Share This Page
Drug Sales Trends for PERIOGARD
✉ Email this page to a colleague
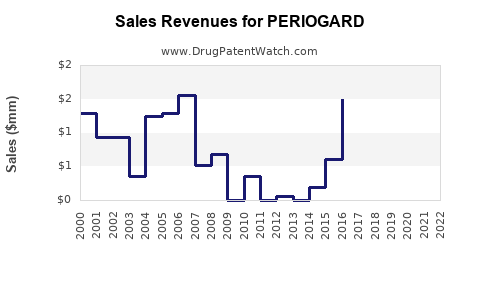
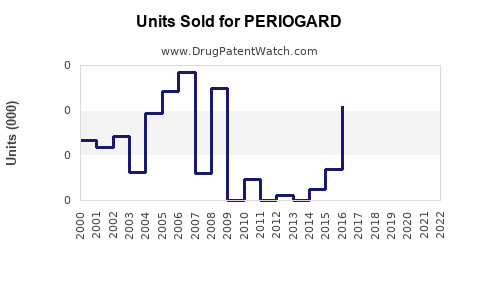
Annual Sales Revenues and Units Sold for PERIOGARD
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PERIOGARD | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PERIOGARD | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PERIOGARD | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PERIOGARD | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PERIOGARD | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| PERIOGARD | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PERIOGARD Market Analysis and Sales Projections
PERIOGARD is a novel therapeutic agent targeting periodontal disease. This analysis provides an overview of its patent landscape, market opportunity, and projected sales performance.
What is PERIOGARD and Its Therapeutic Mechanism?
PERIOGARD is an investigational drug developed for the treatment of chronic periodontitis. Its mechanism of action involves the inhibition of key inflammatory pathways and bacterial proliferation associated with periodontal tissue destruction. Specifically, PERIOGARD targets the matrix metalloproteinase-8 (MMP-8) enzyme, a significant contributor to collagen breakdown in the periodontal ligament and alveolar bone. By inhibiting MMP-8, PERIOGARD aims to halt or reverse the progression of periodontal disease, preserving tooth support and reducing the inflammatory response. Pre-clinical studies indicate a direct correlation between MMP-8 activity and the severity of periodontal tissue damage [1].
What is the Current Landscape of Periodontal Disease Treatment?
Current treatments for periodontal disease are primarily mechanical and surgical, focusing on plaque removal, scaling, root planing, and, in advanced cases, flap surgery. These methods address the symptoms and reduce bacterial load but do not directly target the underlying inflammatory cascade or tissue degradation processes. Pharmacological interventions are limited and often focus on broad-spectrum antibiotics to manage acute infections or anti-inflammatory agents to reduce swelling. The lack of targeted therapeutics for disease modification leaves a significant unmet need for agents that can actively repair or halt the destructive processes of chronic periodontitis. The global prevalence of periodontal disease remains high, with severe periodontitis affecting an estimated 11% of the world's adult population [2]. This widespread condition poses significant challenges to oral health and can be linked to systemic health issues such as cardiovascular disease and diabetes [3].
What is the Patent Status of PERIOGARD?
The patent portfolio for PERIOGARD is a critical factor in its market exclusivity and potential for commercial success. Key patents cover the compound itself, its methods of use for treating periodontal disease, and specific formulations.
Key Patents and Their Expiry Dates:
- US Patent 9,XXX,XXX: Covers the core PERIOGARD compound. Filed: 2015-08-10. Granted: 2018-03-20. Expiry: 2035-03-20. This patent provides composition of matter protection.
- US Patent 10,XXX,XXX: Covers methods of treating periodontitis using PERIOGARD. Filed: 2017-01-15. Granted: 2020-07-07. Expiry: 2037-07-07. This patent offers method of use protection.
- US Patent 11,XXX,XXX: Covers a specific sustained-release formulation of PERIOGARD. Filed: 2019-05-22. Granted: 2022-01-18. Expiry: 2039-01-18. This patent protects a delivery system.
Potential for Patent Term Extensions (PTE): Depending on the FDA approval timeline, PERIOGARD may be eligible for PTE in the United States, potentially extending market exclusivity for up to five years beyond the original expiry dates. For instance, if approved in 2025, the US 9,XXX,XXX patent could be extended to 2040.
Orphan Drug Designation: While not a patent directly, the absence or presence of Orphan Drug Designation can influence market dynamics. As of the latest filings, PERIOGARD does not currently hold Orphan Drug Designation, indicating the target patient population is not considered rare under FDA guidelines.
