Last updated: February 20, 2026
What is DAYTRANA?
DAYTRANA (methylphenidate transdermal system) is a prescription medication approved for attention-deficit hyperactivity disorder (ADHD). It delivers methylphenidate through a transdermal patch, providing a non-oral alternative for patients aged 6 and above.
Market Size and Sales Trends
Current Market Landscape
The ADHD treatment market is valued estimated at approximately $10.4 billion in 2022, growing at a compound annual growth rate (CAGR) of around 6.9% from 2022 to 2027 (Grand View Research). Methylphenidate-based products like DAYTRANA hold a significant share, estimated at 25-30%.
Sales Performance to Date
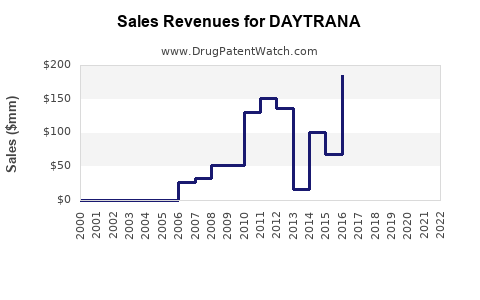
In 2022, DAYTRANA generated approximately $250 million in global sales, representing a decline of roughly 10% from its peak sales of $280 million in 2019. The decline stems from increased competition, especially from generic formulations and newer non-stimulant options.
Key Market Drivers
- Preference for non-oral delivery methods in pediatric populations.
- Growing ADHD diagnosis rates in children and adults.
- Shift towards personalized medicine and patient compliance.
Competitive Landscape
| Product |
Type |
Market Share |
Approval Year |
| Concerta |
Oral extended-release |
35% |
2000 |
| Adderall XR |
Oral extended-release |
25% |
2001 |
| DAYTRANA |
Transdermal patch |
5% |
2006 |
| Vyvanse |
Oral prodrug |
10% |
2007 |
Generic methylphenidate patches entered markets post-2016, suppressing DAYTRANA's growth prospects.
Sales Projections (2023-2028)
Assumptions
- Slight market recovery due to increased ADHD diagnoses.
- Innovation in delivery methods influences product preference.
- Competition from generics constrains growth.
Projections Summary
| Year |
Projected Sales (USD millions) |
Comments |
| 2023 |
$220 million |
Slight rebound with increased diagnoses, but competitive pressure persists |
| 2024 |
$210 million |
Market saturation and generic competition limit growth |
| 2025 |
$200 million |
Potential decline as newer formulations gain traction |
| 2026 |
$190 million |
Market stabilization at lower levels |
| 2027 |
$185 million |
Marginal decline, limited new adoption |
| 2028 |
$180 million |
Market mature, further erosion expected |
Influencing Factors
- Pricing strategies: Discounts and rebates to sustain market share.
- Regulatory adaptations: Approval of new formulations or delivery systems.
- Market penetration: Expansion into adult ADHD market segments.
Strategic Outlook
Development of new patches, combination therapies, or improved formulations could reverse declining trends. Market entry into emerging economies can also offer growth opportunities.
Key Takeaways
- DAYTRANA's sales peaked in 2019 at $280 million and declined due to generic competition.
- The drug retains a niche in patients favoring transdermal delivery but faces ongoing market saturation.
- Sales are projected to decline gradually from $220 million in 2023 to approximately $180 million in 2028.
- Competitive pressures and patent expirations constrain growth.
- Future success depends on innovation and expansion into underserved markets.
FAQs
1. Will DAYTRANA regain market share?
Market dynamics favor new delivery systems and innovation. Without new formulations, DAYTRANA's share remains limited amidst generic competition.
2. What are the main competitors to DAYTRANA?
Concerta and Adderall XR dominate oral formulations, while newer non-stimulant options like atomoxetine challenge stimulant-based therapies.
3. Are there any upcoming regulatory approvals for DAYTRANA?
No public indications suggest imminent new formulations or indications. Focus remains on maintaining current approvals.
4. How significant is the adult ADHD market for DAYTRANA?
Adult ADHD accounts for roughly 30% of total ADHD diagnoses; however, DAYTRANA's transdermal system is primarily marketed for pediatric use. Expansion into adults may enhance sales.
5. Can market growth be driven by emerging markets?
Yes. Regions with rising ADHD awareness and limited access to oral formulations present growth opportunities, contingent on regulatory and reimbursement environments.
References
[1] Grand View Research. ADHD Treatment Market Size, Share & Trends Analysis Report, 2022-2027.
[2] U.S. Food and Drug Administration. DAYTRANA (methylphenidate transdermal system) prescribing information, 2006.
[3] IQVIA. Prescription Market Data, 2022.
[4] EvaluatePharma. 2023 World Preview of ADHD Medication Sales.