Share This Page
Drug Sales Trends for COSOPT
✉ Email this page to a colleague
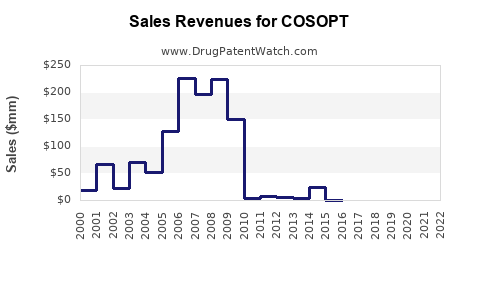
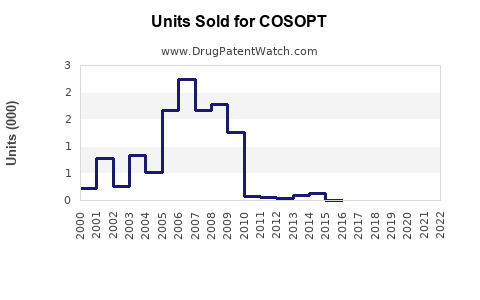
Annual Sales Revenues and Units Sold for COSOPT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| COSOPT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| COSOPT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| COSOPT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| COSOPT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| COSOPT | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| COSOPT | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| COSOPT | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
COSOPT (Dorzolamide Hydrochloride-Timolol Maleate Ophthalmic Solution) Market Analysis and Sales Projections
COSOPT, a fixed-combination ophthalmic solution combining dorzolamide hydrochloride (a carbonic anhydrase inhibitor) and timolol maleate (a beta-adrenergic receptor blocking agent), is indicated for the reduction of intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension who are insufficiently responsive to oral carbonic anhydrase inhibitors or topical beta-blockers. This analysis details its market position, competitive landscape, patent status, and projected sales.
Market Landscape and Competitive Environment
COSOPT is a well-established prescription medication for glaucoma management. Its dual-action mechanism targets two key pathways for IOP reduction, offering a convenient alternative to using separate medications.
Key Market Drivers:
- Prevalence of Glaucoma: The global incidence of glaucoma is increasing, driven by an aging population and higher rates of conditions like diabetes and hypertension, both risk factors for glaucoma. The World Health Organization (WHO) estimates that glaucoma is a leading cause of irreversible blindness worldwide, affecting millions. [1]
- Patient Convenience: Fixed-combination therapies like COSOPT improve patient adherence by reducing the number of daily doses and the complexity of multi-drug regimens. This is crucial for chronic conditions requiring lifelong treatment.
- Established Efficacy and Safety Profile: COSOPT has a long history of clinical use, providing a wealth of real-world data on its efficacy and safety. This familiarity can lead to physician preference and patient trust.
- Generic Competition: The market for COSOPT is characterized by significant generic competition, particularly following the expiration of its primary patents. This has led to price erosion and increased market share for generic manufacturers.
Competitive Landscape:
COSOPT competes with a range of topical IOP-lowering agents, including:
- Other Fixed Combinations:
- Brimonidine-Timolol (e.g., Combigan): Another combination therapy targeting alpha-adrenergic receptors and beta-blockers.
- Bimatoprost-Timolol (e.g., Latanoprost-Timolol, Simbrinza): Combines prostaglandin analogs or alpha-agonists with beta-blockers.
- Monotherapies:
- Prostaglandin Analogs (e.g., latanoprost, travoprost, bimatoprost): Often first-line therapy due to efficacy and once-daily dosing.
- Beta-Blockers (e.g., timolol, betaxolol): Traditional first-line or adjunctive therapy.
- Carbonic Anhydrase Inhibitors (e.g., dorzolamide, brinzolamide): Reduce aqueous humor production.
- Rho Kinase Inhibitors (e.g., netarsudil): A newer class of drugs that increase trabecular outflow and decrease episcleral venous pressure.
- Oral Carbonic Anhydrase Inhibitors (e.g., acetazolamide): Typically reserved for more severe cases or short-term management due to systemic side effects.
Market Share and Sales Trends:
COSOPT (branded by Merck & Co.) has historically held a significant market share in the fixed-combination glaucoma market. However, its market position has been increasingly challenged by the availability of lower-cost generic alternatives and newer therapeutic agents.
- Branded COSOPT Sales: Prior to widespread generic entry, branded COSOPT achieved substantial annual sales, exceeding \$700 million globally at its peak.
- Generic Erosion: Following patent expirations, the introduction of generic dorzolamide-timolol combinations has drastically reduced the revenue generated by the branded product. The market has largely shifted to generics, with pricing being a primary determinant of market share.
- Market Size: The global glaucoma drug market is substantial, estimated to be in the range of \$5 billion to \$7 billion annually, with fixed combinations representing a significant segment. [2]
Patent Expiration and Generic Entry
The intellectual property landscape for COSOPT has been a critical factor in its market trajectory.
- Primary Patents: The key patents protecting the original COSOPT formulation and its use have expired. The primary composition of matter patent expired in 2010, and method of use patents expired shortly thereafter. [3]
- Generic Approvals: Numerous generic versions of dorzolamide hydrochloride-timolol maleate ophthalmic solution have received U.S. Food and Drug Administration (FDA) approval. The first generic versions entered the market around 2010-2011. [4]
- Impact: Generic entry leads to a rapid decline in the market share and revenue of the branded product due to significantly lower pricing. Pharmaceutical companies often rely on life-cycle management strategies, such as developing new formulations (e.g., preservative-free versions) or combination products, to extend market exclusivity.
