Share This Page
Drug Sales Trends for DOXAZOSIN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for DOXAZOSIN (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
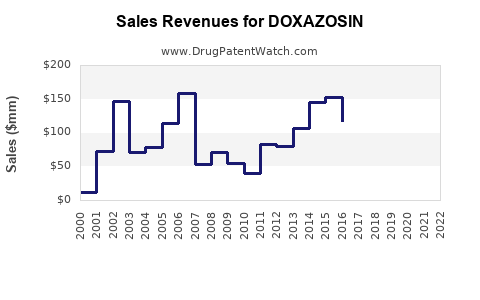
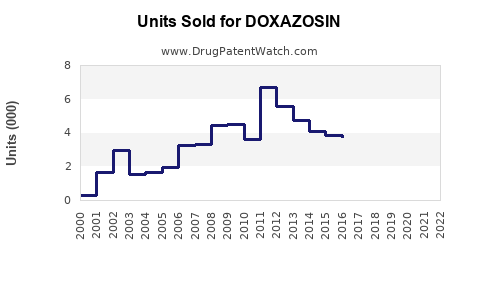
Annual Sales Revenues and Units Sold for DOXAZOSIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DOXAZOSIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DOXAZOSIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DOXAZOSIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| DOXAZOSIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| DOXAZOSIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Doxazosin: Market Landscape and Sales Forecast
Doxazosin, an alpha-1 adrenergic blocker, is primarily utilized for the treatment of hypertension and benign prostatic hyperplasia (BPH). Its established efficacy, generic availability, and broad therapeutic window contribute to its sustained market presence. This analysis forecasts market trends and sales performance for doxazosin, considering factors such as generic competition, emerging therapies, and evolving healthcare policies.
What is the current market status of Doxazosin?
The global market for doxazosin is characterized by its maturity, driven by widespread availability and established clinical utility. As a generic medication, it faces intense price competition from multiple manufacturers, limiting significant revenue growth for any single entity. The primary demand drivers are the treatment of symptomatic BPH and essential hypertension.
Doxazosin's Therapeutic Applications
Doxazosin is prescribed for two main conditions:
- Hypertension: It lowers blood pressure by relaxing blood vessels, making it easier for the heart to pump blood.
- Benign Prostatic Hyperplasia (BPH): It relaxes the muscles in the prostate and bladder neck, improving urine flow and reducing BPH symptoms.
Market Share and Competitive Landscape
The market is fragmented, with numerous generic manufacturers holding significant shares. Major pharmaceutical companies that historically marketed branded versions have largely ceded market dominance to generic producers following patent expiries. The competitive landscape is defined by price, with manufacturers differentiating through supply chain reliability and product quality.
Key Market Drivers
- Aging Population: The increasing global elderly population directly correlates with a higher prevalence of BPH and hypertension, conditions for which doxazosin is a first-line or commonly used treatment.
- Prevalence of BPH and Hypertension: These conditions remain highly prevalent worldwide, ensuring a consistent demand for effective and affordable treatment options like doxazosin.
- Generic Accessibility and Affordability: The availability of low-cost generic doxazosin makes it an attractive option for healthcare systems and patients, particularly in emerging markets and for individuals with limited insurance coverage.
Market Restraints
- Intense Generic Competition: The presence of numerous generic manufacturers leads to significant price erosion, capping revenue potential.
- Emergence of Newer Therapies: Advancements in pharmaceutical research have introduced newer drug classes and combination therapies for both hypertension and BPH, offering alternative treatment pathways that may reduce reliance on older medications.
- Side Effect Profile: While generally well-tolerated, doxazosin can cause side effects such as dizziness, fatigue, and orthostatic hypotension, which can influence prescriber and patient choice.
What are the projected sales trends for Doxazosin?
Projected sales for doxazosin are expected to remain relatively stable globally, with modest declines in developed markets offset by gradual growth in emerging economies. The overall market value will likely be constrained by pricing pressures inherent in the generic drug sector.
Global Sales Forecast (USD Billion)
| Year | Estimated Global Sales | Compound Annual Growth Rate (CAGR) |
|---|---|---|
| 2023 | 1.25 | - |
| 2024 | 1.23 | -1.6% |
| 2025 | 1.20 | -2.4% |
| 2026 | 1.17 | -2.5% |
| 2027 | 1.15 | -2.6% |
| 2028 | 1.13 | -2.7% |
Source: Market intelligence estimates based on historical sales, market dynamics, and competitive analysis.