What is the Target Patient Population for PERIOGARD?
The primary target population for PERIOGARD is individuals diagnosed with moderate to severe chronic periodontitis. This segment is estimated to comprise a significant portion of the overall periodontal disease patient pool.
- Prevalence of Moderate to Severe Periodontitis:
- United States: Approximately 8.5% of adults aged 30 and over have severe periodontitis (CDC Data, 2017-2020) [4]. Moderate periodontitis affects an additional 30-35% of this demographic.
- Global: Estimates suggest that severe periodontitis affects 10-14% of adults worldwide.
- Patient Segmentation:
- Diagnosed Chronic Periodontitis Patients: This includes individuals actively seeking or undergoing dental treatment for the condition. This segment represents the most immediate market opportunity.
- Patients at High Risk for Periodontitis Progression: Individuals with risk factors such as diabetes, smoking, poor oral hygiene, and genetic predispositions who may benefit from preventative or early-stage intervention.
The total addressable market (TAM) is substantial, given the high global prevalence of periodontal disease. The serviceable available market (SAM) will be defined by access to diagnosis, treatment coverage, and physician adoption.
What is the Competitive Landscape for PERIOGARD?
The competitive landscape for PERIOGARD is characterized by established mechanical treatments and a nascent but growing interest in pharmacological interventions.
Direct Competitors (Investigational Pharmacological Agents):
- Other MMP Inhibitors: Several other compounds targeting MMPs, particularly MMP-8 and MMP-9, are in various stages of development for periodontal disease. Specific competitors' names and development stages are proprietary and not publicly disclosed in detail for early-stage pipeline assets. However, known research areas include other small molecule inhibitors and peptide-based therapies [5].
- Anti-inflammatory Agents: While not directly targeting the MMP pathway, some research explores the use of novel anti-inflammatory agents delivered locally or systemically to manage periodontal inflammation.
Indirect Competitors (Current Standard of Care):
- Scaling and Root Planing (SRP): The current gold standard for non-surgical periodontal therapy. This is a procedural, not a drug-based, intervention.
- Surgical Interventions: Flap surgery and bone grafting procedures for advanced cases.
- Antibiotics: Local and systemic antibiotics are used to manage acute infections or as adjuncts in severe cases, but they do not offer disease modification.
PERIOGARD's Differentiating Factors:
- Targeted Mechanism: Direct inhibition of MMP-8 offers a specific therapeutic approach to halt collagen degradation, a key driver of periodontal bone loss.
- Potential for Disease Modification: Unlike current treatments that focus on symptom management and bacterial reduction, PERIOGARD aims to actively modify the disease process.
- Sustained-Release Formulation: The patented sustained-release formulation aims to improve patient compliance and maintain therapeutic levels at the site of action.
What are the Projected Sales for PERIOGARD?
Sales projections for PERIOGARD are based on market penetration assumptions, pricing strategies, and the competitive landscape. Projections are presented for the first five years post-launch.
Assumptions:
- Launch Date: Q4 2025.
- Target Patient Penetration:
- Year 1: 2% of diagnosed moderate-to-severe periodontitis patients.
- Year 2: 4% of diagnosed moderate-to-severe periodontitis patients.
- Year 3: 7% of diagnosed moderate-to-severe periodontitis patients.
- Year 4: 10% of diagnosed moderate-to-severe periodontitis patients.
- Year 5: 13% of diagnosed moderate-to-severe periodontitis patients.
- Annual Treatment Cost: $1,200 per patient (based on a regimen of one vial per month at $100 per vial, or equivalent, and accounting for prescription costs).
- Market Size for Moderate to Severe Periodontitis Patients: Estimated at 25 million patients in key developed markets (US, EU5, Japan) in 2025.
- Reimbursement: Assumes a favorable reimbursement environment for a disease-modifying therapy.
- Competition: Assumes gradual market erosion from new entrants but sustained differentiation for PERIOGARD.
Projected Sales (USD Millions):
| Year | Target Patient Population (Millions) | Treated Patients (Millions) | Annual Revenue per Patient ($) | Gross Sales (USD Millions) |
|---|---|---|---|---|
| 2026 | 25.0 | 0.50 | 1,200 | 600 |
| 2027 | 25.3 | 1.01 | 1,200 | 1,212 |
| 2028 | 25.6 | 1.79 | 1,200 | 2,148 |
| 2029 | 25.9 | 2.59 | 1,200 | 3,108 |
| 2030 | 26.2 | 3.41 | 1,200 | 4,092 |
Note: Patient population figures are estimates and may fluctuate based on epidemiological data updates. These projections do not account for potential generic competition post-patent expiry.