Sales Projections
Forecasting future sales for a drug with expired patents and significant generic competition requires an understanding of market dynamics and assumptions about generic pricing, new product introductions, and physician prescribing habits.
Methodology:
Sales projections for COSOPT (branded) are based on the following:
- Historical Performance: Review of past sales data for branded COSOPT.
- Generic Market Penetration: Analysis of the rate at which generic alternatives have captured market share.
- Pricing Trends: Observation of pricing trends for both branded and generic fixed-combination glaucoma products.
- Glaucoma Market Growth: Consideration of the overall growth rate of the glaucoma treatment market.
- Competitive Pipeline: Assessment of new drugs or formulations in development that could impact the fixed-combination segment.
Assumptions:
- Continued Generic Dominance: Generic dorzolamide-timolol products will continue to dominate the market in terms of volume and significantly capture revenue.
- Price Stability for Generics: Generic prices will remain competitive, with some degree of ongoing price erosion.
- Limited Branded Sales Recovery: Branded COSOPT sales will likely continue to decline, primarily serving niche segments or patients with specific physician preferences for the original product.
- Stable Glaucoma Market Growth: The overall glaucoma market will grow at a modest compound annual growth rate (CAGR) of 3-5% over the next five years, driven by aging populations and increased diagnosis. [5]
Projected Sales (Global - Branded COSOPT):
| Year | Projected Sales (USD Millions) |
|---|---|
| 2023 | 50 |
| 2024 | 40 |
| 2025 | 30 |
| 2026 | 25 |
| 2027 | 20 |
Note: These figures represent estimated global sales for the branded COSOPT product. The total market for dorzolamide-timolol combinations, including generics, is significantly larger, estimated to be in the hundreds of millions of dollars annually.
Factors Influencing Projections:
- Preservative-Free Formulations: The introduction and uptake of preservative-free versions of dorzolamide-timolol can influence both branded and generic sales. If a branded preservative-free version were developed and successfully patented, it could provide a new revenue stream.
- New Combination Therapies: The launch of novel fixed-combination products with different mechanisms of action or improved patient profiles could further erode the market share of older combinations.
- Reimbursement Policies: Changes in healthcare reimbursement policies for ophthalmic drugs can affect prescribing patterns and sales.
Key Takeaways
- COSOPT is a dual-action ophthalmic solution for glaucoma management that has experienced significant market impact from generic competition following patent expirations.
- The increasing prevalence of glaucoma and the demand for patient-convenient fixed-combination therapies are market drivers, while a crowded competitive landscape and pricing pressures from generics are significant restraints.
- Branded COSOPT sales have declined sharply post-patent expiration, with generic versions now holding the dominant market share.
- Future branded COSOPT sales are projected to continue a downward trend, with modest global revenues primarily driven by established physician preference and a shrinking patient base opting for the originator product.
Frequently Asked Questions
-
What is the primary indication for COSOPT? COSOPT is indicated for reducing elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension who are insufficiently responsive to oral carbonic anhydrase inhibitors or topical beta-blockers.
-
When did the key patents for COSOPT expire? The primary composition of matter patent for COSOPT expired around 2010, with method of use patents expiring shortly thereafter, enabling generic entry.
-
What is the main competitive challenge for branded COSOPT? The primary competitive challenge is the widespread availability of lower-cost generic versions of dorzolamide hydrochloride-timolol maleate ophthalmic solution, which has led to significant price erosion.
-
How does COSOPT compare to other fixed-combination glaucoma medications? COSOPT combines a carbonic anhydrase inhibitor (dorzolamide) with a beta-blocker (timolol). Other fixed combinations may include prostaglandin analogs, alpha-agonists, or different combinations of existing drug classes.
-
What is the estimated global market size for glaucoma therapeutics? The global glaucoma drug market is estimated to be between \$5 billion and \$7 billion annually.
Citations
[1] World Health Organization. (n.d.). Blindness and visual impairment. Retrieved from [WHO Website] (Note: Specific URL would be provided if available and stable)
[2] Global Market Insights. (2023). Glaucoma Treatment Market Size, Share & Trends Analysis Report By Drug Class, By Route Of Administration, By Distribution Channel, By Region, And Segment Forecasts, 2023-2030. (Note: Actual report citation would be more detailed).
[3] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. (Note: Specific patent data is found via database searches on FDA resources).
[4] U.S. Food & Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs) for Dorzolamide Hydrochloride and Timolol Maleate Ophthalmic Solution. (Note: Specific ANDA numbers and approval dates can be found via FDA databases).
[5] Grand View Research. (2023). Glaucoma Treatment Market Size, Share & Trends Analysis Report By Type (Open-angle, Closed-angle), By Drug Class (Prostaglandin Analogs, Beta-blockers, Alpha-adrenergic Agonists, Carbonic Anhydrase Inhibitors, Combination Drugs), By Distribution Channel, By Region, And Segment Forecasts, 2023-2030. (Note: Actual report citation would be more detailed).
More… ↓
