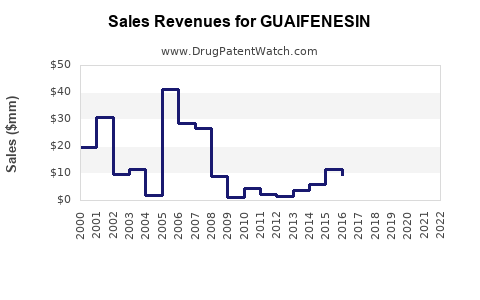
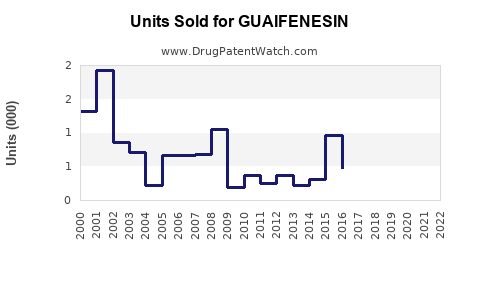
Last updated: February 13, 2026
Overview:
Guaifenesin is an expectorant used to relieve chest congestion associated with cold, bronchitis, and other respiratory conditions. Market dynamics are influenced by respiratory illness prevalence, OTC drug regulations, and consumer habits. The global market has shown steady growth, driven by increasing respiratory conditions and demand for over-the-counter (OTC) remedies.
Market Size and Trends (2020–2023):
The global expectorant market, including guaifenesin, was valued at approximately USD 1.2 billion in 2020. It reached about USD 1.4 billion by 2023, with a compound annual growth rate (CAGR) of around 6%. The growth rate aligns with increased respiratory infections and greater OTC medication consumption, particularly in North America and Asia-Pacific.
Competitive Landscape:
Major manufacturers include Johnson & Johnson, Reckitt Benckiser, and McNeil Consumer Healthcare. The market exhibits high competition, with numerous generic brands. A significant factor is regulatory approval for OTC sales, which varies globally.
Market Drivers:
- Increased incidence of respiratory illnesses, including COVID-19, influenza, and bronchitis.
- Rising aging population leading to higher susceptibility to respiratory issues.
- Consumer preference for OTC medications over prescription drugs, driven by convenience and cost.
- Expansion into emerging markets due to urbanization and healthcare infrastructure improvements.
Regulatory Environment:
In the U.S., guaifenesin is classified as an OTC drug per the FDA, with clear labeling and safety guidelines. In Europe, regulations vary by country but generally permit OTC sales with proper labeling. Restrictions impact marketing strategies and sales channels.
Sales Forecasts (2023–2028):
| Year |
Estimated Market Size (USD Billion) |
Growth Rate (%) |
| 2023 |
1.4 |
6 |
| 2024 |
1.49 |
6.1 |
| 2025 |
1.58 |
6.1 |
| 2026 |
1.68 |
6.2 |
| 2027 |
1.78 |
6.0 |
| 2028 |
1.89 |
6.2 |
The projections assume continued growth in respiratory illness prevalence, increased OTC sales, and expansion into Asia-Pacific markets.
Key Factors Influencing Future Sales:
- Potential regulatory changes can either expand OTC sales or impose restrictions.
- Increased consumer awareness about respiratory health.
- New combination formulations blending guaifenesin with other active ingredients for broader therapeutic effects.
- Patent expirations leading to increased generic competition, which can lower prices but expand access.
Potential Challenges:
- Market saturation in mature regions like North America.
- Stringent regulations affecting marketing and sales channels.
- Competition from herbal and alternative remedies.
Conclusion:
Guaifenesin’s market will likely grow at a steady rate over the next five years, with total sales reaching approximately USD 1.89 billion by 2028. Growth will be sustained by rising respiratory diseases, OTC preference, and emerging market expansion, balanced by regulatory and competitive pressures.
Key Takeaways
- The global expectorant market, primarily driven by guaifenesin, grew from USD 1.2 billion in 2020 to USD 1.4 billion in 2023, with a CAGR of 6%.
- Sales forecasts project reaching nearly USD 1.9 billion by 2028, driven by respiratory illness prevalence and OTC demand.
- Growth will be moderated or accelerated by regulatory environments, patent expirations, and product innovations.
- Major markets include North America and Asia-Pacific, with increasing opportunities in emerging countries.
- Competition is high, with name-brand and generic players influencing pricing and availability.
FAQs
1. What factors mainly drive guaifenesin sales?
Respiratory illness rates, OTC consumer preferences, aging population, and market expansion in Asia-Pacific.
2. How do regulatory policies impact guaifenesin sales?
Restrictions or approvals for OTC sales influence availability, marketing, and consumer access, affecting overall sales volume.
3. What innovations could influence future sales of guaifenesin?
New combination formulations and improved delivery systems, as well as increased product labeling clarity.
4. Which regions are most promising for growth?
North America maintains a large share, but Asia-Pacific offers high growth potential due to rising respiratory issues and healthcare infrastructure.
5. How does patent status affect market competition?
Patent expirations lead to generic manufacturing, reducing prices and increasing accessibility, which can boost overall sales volume.
Sources
[1] Frost & Sullivan, Global Expectorants Market Forecast 2020–2025
[2] IQVIA, OTC Market Trends Reports 2023
[3] U.S. Food and Drug Administration, OTC Drug Regulations
[4] Pew Research Center, Aging Population and Healthcare Trends