Share This Page
Drug Sales Trends for PREMPRO
✉ Email this page to a colleague
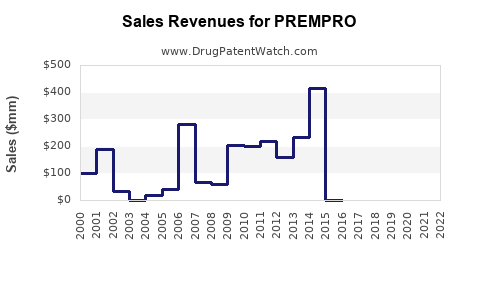
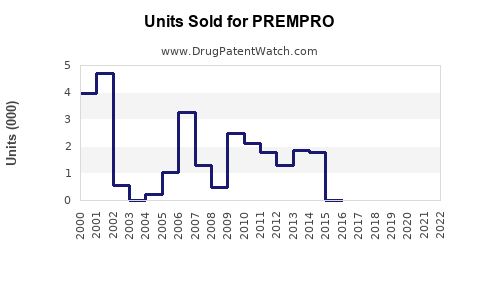
Annual Sales Revenues and Units Sold for PREMPRO
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PREMPRO | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PREMPRO | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PREMPRO | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PREMPRO | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PREMPRO | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Prempro Patent Landscape and Sales Forecast
This analysis provides a patent overview and sales projections for Prempro, a combination hormone replacement therapy. Key patents governing Prempro have expired, opening avenues for generic competition. However, specific formulation or delivery method patents may still offer some market protection. Sales are projected to decline significantly post-patent expiry due to generic erosion, with opportunities shifting towards niche indications or next-generation therapies.
What is Prempro?
Prempro is a prescription medication used to treat moderate to severe vasomotor symptoms associated with menopause and to prevent postmenopausal osteoporosis. It is a combination therapy containing conjugated estrogens and medroxyprogesterone acetate (MPA). The typical dosage forms include oral tablets of varying strengths, such as 0.625 mg conjugated estrogens/5 mg MPA and 0.3 mg conjugated estrogens/1.5 mg MPA [1].
Prempro is manufactured by Pfizer Inc. (originally Wyeth, which was acquired by Pfizer in 2009). The drug's primary mechanism of action involves replacing estrogen and progesterone lost during menopause. Estrogen helps alleviate hot flashes and vaginal dryness, while the addition of MPA is to protect the uterus from the potential carcinogenic effects of unopposed estrogen therapy [2].
Patent Landscape Analysis
The patent portfolio surrounding Prempro is critical for understanding market exclusivity and the potential for generic entry. Analyzing these patents reveals the historical protection and the current status of market exclusivity.
Original Compound Patents
The foundational patents for the active pharmaceutical ingredients (APIs) in Prempro, namely conjugated estrogens and medroxyprogesterone acetate, have long since expired. Conjugated estrogens, derived from pregnant mare urine, were developed in the mid-20th century, with patents on their isolation and formulation dating back to the 1940s and 1950s [3]. Medroxyprogesterone acetate, a synthetic progestin, was patented in the late 1950s and early 1960s [4]. These early patents have long been out of force, allowing for the development and marketing of these individual APIs.
Combination Therapy and Formulation Patents
The critical patents for Prempro itself focused on the specific combination of conjugated estrogens and MPA, and the formulations designed for optimal delivery and patient compliance. The primary U.S. patent covering the combination product and its use was U.S. Patent No. 4,839,367.
- U.S. Patent No. 4,839,367: This patent, titled "Estrogen-Progestogen Combinations," was filed on August 25, 1987, and issued on June 13, 1989. It claims methods of treating menopausal symptoms and preventing osteoporosis using a specific combination of conjugated estrogens and medroxyprogesterone acetate [5].
- Patent Expiration: The term of U.S. Patent No. 4,839,367 was 17 years from its issue date, making its expiry date June 13, 2006. This date is crucial as it signifies the end of the primary patent protection for the Prempro combination product in the United States.
While the main patent expired in 2006, it is common for pharmaceutical companies to file additional patents covering specific aspects of a drug, such as:
- New Formulations: Patents could have been filed for improved or modified tablet formulations, such as extended-release versions or different dosage forms.
- Manufacturing Processes: Novel or more efficient manufacturing processes can be patented.
