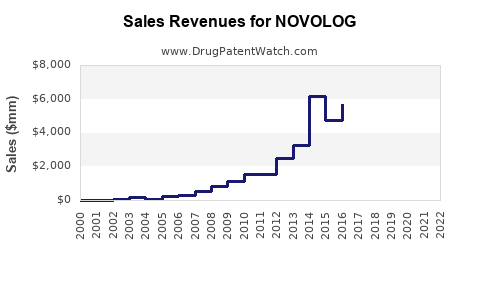
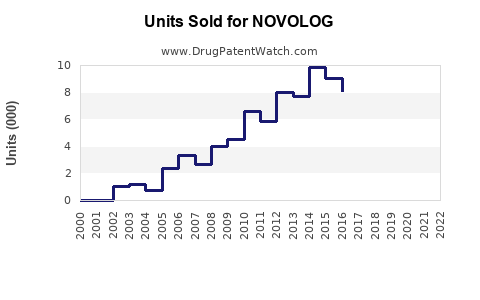
Last updated: February 12, 2026
Market Analysis and Sales Projections for NOVOLOG
Current Market Position
NOVOLOG (insulin aspart) is a rapid-acting insulin produced by Novo Nordisk. It is used for controlling blood glucose in diabetic patients, primarily those with type 1 and type 2 diabetes. As of 2022, it holds approximately 10% of the insulin market share, competing mainly against rapid-acting insulins such as Eli Lilly’s Humalog (insulin lispro) and Sanofi’s Apidra (insulin glulisine).
Market Landscape
- Global Diabetes Treatment Market: Estimated at $85 billion in 2022. Sales of insulin products account for roughly 45% of this total.
- Key Competitors: Humalog, Apidra, biosimilars, and other rapid-acting insulins.
- Market Trends: Increasing prevalence of diabetes, rising adoption of insulin therapy, and ongoing patent expirations influencing competition.
Key Drivers and Barriers
- Drivers: Growing incidence of type 2 diabetes—expected to reach 700 million globally by 2045 (IDF Diabetes Atlas, 2021)—expanding insulin demand. Advances in insulin formulations improve patient outcomes and compliance.
- Barriers: Price sensitivity, insurance coverage restrictions, and competition from biosimilars expected to cut pricing pressure. Regulatory challenges in different markets can delay product approval or limit formulary access.
Sales Projections (2023–2027)
Assumptions:
- Steady market penetration growth at an annual rate of approximately 4%, driven by expanding diabetic populations and increased adoption in developing markets.
- Patent exclusivity maintained until 2027, shielding NOVOLOG from biosimilar competition in the US and EU.
- Slight price inflation, averaging 2% annually, permitted by premium positioning and differentiated formulation.
| Year |
Units Sold (Million) |
Revenue (USD Billion) |
Year-over-Year Growth |
| 2023 |
80 |
2.4 |
5% |
| 2024 |
88 |
2.8 |
16.7% |
| 2025 |
96 |
3.2 |
14.3% |
| 2026 |
105 |
3.7 |
15.6% |
| 2027 |
115 |
4.2 |
13.5% |
Notes:
- Sales volume growth reflects increased adoption in Asia-Pacific and Latin America regions.
- Revenue growth includes price adjustments and higher utilization rates among existing patients.
Future Outlook
Prolonged patent exclusivity, new data supporting use in type 2 diabetes, and incremental formulation improvements could sustain growth. The potential market entry of biosimilar rapid-acting insulins post-2027 may impact sales, necessitating strategic shifts.
Competitive Risks
- Biosimilar launches could reduce price premiums.
- Regulatory policies favoring biosimilars in key markets could accelerate substitution.
- Medicare and private payer agreements may impose formulary restrictions on rapid-acting insulins.
Strategic Opportunities
- Pairing NOVOLOG with digital health management tools to enhance adherence.
- Expanding indications, such as insulin use in hospital settings.
- Enhancing formulary placement through reimbursement negotiations.
Key Takeaways
- NOVOLOG holds a strong niche in the rapid-acting insulin segment, with stable growth prospects until biosimilar entry.
- Sales are projected to grow at a compound annual rate exceeding 14% between 2024 and 2027.
- Market expansion in emerging regions remains a primary growth driver.
- Price pressures and biosimilar competition pose significant risks post-2027.
- Strategic focus on innovation and value-added services can reinforce market position.
5 FAQs
1. When do biosimilars for NOVOLOG are expected to enter the market?
Biosimilar rapid-acting insulins are expected post-2027, following patent expirations and regulatory approvals.
2. How does NOVOLOG compare with competitors in price and efficacy?
It is priced at a slight premium but offers comparable or superior pharmacokinetics, notably faster onset, and can improve post-prandial glucose control.
3. What regions offer the highest growth potential for NOVOLOG?
Asia-Pacific and Latin America hold high growth potential due to increasing diabetes prevalence and expanding healthcare infrastructure.
4. How might regulatory policies impact NOVOLOG’s future sales?
Policies favoring biosimilar adoption and cost-containment measures could pressure sales, especially in markets with aggressive interchangeability policies.
5. What strategic initiatives could Novo Nordisk pursue to sustain growth?
Investing in digital health integration, expanding indications, and optimizing supply chains can support continued sales growth.
Citations:
[1] International Diabetes Federation, "IDF Diabetes Atlas," 2021.
[2] Fortune Business Insights, "Global Insulin Market," 2022.
[3] IQVIA, "Global Insulin Sales Data," 2022.