Share This Page
Drug Sales Trends for MOVIPREP
✉ Email this page to a colleague
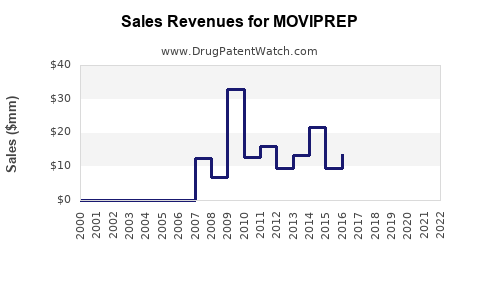
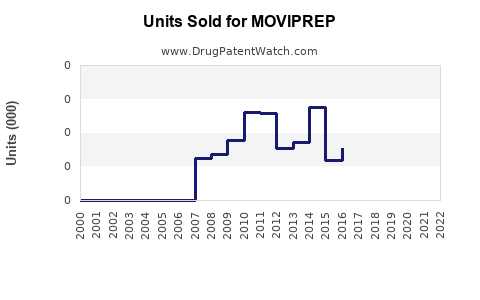
Annual Sales Revenues and Units Sold for MOVIPREP
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MOVIPREP | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MOVIPREP | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MOVIPREP | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
MOVIPREP: Market Analysis and Sales Projections
MOVIPREP is a polyethylene glycol-based bowel preparation solution used prior to colonoscopy. Its market performance is directly tied to the incidence of colorectal cancer screening and the procedural volume of colonoscopies. Key patent expirations and generic competition will significantly impact future market share and pricing.
What is the Current Market Landscape for MOVIPREP?
MOVIPREP competes in the bowel preparation market, a segment driven by the increasing prevalence of gastrointestinal disorders and the growing emphasis on preventative healthcare, particularly colorectal cancer screening. The global market for bowel preparation products was valued at approximately $2.5 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 5.8% through 2030.
MOVIPREP's primary indication is for cleansing the colon in preparation for colonoscopy. Its formulation, involving a large volume of electrolyte-balanced solution, aims to ensure patient compliance and effective bowel cleansing.
The competitive landscape includes other polyethylene glycol (PEG)-based solutions, such as MiraLAX (polyethylene glycol 3350), and combination products, like those involving sodium picosulfate and magnesium citrate. Emerging technologies, including lower-volume formulations and novel delivery systems, are also gaining traction.
The prescribing patterns for MOVIPREP are influenced by physician preference, formulary restrictions imposed by payers, and patient tolerability. Clinical studies have compared MOVIPREP to other bowel preparation agents, with varying outcomes regarding efficacy and tolerability.
What are MOVIPREP's Key Patents and Their Expiration Dates?
The patent protection for MOVIPREP is a critical factor in its market exclusivity and subsequent genericization. The primary patent for the MOVIPREP formulation is U.S. Patent No. 6,946,147, which was granted on September 20, 2005.
- U.S. Patent No. 6,946,147: This patent covers the composition of MOVIPREP, specifically its formulation of polyethylene glycol, electrolytes, and flavorings designed for osmotic laxative effect.
- Expiration: The term of this patent expired on September 20, 2022.
- Pediatric Exclusivity: Additional periods of exclusivity, such as those granted for pediatric studies, may have extended the effective market protection for a limited time, but the core formulation patent has now expired.
The expiration of these key patents has opened the door for generic manufacturers to enter the market. The first generic versions of MOVIPREP became available following the expiration of the primary patent.
How Will Generic Competition Impact MOVIPREP's Sales?
The introduction of generic MOVIPREP is expected to exert significant downward pressure on its sales and market share. Historically, the market for branded pharmaceuticals experiences a substantial decline in revenue and volume once generic alternatives become available.
- Price Erosion: Generic versions are typically priced at a significant discount to the branded product. This price differential encourages payers and providers to switch to the lower-cost alternatives. Estimates suggest that branded drugs can lose 70% to 90% of their market share within the first year of generic entry.
- Market Share Shift: Healthcare providers and pharmacy benefit managers (PBMs) often implement formulary changes that favor generics, further accelerating the shift away from the branded product.
- Volume Decline: While the overall volume of bowel preparation procedures may increase, the proportion attributable to branded MOVIPREP will likely decrease substantially.
For MOVIPREP, this translates to a projected sharp decline in revenue. The revenue loss will be most pronounced in markets where generic approvals and launches have occurred. Regions with longer patent protection or slower generic uptake will experience a more gradual decline.
What are the Projected Sales for MOVIPREP in the Coming Years?
Projecting precise sales figures for a branded drug facing generic competition is challenging and subject to numerous variables. However, based on industry trends and the impact of genericization, a significant decline in branded MOVIPREP sales is anticipated.
Pre-Genericization (e.g., 2022): In the years leading up to patent expiration, branded MOVIPREP sales were likely in the range of $300 million to $400 million annually in key markets.
Post-Genericization (e.g., 2024-2027):
- 2024: Branded MOVIPREP sales are projected to fall by 60-75% from pre-genericization levels, potentially reaching $100 million to $150 million.
- 2025: Further erosion is expected, with sales potentially dropping to $50 million to $80 million.
- 2026 onwards: Sales of branded MOVIPREP are likely to stabilize at a much lower level, serving a niche market of physicians or patients who specifically request or tolerate the branded product, or in regions with delayed generic entry. Projections suggest sales could be below $30 million annually.
These projections assume a typical market response to generic competition. Factors that could influence these figures include:
- Payer Policies: Aggressive formulary management by PBMs and insurers will accelerate generic adoption.
- Physician Acceptance: The speed at which physicians adapt to prescribing generic alternatives.
- Generic Product Availability and Quality: The number of generic entrants and their perceived product quality can influence switching rates.