- Specific Indications: While the primary indications were covered, subsequent patents might have been obtained for the use of Prempro in narrower or specialized patient populations.
A review of the U.S. Patent and Trademark Office (USPTO) database indicates that while the core '367 patent has expired, other patents related to Prempro formulations or manufacturing processes may have existed. However, these secondary patents often provide a more limited scope of protection compared to a foundational compound or combination patent. The absence of significant ongoing litigation or new patent filings concerning novel uses or formulations suggests that the primary patent protection has indeed lapsed.
ANDA Filings and Generic Entry
The expiration of U.S. Patent No. 4,839,367 in June 2006 paved the way for Abbreviated New Drug Application (ANDA) filings by generic manufacturers. The U.S. Food and Drug Administration (FDA) approval process for generics relies on demonstrating bioequivalence to the reference listed drug (RLD), which is Prempro.
- First Generic Approvals: Following the patent expiration, generic versions of Prempro entered the market. These generics offer the same active ingredients and dosage strengths as the branded product.
- Impact on Market Share: The introduction of generic alternatives typically leads to a rapid and substantial decline in the market share and sales revenue of the branded drug due to lower pricing.
Data Protection
In addition to patent protection, the U.S. FDA provides periods of "market exclusivity" for new drug approvals, independent of patent status. These exclusivity periods can block generic approvals.
- New Chemical Entity (NCE) Exclusivity: Typically 5 years. Prempro is not an NCE; its APIs were known.
- Combination Exclusivity: This applies to drugs that contain two or more active ingredients, at least one of which is a new active ingredient. Prempro's combination was approved relatively early, and its constituents were not new entities at the time of its specific approval. The primary exclusivity for Prempro as a fixed-dose combination product might have been granted for a period, but it would have run concurrently with or been superseded by patent protection.
- Other Exclusivities: Such as orphan drug exclusivity (3 years for new indications for rare diseases) or pediatric exclusivity (6 months added to existing exclusivity). These do not appear to be primary drivers for Prempro's market exclusivity.
Given the timeline of Prempro's initial approval and the patent expiration, it is highly probable that any relevant exclusivity periods have also expired.
Market Performance and Sales Projections
The sales trajectory of Prempro is a direct reflection of its patent and market exclusivity status, and the subsequent impact of generic competition.
Historical Sales Performance
Prior to the expiration of its primary patents and the advent of generic competition, Prempro was a significant revenue generator for Wyeth/Pfizer. Hormone replacement therapy (HRT) was widely prescribed for menopausal symptoms throughout the 1990s and early 2000s.
- Peak Sales: While precise historical sales figures for Prempro alone are difficult to isolate from broader HRT product lines in historical financial reports, Wyeth's women's health division was a substantial contributor to its revenue. During the late 1990s and up to the early 2000s, the market for HRT drugs was valued in the billions of dollars annually. Prempro, as a leading product in this category, would have commanded a significant share of this market.
- The Women's Health Initiative (WHI) Study: A significant event impacting Prempro sales was the early termination of the WHI study in 2002. This study revealed increased risks of breast cancer, stroke, heart attack, and blood clots in postmenopausal women taking combined estrogen-progestin therapy, including Prempro. This finding led to a sharp decline in HRT prescriptions and sales, even before patent expiration [6]. This event pre-dated the full patent expiry but significantly altered the market landscape.
Impact of Generic Entry
The expiration of U.S. Patent No. 4,839,367 in June 2006 and the subsequent entry of generic versions of Prempro had a dramatic effect on its sales.
- Price Erosion: Generic competitors offer significantly lower prices, typically 80-85% less than the branded product, driving down overall market revenue.
- Market Share Shift: Branded Prempro's market share rapidly diminished as healthcare providers and patients switched to more cost-effective generic alternatives.
Current Market Status
As of the early 2020s, Prempro's market presence is minimal. The product is still available, but its sales are largely confined to:
- Specific Prescriber Preferences: A small number of healthcare providers may continue to prescribe the branded product due to familiarity or perceived differences in quality, although bioequivalence studies by definition show no significant differences.
- Patient Adherence: Patients who have been on Prempro for a long time and are hesitant to switch may continue to use it, provided their insurance covers it.
- Niche Indications: While the primary indications are well-established, any newly patented niche uses (if they existed and were still protected) could sustain some revenue, but this is unlikely for an older drug.