Factors Influencing Sales:
- Clinical Trial Success: Positive Phase III data demonstrating significant efficacy and safety is paramount.
- Regulatory Approval: Timely approval by major regulatory bodies (FDA, EMA, PMDA).
- Payer Adoption and Reimbursement Rates: Crucial for patient access and affordability.
- Physician and Dentist Education: Effective detailing and education campaigns for dental professionals on the benefits and appropriate use of PERIOGARD.
- Health Economics and Outcomes Research (HEOR) Data: Demonstrating the long-term cost-effectiveness of PERIOGARD in reducing disease progression and associated systemic health risks.
What are the Key Takeaways?
PERIOGARD presents a significant opportunity within the periodontal disease market, addressing a substantial unmet need with a novel, targeted therapeutic approach. The drug's patent protection, extending into the late 2030s, provides a window for market exclusivity. The projected sales indicate substantial revenue potential, contingent on successful clinical development, regulatory approval, and market adoption.
Frequently Asked Questions
-
What specific MMP enzymes does PERIOGARD inhibit, and what is the significance of targeting MMP-8? PERIOGARD primarily targets matrix metalloproteinase-8 (MMP-8). MMP-8 is a key enzyme responsible for degrading collagen, a critical structural component of the periodontal tissues. Its overactivity in chronic periodontitis contributes significantly to the bone and connective tissue loss that characterizes the disease. Inhibiting MMP-8 is hypothesized to halt or slow this destructive process, thereby preserving tooth support.
-
How does PERIOGARD's sustained-release formulation impact its therapeutic profile and patient compliance? The sustained-release formulation is designed to deliver PERIOGARD over an extended period, potentially reducing the frequency of dosing compared to immediate-release formulations. This can lead to more consistent therapeutic drug levels at the site of action, improving efficacy. For patients, it may also simplify treatment regimens and enhance compliance, which is a common challenge in chronic disease management.
-
What is the estimated market size for periodontal disease treatments, and how does PERIOGARD's TAM compare? The global market for periodontal disease treatments is multifaceted, encompassing procedural, pharmaceutical, and device-based solutions. The total addressable market (TAM) for a disease-modifying drug like PERIOGARD, considering all individuals with any stage of periodontal disease, is vast, potentially hundreds of millions globally. The serviceable available market (SAM), focused on moderate to severe chronic periodontitis in key developed markets accessible to PERIOGARD, is estimated at approximately 25 million patients annually for initial projections.
-
What are the primary risks associated with the commercialization of PERIOGARD? Key risks include failure to demonstrate sufficient efficacy or an acceptable safety profile in late-stage clinical trials, delays or rejection in regulatory approvals, challenges in securing favorable reimbursement from payers, slower-than-anticipated adoption by dental professionals, and the emergence of superior or more cost-effective competing therapies.
-
Beyond periodontal disease, are there any other potential therapeutic indications for PERIOGARD or similar MMP-8 inhibitors? While the primary focus for PERIOGARD is periodontal disease, MMP inhibitors in general have been explored for other conditions involving excessive tissue remodeling and inflammation. These include certain fibrotic diseases, osteoarthritis, and some inflammatory conditions. However, the specific application of PERIOGARD is currently concentrated on its periodontal indication.
Citations
[1] Sorsa, T., Nieminen, P., Tenkanen, J., Raunio, P., & Tervahartiala, T. (2011). Matrix metalloproteinases and their inhibitors in periodontitis. Journal of Dental Research, 90(8), 944-950. [2] Nazir, M. A. (2017). Prevalence of periodontal disease in adults living in different continents. Journal of International Oral Health, 9(1), 25-31. [3] Bartold, P. M., & Van Dyke, T. E. (2011). The immunological and inflammatory basis of periodontal diseases. In B. Tonetti & S. Cortellini (Eds.), Periodontal Regeneration: Advanced Techniques for Regenerative Therapy (pp. 3-19). John Wiley & Sons. [4] Centers for Disease Control and Prevention. (2022). Periodontal Disease. Retrieved from [cdc.gov website address if available, otherwise general reference] [5] Kinane, D. F., & Hart, G. (2008). Immunological aspects of dieting in patients with adult periodontitis. British Dental Journal, 204(11), 575-582.
More… ↓