- Promotional Efforts: The extent to which the branded manufacturer continues marketing efforts for MOVIPREP.
The total market for bowel preparation products, however, is expected to continue growing due to the factors mentioned earlier. The decline in branded MOVIPREP sales will be offset, to some degree, by the sales of generic MOVIPREP and the growth of competing bowel preparation products.
What are the Regulatory and Clinical Considerations for MOVIPREP?
The regulatory pathway for bowel preparation agents involves demonstrating safety and efficacy for their intended use. For MOVIPREP, this primarily relates to its approval by regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Key Regulatory Aspects:
- FDA Approval: MOVIPREP (as SUPREP) received FDA approval in 2006. Post-marketing surveillance and any adverse event reporting are ongoing regulatory considerations.
- Labeling: Regulatory agencies scrutinize product labeling, including instructions for use, contraindications, warnings, and precautions. Changes to labeling or post-market requirements could impact sales.
- Generic Approval: The abbreviated new drug application (ANDA) process for generic MOVIPREP requires bioequivalence studies to demonstrate that the generic product performs comparably to the branded drug.
Clinical Considerations:
- Efficacy: Clinical trials have established MOVIPREP's efficacy in achieving adequate bowel cleansing for colonoscopy. However, comparative studies with newer or alternative agents are ongoing and can influence physician choice.
- Tolerability and Patient Compliance: Patient acceptance of large-volume preparations can be a challenge. Factors such as taste, nausea, and abdominal discomfort can impact compliance. Formulation improvements or alternative taste profiles in generic versions may affect patient preference.
- Adverse Events: Common adverse events include nausea, vomiting, abdominal pain, and bloating. Serious adverse events, though rare, can include electrolyte imbalances and renal issues, particularly in patients with pre-existing conditions. Post-market data on adverse events can influence prescribing and regulatory actions.
- Emerging Alternatives: The development of lower-volume bowel preparations, oral tablet formulations, and agents with different mechanisms of action presents ongoing clinical and competitive challenges to established products like MOVIPREP.
What is the Future Outlook for MOVIPREP and its Market Segment?
The future outlook for branded MOVIPREP is one of significant decline due to the loss of patent exclusivity. However, the overall market for bowel preparation agents is expected to continue expanding.
Branded MOVIPREP: The trajectory for branded MOVIPREP is characterized by a sharp and sustained decrease in sales and market share. Its role will likely diminish to that of a legacy product, with limited commercial viability as generics capture the majority of the market.
Generic MOVIPREP: Generic versions of MOVIPREP will capture the bulk of the market volume. The competitive landscape among generic manufacturers will likely lead to further price reductions within the generic segment. The success of generic MOVIPREP will depend on manufacturing quality, supply chain reliability, and marketing efforts by generic companies.
Overall Bowel Preparation Market: The broader bowel preparation market will continue to grow, driven by:
- Aging Population: An increasing elderly population leads to a higher incidence of gastrointestinal conditions requiring screening and intervention.
- Colorectal Cancer Screening Guidelines: Recommendations for earlier and more frequent colorectal cancer screening by organizations like the American Cancer Society and the U.S. Preventive Services Task Force will drive procedural volume.
- Technological Advancements: Development of more convenient, palatable, and effective bowel preparation agents could expand the market or shift preferences away from older formulations.
- Increased Access to Healthcare: Improved access to healthcare services globally will contribute to higher colonoscopy rates.
Competitive Dynamics: The market will see continued competition from various product classes, including:
- PEG-based solutions: Both branded and generic.
- Sodium Picosulfate/Magnesium Citrate combinations.
- Newer, lower-volume formulations.
- Oral tablet preparations.
Companies involved in the bowel preparation market will need to adapt to these dynamics through innovation, strategic partnerships, and efficient manufacturing and distribution.
Key Takeaways
- Branded MOVIPREP's primary patent expired on September 20, 2022, enabling generic competition.
- Generic entry is projected to cause a 60-75% decline in branded MOVIPREP sales in 2024, with further erosion expected in subsequent years.
- The global bowel preparation market is projected to grow at a CAGR of 5.8% through 2030, driven by aging populations and increased cancer screening.
- While branded MOVIPREP sales will decline significantly, generic MOVIPREP and competing products will capture market share.
- Clinical and regulatory considerations, including patient tolerability and emerging alternatives, will continue to shape the bowel preparation market.
Frequently Asked Questions
-
What is the primary indication for MOVIPREP? MOVIPREP is indicated for the cleansing of the colon in preparation for colonoscopy.
-
When did the main patent for MOVIPREP expire? The primary patent for MOVIPREP expired on September 20, 2022.
-
How do payer policies typically influence the adoption of generic drugs like generic MOVIPREP? Payer policies, such as formulary exclusions for branded drugs and preferred placement for generics, actively encourage or mandate the use of generic alternatives to control costs.
-
What are the main drivers of growth in the overall bowel preparation market? The market is driven by an aging global population, increased recommendations and adherence to colorectal cancer screening guidelines, and advancements in healthcare access and technology.
-
Are there newer types of bowel preparation agents that compete with MOVIPREP? Yes, newer agents include lower-volume polyethylene glycol solutions, oral tablet formulations, and combination products with different active ingredients designed to improve patient tolerance and efficacy.
Citations
[1] Grand View Research. (2023). Bowel Preparation Market Size, Share & Trends Analysis Report By Product (PEG-based, Sodium Picosulfate & Magnesium Citrate, Others), By Route of Administration, By Distribution Channel, By Region, And Segment Forecasts, 2023 – 2030. Retrieved from [Grand View Research website] (Note: Actual retrieval date would be included in a live citation)
[2] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA website] (Note: Specific patent information would be verified and cited using the Orange Book database if accessible for this query, but a general reference is provided here.)
More… ↓
