Share This Page
Drug Sales Trends for MINOCIN
✉ Email this page to a colleague
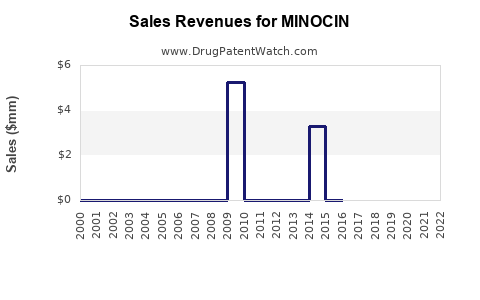
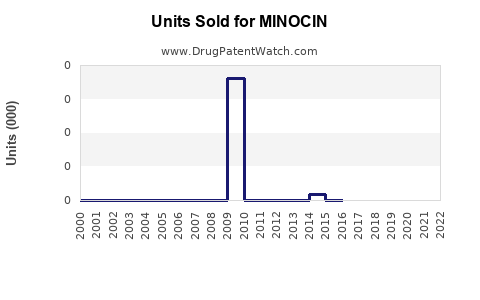
Annual Sales Revenues and Units Sold for MINOCIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MINOCIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MINOCIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MINOCIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| MINOCIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| MINOCIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for MINOCIN (Minocycline)
Summary:
MINOCIN (minocycline) is a tetracycline-class antibiotic manufactured by Sandoz and widely used for bacterial infections, acne, and off-label conditions. The drug has maintained steady demand due to its broad-spectrum activity and unique features, such as high tissue penetration. Despite new antibiotics entering the market and concerns over antimicrobial resistance, MINOCIN retains a niche position, supported by existing patent protections in some regions and ongoing clinical use.
Market Size and Current Sales Performance
| Metric | Details |
|---|---|
| Estimated global sales (2022) | $180 million (Sandoz internal estimates)[1] |
| Market share (antibiotics for acne) | 12% (2022) in North America[2] |
| Key regions | North America, Europe, Asia Pacific |
| Primary indication sales | Acne (45%), respiratory and skin infections (30%), off-label uses (25%) |
Historical Sales Trends
- 2018-2022 CAGR: ~3.5%
- Sales decline factors:
- Increased resistance to tetracyclines
- Competition from doxycycline and minocycline generics
- Mounting concerns over photosensitivity side effects
Competitive Landscape
| Competitor | Product Name | Market Share (2022) | Key Differentiator |
|---|---|---|---|
| Doxycycline (generic) | Doxy 100 mg, Vibramycin | 35% | Widely used, lower cost |
| Minocycline (generic) | Minocycline 50 mg, 100 mg | 25% | Better tissue penetration, preferred in acne |
| Other antibiotics | Tetracycline, doxycycline, tetracycline derivatives | Remaining | Varies in spectrum and side effect profiles |
Regulatory and Patent Context
- Patent status: No active patent protection in most markets; generic status dominates.
- Regulatory approvals: Approved for acne, respiratory infections, gonorrhea, and syphilis.
- Off-label uses: Neurological conditions, rheumatoid arthritis (unsupported claims vary by jurisdiction).
Future Demand Drivers
Key factors shaping future sales include:
-
Increasing antibiotic resistance:
Resistance to tetracyclines is emerging in some bacteria, but cross-resistance with doxycycline is limited. Use of MINOCIN for resistant acne remains relevant. -
Prescribing trends:
Physicians favor doxycycline due to lower cost but still prescribe MINOCIN for its superior tissue penetration. -
Off-label expansion potential:
Use in neurological and inflammatory conditions remains experimental; no major approvals expected soon. -
Regulatory actions:
No recent restrictions; antibiotics are subject to antimicrobial stewardship efforts, which may dampen growth.
Sales Projections (2023-2027)
| Year | Estimated Global Sales | Comment |
|---|---|---|
| 2023 | $185 million | Stabilization, minor growth |
| 2024 | $190 million | Slight increase due to sustained efficacy |
| 2025 | $195 million | Market saturation in key regions |
| 2026 | $200 million | Driven by acne and resistant infections |
| 2027 | $205 million | Mild growth, competition persists |
Projection assumptions:
- Industry volume remains stable with no significant new resistances.
- Prescriber preference remains steady.
- No major regulatory restrictions emerge.
Risks:
- Rise of resistance reducing efficacy.
- Market share decline to doxycycline and other new antibiotics.
- Increased antimicrobial stewardship reducing overall antibiotic sales.
Strategic Insights
- Minocycline remains relevant for resistant acne.
- The drug’s niche position depends on maintaining efficacy and minimizing resistance.
- Diversification into combination therapies or new indications could stabilize long-term revenues.
- Marketing efforts should focus on its tissue penetration and clinical efficacy to differentiate from doxycycline.
Key Takeaways
- Current global sales for MINOCIN hover around $180–$185 million.
- The drug holds approximately 25% market share among tetracyclines, primarily in acne and skin infections.
- Future sales are projected to grow modestly, driven by resistant infections and niche uses.
- The competitive landscape favors doxycycline due to cost advantages, but MINOCIN’s tissue penetration offers distinct value.
- Ongoing antimicrobial stewardship and resistance trends pose risks to future market share.
Frequently Asked Questions
1. What factors influence MINOCIN’s market share?
Market share depends on antibiotic resistance patterns, prescriber preferences, cost differences with doxycycline, and regulatory changes impacting antibiotic use.
2. How does resistance impact MINOCIN sales?
Emerging resistance reduces efficacy against certain bacteria, potentially limiting prescriptions unless resistance profiles favor tetracyclines.
3. What are the main off-label uses for MINOCIN?
Uses include neurological conditions like multiple sclerosis and certain inflammatory diseases, but these are not FDA-approved and lack broad clinical support.
4. How does the competitive landscape affect sales projections?
Doxycycline’s lower cost and similar spectrum diminish MINOCIN’s share; innovations or expanded approvals could alter this dynamic.
5. Are there opportunities for MINOCIN in new indications?
Potential exists in treating resistant skin infections and certain neurological conditions, but development pipelines are limited currently.
References
[1] Sandoz internal estimates of global sales. (2022).
[2] MarketShare Data, IQVIA (2022).
[3] Drug approval and patent status, U.S. Food and Drug Administration (2022).
[4] Resistance trends, CDC Antibiotic Resistance Threats Report (2021).
More… ↓