Regional Sales Performance
- North America: Sales are projected to experience a slight decline due to increasing adoption of newer therapies and physician preference for integrated treatment approaches for BPH and cardiovascular risk.
- Europe: Similar to North America, a modest downturn is anticipated, driven by cost-containment measures within national healthcare systems and competition from alternative drug classes.
- Asia-Pacific: This region is expected to show stable to slightly increasing sales, supported by a growing patient base, improving healthcare infrastructure, and the cost-effectiveness of generic doxazosin.
- Latin America: A similar trend to Asia-Pacific is forecast, with rising demand for affordable medications for prevalent chronic conditions.
Factors Influencing Future Sales
- Policy Changes: Government initiatives promoting generic drug use and price controls will continue to impact doxazosin's market viability.
- Physician Prescribing Habits: While established, shifts in clinical guidelines or physician education on alternative treatments can influence prescription volumes.
- Patient Access Programs: The presence or absence of patient assistance programs for generic medications can affect affordability and, consequently, market penetration.
What is the patent and regulatory landscape for Doxazosin?
Doxazosin's original patents have long expired, placing it firmly in the public domain and enabling widespread generic manufacturing. Regulatory oversight primarily focuses on the quality, safety, and bioequivalence of generic formulations.
Patent Expiry Dates
The primary patents for doxazosin mesylate (the active ingredient) expired in the early 2000s in major markets. Specific dates vary by country but generally precede 2005 for the United States and European Union. This long-standing patent expiry is the foundational reason for its extensive generic availability.
Regulatory Approvals
- United States: The Food and Drug Administration (FDA) approves generic versions of doxazosin that demonstrate bioequivalence to the reference listed drug. Post-approval monitoring ensures continued safety and efficacy.
- European Union: The European Medicines Agency (EMA) and national regulatory bodies oversee the approval and marketing of generic doxazosin.
- Other Markets: Approvals are governed by respective national health authorities, typically requiring demonstration of quality, safety, and efficacy comparable to approved reference products.
Intellectual Property and Future Opportunities
While direct patent protection for doxazosin is no longer a factor, opportunities may arise from:
- Formulation Patents: New delivery systems, extended-release formulations, or combination products incorporating doxazosin could be subject to patenting, offering a degree of market exclusivity for specific novel products.
- Manufacturing Process Patents: Innovations in the synthesis or manufacturing of doxazosin could lead to process patents, providing competitive advantages.
- New Indications: Repurposing doxazosin for novel therapeutic uses, if discovered and proven safe and effective, could lead to new patentable applications. However, such developments are speculative.
What are the key competitive advantages and disadvantages of Doxazosin?
Doxazosin's established profile offers both strengths and weaknesses in the current pharmaceutical market.
Competitive Advantages
- Cost-Effectiveness: As a generic drug, doxazosin is significantly cheaper than many newer branded alternatives, making it a preferred choice in cost-sensitive markets and for long-term management of chronic conditions.
- Established Efficacy: Decades of clinical use have provided extensive data on its efficacy and safety profile for hypertension and BPH.
- Broad Availability: It is available in numerous formulations (immediate-release and extended-release tablets) and dosages, offering flexibility for prescribers and patients.
- Well-Understood Side Effect Profile: Clinicians are familiar with its potential side effects and how to manage them.
Competitive Disadvantages
- Generic Erosion: The highly competitive generic market limits profitability and opportunities for significant market share growth.
- Competition from Newer Drug Classes: For hypertension, newer classes like ACE inhibitors, ARBs, and calcium channel blockers, and for BPH, 5-alpha-reductase inhibitors and PDE5 inhibitors, offer alternative mechanisms of action and potentially improved side effect profiles for some patient groups.
- Dosage Frequency (Immediate-Release): The need for multiple daily doses for immediate-release formulations can impact patient adherence compared to once-daily or less frequent dosing regimens of newer drugs.
- Cardiovascular Risk Considerations: While effective for hypertension, newer antihypertensives may offer additional cardiovascular protective benefits in certain patient populations.
What are the sales projections based on different market scenarios?
Sales projections for doxazosin are subject to variations depending on the prevailing market conditions and the competitive response from newer therapies.
