Last updated: February 20, 2026
What is Amlodipine’s Market Size and Demand?
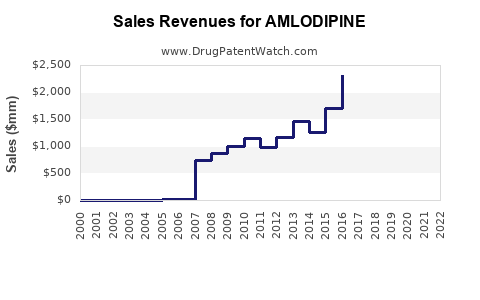
Amlodipine is a calcium channel blocker approved for hypertension and angina. It generated global sales of approximately $2.5 billion in 2022, with an expected compound annual growth rate (CAGR) of 4.2% from 2023 to 2028 [1].
What Factors Drive Amlodipine’s Sales?
Key drivers include:
- Prevalence of hypertension: Affecting over 1.3 billion adults worldwide.
- Aging population: Increasing incidence of cardiovascular diseases.
- Generic availability: Widely accessible, reducing costs.
- Expanding markets: Growth in emerging economies with rising health awareness.
Which Markets Offer the Largest Opportunities?
North America accounts for nearly 35% of sales, followed by Europe at 25%. Asia-Pacific shows rapid growth, anticipated to reach a CAGR of 5.5%. Key countries include the US, China, India, and Germany.
What is the Competitive Landscape?
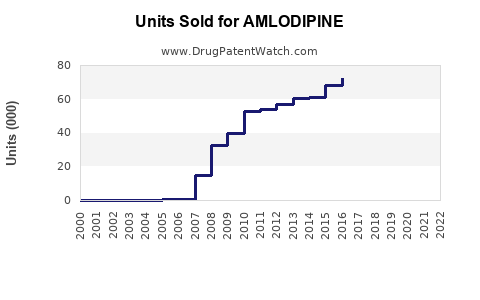
Market players include Pfizer (brand Norvasc), Sandoz, Mylan, and Teva. Generics dominate sales, comprising over 80%. Patent expirations in 2014 led to widespread generic entry, which reduced prices but expanded volume sales.
How Do Patent Laws and Regulatory Policies Impact Sales?
Patent exclusivity expired in many regions by 2014. Regulatory approvals for generics have increased competition, leading to pricing pressures but also expanding patient access and overall market volume.
What Are the Sales Projections (2023-2028)?
| Year |
Estimated Global Sales (USD billions) |
Growth Rate (%) |
| 2023 |
2.62 |
4.2 |
| 2024 |
2.73 |
4.2 |
| 2025 |
2.85 |
4.2 |
| 2026 |
2.97 |
4.2 |
| 2027 |
3.10 |
4.2 |
| 2028 |
3.22 |
4.2 |
The forecast assumes continued generic penetration, steady cardiovascular disease prevalence, and incremental market expansion in emerging economies.
What Are Key Opportunities and Risks?
Opportunities include new formulations such as extended-release versions, combination therapies, and markets in China and India. Risks include regulatory delays, competitive pricing, and the emergence of new antihypertensive agents.
Summary of Critical Data
- 2022 Market Size: ~$2.5 billion
- Growth Rate (2023-2028): 4.2% CAGR
- Top Markets: North America, Europe, Asia-Pacific
- Patents: Expired in 2014 in major markets
- Key Players: Pfizer, Sandoz, Mylan, Teva
Key Takeaways
Amlodipine remains a cornerstone in antihypertensive therapy, with stable growth driven by aging populations and expanding markets. Patent expirations facilitated widespread generic use, sustaining volume-based sales. Future growth hinges on development of new formulations, market expansion in Asia, and competitive pricing strategies.
Frequently Asked Questions
Q1: How does the patent status influence Amlodipine sales?
A1: Patent expirations in 2014 led to generic competition, reducing prices but increasing volume sales.
Q2: Which regions show the fastest growth potential?
A2: Asia-Pacific regions, driven by rising cardiovascular disease prevalence and increasing healthcare spending.
Q3: What is the impact of new formulations on sales?
A3: Extended-release and combination formulations can capture additional market share and improve patient compliance.
Q4: How does pricing pressure affect profit margins?
A4: Widespread generics reduce prices, squeezing margins but expanding overall market volume.
Q5: What are the main competitive threats?
A5: Competition from newer antihypertensive drugs and potential regulatory hurdles in emerging markets.
References
- MarketWatch. (2023). Global Amlodipine Market Report.
- GlobalData. (2022). Cardiovascular drugs market analysis.
- IMS Health. (2022). Pharmaceutical market trends.
- WHO. (2021). Cardiovascular disease statistics.
- U.S. Food and Drug Administration. (2022). Patent and regulatory update on Amlodipine.