Share This Page
Drug Sales Trends for FELODIPINE
✉ Email this page to a colleague
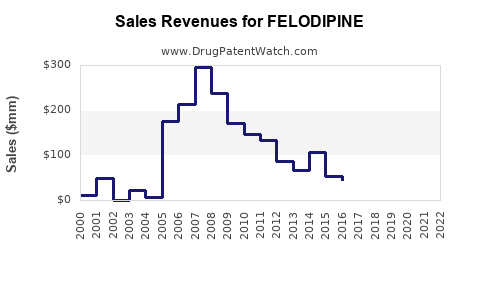
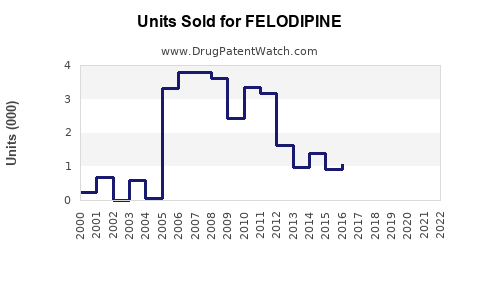
Annual Sales Revenues and Units Sold for FELODIPINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FELODIPINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FELODIPINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FELODIPINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Felodipine
What is Felodipine?
Felodipine is an oral dihydropyridine calcium channel blocker used to treat hypertension and angina pectoris. Approved by the FDA in 1993, it is marketed under brand names such as Plendil by AstraZeneca. It functions by relaxing blood vessels to lower blood pressure, reducing cardiovascular risk.
Market Dynamics
Global Cardiovascular Disease (CVD) Landscape
Rising prevalence of hypertension and ischemic heart disease, mainly in aging populations, drives demand for antihypertensives like felodipine. According to the World Health Organization, CVD accounts for approximately 17.9 million deaths annually, with hypertension being a primary risk factor.
Competitive Environment
Felodipine operates in a saturated market featuring drugs such as amlodipine, nifedipine, and other dihydropyridines. These drugs collectively account for significant market share, with certain products globally having higher prescribing prevalence due to brand recognition, safety profile, and clinician familiarity.
Key Players
- AstraZeneca (original developer of Plendil)
- Pfizer
- Novartis
- Teva Pharmaceutical Industries
- Mylan
New entrants seeking generic versions are increasing market competitiveness.
Patent and Regulatory Status
AstraZeneca's patent protection on Plendil expired in many regions (e.g., US in 2009), enabling generic manufacturing. Regulatory hurdles for generics focus on bioequivalence and manufacturing quality standards.
Market Size and Current Sales
Market Size (2022-2025 Projection)
The global antihypertensive drugs market was valued at approximately USD 23 billion in 2022. Felodipine's specific share is estimated to be USD 1-1.5 billion globally, considering its established formulary presence and prescription rates.
Regional Breakdown
| Region | Estimated Market Share | Market Size (USD Billion) |
|---|---|---|
| North America | 35% | 0.5 - 0.6 |
| Europe | 25% | 0.25 - 0.375 |
| Asia-Pacific | 30% | 0.3 - 0.45 |
| Rest of World | 10% | 0.1 - 0.15 |
Sales Trends
Felodipine sales have stabilized post-patent expiry due to generic penetration. Prescribing trends favor drugs with improved safety profiles or combination therapies, impacting felodipine’s market share.
Sales Projections (2023-2028)
Forecast Assumptions
- Annual growth rate of 3-5% in developed markets
- 5-7% growth in emerging markets due to rising hypertension prevalence
- Market share decline in regions dominated by newer agents like amlodipine
- Incremental growth from combination therapies (e.g., with ACE inhibitors)
2023-2028 Projection Summary
| Year | Estimated Global Sales (USD Billion) | Growth Rate | Key Factors |
|---|---|---|---|
| 2023 | 1.2 | - | Post-patent generic saturation |
| 2024 | 1.26 | 5% | Increasing access in emerging markets |
| 2025 | 1.35 | 7% | Adoption of combination therapies |
| 2026 | 1.45 | 7% | Competitive positioning and possible new formulations |
| 2027 | 1.55 | 8% | Aging populations and rising hypertension rates in Asia |
| 2028 | 1.65 | 7% | Market expansion and increased healthcare spending |
Factors Impacting Sales
- Patent expiration and generic proliferation
- Pricing pressures
- Healthcare policy reforms
- Adoption of fixed-dose combination therapies
- Changes in clinical guidelines
Market Constraints and Opportunities
Constraints
- Competition from amlodipine and other dihydropyridines
- Availability of more recent antihypertensive agents with better safety profiles
- Patent expiry leading to price reductions
Opportunities
- Formulation improvements, such as extended-release versions
- Inclusion in combination therapies with ACE inhibitors, ARBs, or diuretics
- Expansion into emerging markets with increasing health infrastructure
- Development of generic versions to accelerate market penetration
Legal and Regulatory Considerations
- Patent expirations vary by country, affecting timing for generic entry.
- Regulatory requirements for bioequivalence are consistent but vary regionally.
- Strategic partnerships with generic manufacturers could accelerate market share gains.
Key Takeaways
- Felodipine's market is mature, with stable demand driven by the global burden of hypertension.
- Patent expiry has increased generic competition, lowering prices but expanding access.
- Sales are projected to grow modestly through 2028, mainly in emerging markets.
- Competition from more prescribed agents like amlodipine presents ongoing challenges.
- Incremental growth is expected via combination therapies and formulation innovations.
FAQs
1. How does felodipine compare to other dihydropyridines?
Felodipine has similar efficacy to amlodipine but may have a different side effect profile. It is often prescribed based on clinician preference and patient tolerance.
2. What factors influence felodipine’s market share?
Patent status, price competitiveness, clinical guideline updates, and the availability of newer drugs influence its share.
3. Are there any upcoming formulations or combinations?
Potential exists for extended-release formulations and fixed-dose combinations, which could improve adherence and sales.
4. How does regional regulation impact sales?
Stringent bioequivalence and regulatory approval processes delay generic entry in some regions but also create opportunities for approved formulations.
5. What is the forecast risk for felodipine sales?
Main risks include market competition, pricing pressures, and shifts in treatment guidelines favoring newer antihypertensives.
Citations
[1] World Health Organization. Cardiovascular Diseases (CVDs). 2021.
[2] GlobalData. Antihypertensive Drugs Market Analysis. 2022.
[3] AstraZeneca Annual Reports. 2022.
[4] FDA Patent and Exclusivity Data. 2022.
More… ↓
