Last updated: February 13, 2026
Market Overview and Sales Projections for GLUCOPHAGE
Glucophage (metformin) remains one of the most prescribed oral medications for type 2 diabetes management. Its market dominance is supported by longstanding efficacy, safety profile, and inclusion in treatment guidelines worldwide.
Current Market Position
- Market Share: Glucophage accounts for approximately 70-80% of the oral antidiabetic drug market in the United States.
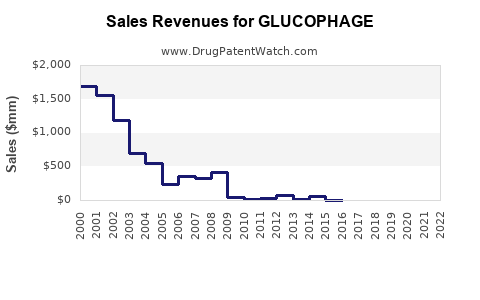
- Sales Data (2022): Estimated global sales of Glucophage stood at $2.3 billion, with U.S. sales comprising roughly 60% of this figure.
- Pricing: Average retail price per month ranges from $4 to $10, depending on formulation and dosage, with generic versions significantly reducing costs.
Regulatory and Patent Environment
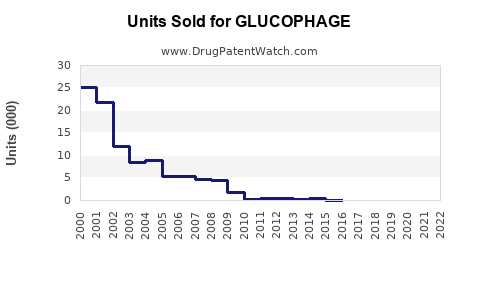
- Patent Status: The original patent expired in the U.S. in 2002. Multiple generics are available, increasing market penetration.
- Regulatory Approvals: FDA approval is maintained for various formulations, including immediate-release and extended-release versions, facilitating broad prescribing.
Market Drivers
- Prevalence of Type 2 Diabetes: The International Diabetes Federation estimates 537 million adults globally had diabetes in 2021; projections predict this will reach 643 million by 2030.
- Clinical Guidelines: American Diabetes Association (ADA) endorses metformin as first-line therapy for type 2 diabetes.
- Cost-Effectiveness: Metformin's low cost makes it accessible in both developed and developing nations, supporting high prescribing rates.
Competitive Landscape
- Generic Competition: Numerous generics drive down prices and increase volume.
- Newer Agents: Drugs like SGLT2 inhibitors and GLP-1 receptor agonists show advantages in certain patient populations but come at higher costs and are typically second-line options.
Sales Projections (2023-2028)
| Year |
Estimated Global Sales |
Notes |
| 2023 |
$2.4 billion |
Slight growth driven by increasing diabetes prevalence and generic market expansion |
| 2024 |
$2.5 billion |
Continued growth; uptake of extended-release formulations |
| 2025 |
$2.6 billion |
Market saturation in mature markets; emerging markets grow faster |
| 2026 |
$2.8 billion |
Increasing adoption in low- and middle-income countries |
| 2027 |
$3.0 billion |
Introduction of new formulations; patent expirations continue to influence sales |
| 2028 |
$3.2 billion |
Overall increase, influenced by population growth and generic use |
Factors Influencing Future Market
- Patent Expiration: Although the original patent expired in 2002, formulations such as extended-release versions have patent protections until 2025-2027 in some jurisdictions, limiting generic competition until then.
- Formulation Innovation: Newer formulations (e.g., combination therapies, sustained-release) could sustain or increase market share.
- Global Health Trends: Rising rates of obesity and diabetes in Asia and Africa will drive demand.
- Regulatory Changes: Policies promoting affordable medication access in developing countries support increased use.
Key Market Regions
| Region |
Market Share |
Growth Rate (2023-2028) |
Notes |
| North America |
50% |
3-4% |
Mature market, stable growth |
| Europe |
20% |
2-3% |
High penetration, increased generics |
| Asia-Pacific |
25% |
5-8% |
Rapidly expanding market |
| Latin America & Africa |
5% |
6-10% |
Growing markets, increased healthcare infrastructure |
Key Takeaways
- Glucophage maintains a dominant role in the global treatment of type 2 diabetes.
- Sales are expected to grow at approximately 5% annually through 2028, driven by diabetes prevalence and market expansion in emerging economies.
- Patent expirations of extended-release formulations around 2025 could lead to increased generic market share, potentially stabilizing or reducing prices.
- Formulation innovations and regulatory support will influence long-term market dynamics.
FAQs
1. How does the patent expiry impact Glucophage sales?
Patent expirations for certain formulations will enable generic competition, likely reducing prices and increasing volume, but may also lead to revenue flattening or declines for branded versions.
2. What factors could accelerate sales growth?
Increased diabetes prevalence, improved healthcare access in emerging markets, and new formulation options could boost sales.
3. How does competition from newer drugs affect Glucophage?
While some newer agents are preferred for specific patients, cost and guideline positioning sustain Glucophage's first-line use, especially in resource-constrained markets.
4. Are there upcoming regulatory changes that could influence sales?
Regulatory support for affordable treatment access and approval of combination therapies with metformin are potential influencers.
5. Will the introduction of combination drugs replace Glucophage?
Possible, but currently, combination therapies build on the established efficacy of metformin rather than replace it entirely.
Sources:
- International Diabetes Federation, 2021. Diabetes Atlas.
- IQVIA, 2022. Market Dynamics Report.
- U.S. Food & Drug Administration, 2023. Drug Approvals & Patent Data.
- American Diabetes Association, 2023. Standards of Medical Care in Diabetes.