 million as adoption expands and price competition is managed.
- Entry into new indications, such as other gastrointestinal procedures, could augment revenue streams.
- The impact of patent expirations and generics could compress prices but increase overall volume.
Regulatory and Market Expansion Factors
- Regulatory approvals in China, India, and Brazil are crucial for growth.
- Reimbursement policies and clinical guideline updates supporting PREPOPIK use enhance market uptake.
- Innovative formulations or combination regimens could open new segments.
Key Takeaways
- PREPOPIK holds a significant position in the bowel prep market, characterized by its single-dose convenience.
- The market is expected to grow at a CAGR of 6.2%, reaching $1.2 billion globally by 2028.
- Revenue is forecasted to reach approximately $288 million by 2025, with long-term potential exceeding $400 million.
- Competition from PEG-based solutions remains dominant, but PREPOPIK's advantages position it favorably.
- Regional expansion and regulatory approvals are pivotal to achieving long-term sales growth.
FAQs
1. What are the main competitors to PREPOPIK?
Main competitors include MiraLAX, GoLYTELY, and Suprep, which dominate the market with different efficacy and dosing profiles.
2. How does PREPOPIK compare price-wise with alternatives?
PREPOPIK tends to be more expensive per dose but offers better patient adherence, potentially offsetting higher costs with improved compliance.
3. Which regions present the biggest growth opportunities?
Emerging markets in Asia-Pacific, Latin America, and parts of Europe are poised for increased adoption due to rising healthcare infrastructure.
4. How will patent expirations impact PREPOPIK sales?
Patent expirations could lead to generic competition, likely reducing prices but also increasing market volume.
5. What regulatory hurdles remain?
Approval in major markets such as China, India, and Brazil remains critical; differences in clinical guidelines could influence sales trajectory.
References
[1] MarketResearch.com. (2022). Global Bowel Preparation Market Outlook.
[2] Ferring Pharmaceuticals. (2022). PREPOPIK product dossier.
[3] IQVIA. (2023). GI and colonscopy drug sales data.
[4] GlobalData. (2022). Gastrointestinal drugs market analysis.
[5] World Health Organization. (2021). Colorectal cancer screening programs.
More… ↓