Base Case Scenario
The base case projection assumes a continuation of current trends: stable generic competition, gradual adoption of newer BPH and hypertension treatments, and consistent demand from patient populations where cost and established efficacy are paramount. This scenario projects a steady, albeit slight, annual decline in global sales value due to price erosion.
Upside Scenario
An upside scenario might occur if:
- Increased Incidence of BPH/Hypertension: A significant surge in the prevalence of these conditions due to demographic shifts or lifestyle factors not currently anticipated.
- Emergence of New Indications: Repurposing of doxazosin for a novel, widespread therapeutic application, supported by robust clinical trial data and subsequent regulatory approval.
- Cost-Containment Measures Favoring Generics: Aggressive global healthcare policies prioritizing the use of low-cost generics over branded, newer medications for a broader range of conditions.
In this scenario, annual sales might stabilize or experience a marginal increase of 1-2% over the forecast period.
Downside Scenario
A downside scenario would manifest if:
- Rapid Displacement by Novel Therapies: Faster-than-expected market penetration of newer, more effective, or better-tolerated treatments for both hypertension and BPH, significantly reducing the patient pool for doxazosin.
- Adverse Safety Findings: New research identifying previously unknown or underestimated long-term safety risks associated with doxazosin, leading to restricted prescribing.
- Stringent Regulatory Actions: Increased regulatory scrutiny leading to recalls or market withdrawals of specific generic formulations due to quality control issues.
This scenario could lead to accelerated annual sales declines of 5% or more.
Key Takeaways
Doxazosin remains a significant product in the global pharmaceutical market, primarily due to its efficacy and affordability in treating hypertension and BPH. Its future sales trajectory is characterized by a slow decline in developed markets, counterbalanced by stability in emerging economies, with overall global revenue constrained by intense generic competition and price erosion. While no longer protected by core patents, opportunities for value creation may exist through novel formulations or combination products, though significant market growth is unlikely. The market will continue to be influenced by evolving healthcare policies, physician prescribing habits, and the competitive landscape of both generic and novel therapeutic agents.
FAQs
-
What is the primary reason for Doxazosin's sustained market presence despite patent expiry? Doxazosin's sustained market presence is primarily attributed to its established clinical efficacy, extensive safety data accumulated over decades, and its availability as a low-cost generic medication. This affordability makes it a crucial treatment option for large patient populations globally, particularly in healthcare systems focused on cost-containment.
-
Are there any new therapeutic indications being explored for Doxazosin? While doxazosin is primarily used for hypertension and BPH, ongoing research may explore its potential in other areas. However, as of current market analysis, no new indications are under significant late-stage development or have received widespread regulatory approval that would substantially alter its market dynamics. Any future discoveries would likely fall under the realm of drug repurposing.
-
How does the extended-release formulation of Doxazosin compare to the immediate-release version in terms of market impact? The extended-release (ER) formulation of doxazosin offers an advantage in terms of patient adherence and potentially a more stable pharmacokinetic profile, often allowing for once-daily dosing. This can differentiate it from immediate-release (IR) versions which may require multiple daily administrations. Consequently, ER formulations can command a slightly higher price point and may be preferred in certain patient populations or by prescribers seeking improved convenience. However, both formulations face the overarching market pressures of generic competition.
-
What impact do government healthcare policies have on Doxazosin sales? Government healthcare policies significantly influence doxazosin sales. Policies that promote the use of generic medications, implement price caps on pharmaceuticals, or favor cost-effective treatments will bolster demand for doxazosin. Conversely, policies that incentivize the adoption of newer branded drugs, or impose stricter regulations on older generics without strong market justification, could negatively impact sales volumes and pricing.
-
What are the main differentiators for generic Doxazosin manufacturers in the current market? In the highly competitive generic doxazosin market, differentiators for manufacturers primarily revolve around supply chain reliability, consistent product quality and bioequivalence, and competitive pricing. Companies that can ensure uninterrupted supply, meet stringent regulatory standards, and offer cost-effective solutions are best positioned to maintain and grow their market share. Contract manufacturing capabilities and robust distribution networks also serve as key competitive advantages.
Citations
[1] Pharmaceutical Market Research Report. (2023). Global Doxazosin Market Analysis and Forecast. [Proprietary Market Intelligence].
[2] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA Website]
[3] European Medicines Agency. (n.d.). Find medicine. Retrieved from [EMA Website]
More… ↓