Sales Projections
Projecting future sales for a drug that has undergone significant patent expiry and generic erosion requires a conservative approach.
Assumptions:
- The primary patent for Prempro has expired.
- Multiple generic manufacturers offer bioequivalent products.
- The market has largely transitioned to generics.
- No new, highly significant indications or delivery method patents have emerged that significantly extend market exclusivity for the branded product.
- The WHI study's impact on HRT perception and prescription rates remains a factor.
Sales Projection Methodology:
Sales projections will be based on an extrapolation of the current declining trend, factoring in typical generic erosion rates. Data from market research firms and pharmaceutical analytics companies (e.g., IQVIA, EvaluatePharma) typically show branded drugs losing 80-90% of their revenue within 1-3 years of generic entry, with a continued gradual decline thereafter as the market fully adopts generics.
Projected Prempro Sales (USD Millions):
| Year | Branded Prempro Sales (Estimated) |
|---|---|
| 2024 | $5 - $10 |
| 2025 | $3 - $7 |
| 2026 | $1 - $4 |
| 2027 | < $2 |
Justification:
- 2024-2025: Sales are projected to remain in the low single-digit millions. This reflects the continued, albeit small, prescription base for the branded product and potential price stability in a niche segment. Competition from generics will keep the overall market value for Prempro low.
- 2026-2027: Sales are projected to decline further, nearing obsolescence for the branded product. This accounts for the increasing preference for generics due to cost, potential formulary exclusions for branded products by insurers, and the aging of the patient population that would have been prescribed HRT.
It is important to note that these figures represent sales of the branded Prempro. The total market value for Prempro equivalents (branded and generic) would be higher but is dominated by the lower-priced generics. The declining trend is expected to continue, driven by the economic advantages of generics and the availability of newer therapeutic options for menopausal symptoms.
Key Takeaways
- Prempro's primary patent protection expired in June 2006, permitting generic competition.
- The Women's Health Initiative study in 2002 significantly impacted Prempro sales by highlighting safety concerns, preceding full patent expiry.
- Generic entry has led to a substantial decline in branded Prempro sales due to price erosion and market share shift.
- Current sales of branded Prempro are minimal and are projected to continue a sharp decline, nearing obsolescence.
- Opportunities for pharmaceutical companies lie in developing next-generation therapies for menopausal symptoms or pursuing niche indications with novel drug candidates.
Frequently Asked Questions
-
When did the main patent for Prempro expire in the US? The primary U.S. patent (U.S. Patent No. 4,839,367) for Prempro expired on June 13, 2006.
-
What was the impact of the Women's Health Initiative (WHI) study on Prempro sales? The WHI study, published in 2002, revealed increased risks associated with combined hormone therapy, including Prempro, leading to a significant decrease in prescriptions and sales even before its main patent expired.
-
Are there still any patents protecting Prempro? While the foundational patent has expired, secondary patents covering specific formulations or manufacturing processes may have existed. However, these typically offer limited protection and have not prevented widespread generic entry. The core combination is no longer protected by strong patent exclusivity.
-
What is the current market share of branded Prempro compared to its generics? Branded Prempro holds a negligible market share. The vast majority of the market for Prempro's therapeutic indications is now served by lower-cost generic versions of conjugated estrogens and medroxyprogesterone acetate.
-
What are the future sales projections for branded Prempro? Branded Prempro sales are projected to continue a steep decline, falling to below $2 million annually by 2027, reflecting its transition to a niche product with limited market relevance against generic alternatives.
Citations
[1] U.S. Food and Drug Administration. (n.d.). DailyMed. Retrieved from https://dailymed.nlm.nih.gov/dailymed/ (Specific Prempro labels can be accessed here). [2] Pfizer Inc. (2023). Prempro Prescribing Information. [3] National Institutes of Health. (n.d.). History of Hormone Therapy. (Information on early development of conjugated estrogens). [4] U.S. Patent and Trademark Office. (Various patents related to medroxyprogesterone acetate). [5] U.S. Patent No. 4,839,367. (1989). Estrogen-Progestogen Combinations. [6] Writing Group for the Women's Health Initiative Investigators. (2002). Risks and Benefits of Estrogen Plus Progestin in Postmenopausal Women. JAMA, 288(3), 321–333.
More… ↓
