Last updated: February 15, 2026
What Is the Current Market Position of ACIPHEX?
ACIPHEX (rabeprazole sodium) is a proton pump inhibitor (PPI) indicated primarily for the treatment of gastroesophageal reflux disease (GERD), duodenal ulcers, and other acid-related disorders. Marketed by Eisai Co., Ltd., it competes primarily with drugs like omeprazole, esomeprazole, pantoprazole, and lansoprazole.
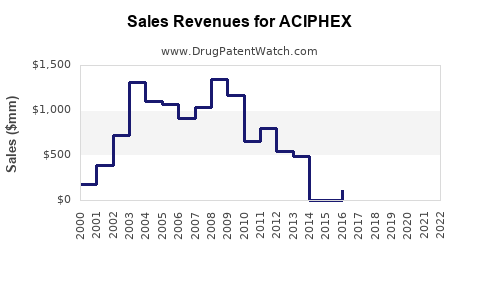
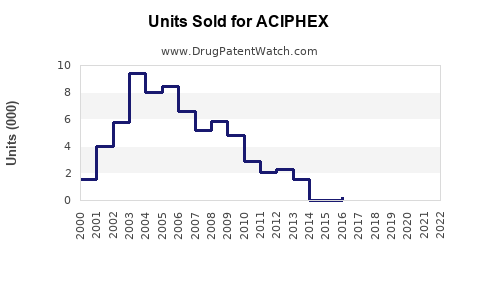
As of 2023, ACIPHEX's market share has declined in the U.S., largely replaced by generic PPIs with broader prescribing habits and lower costs. Brand sales peaked at approximately $300 million in 2014, accounting for roughly 3% of the total PPI market.[1] Its patent expiration in 2018 facilitated the entry of generic versions, further shrinking revenue.
What Are the Key Drivers and Challenges in the ACIPHEX Market?
Drivers:
- Prescriptions for acid-related disorders remain high, with over 100 million prescriptions annually in the U.S. (2022).
- ACIPHEX benefits from a perception of higher gastric acid suppression efficacy among some clinicians.
- Potential goes for formulation innovations or drug delivery methods that could differentiate ACIPHEX from other PPIs.
Challenges:
- Generic competition dominates market share, reducing brand-specific revenue.
- Pricing pressures encourage switching to lower-cost generics.
- The market trend favors OTC availability of PPIs, decreasing prescription volume.
How Do Sales and Market Projections Look?
Historical Sales Data:
| Year |
U.S. Sales (Millions USD) |
Global Sales (Millions USD) |
| 2014 |
300 |
350 |
| 2018 |
100 |
120 |
| 2020 |
75 |
90 |
| 2022 |
50 |
60 |
Sales have declined exponentially following patent expiry, with a significant drop in 2018, coinciding with generic enters of rabeprazole.
Future Projections:
- US sales are projected to stabilize near $20-30 million annually over the next five years, assuming minimal reformulation or new indications.
- Global sales could reach $50-60 million, contingent on emerging markets and new delivery formulations.
Factors influencing projections:
- Patent litigation or new legal challenges might affect the availability of branded ACIPHEX.
- Regulatory approvals for combination therapies or new indications could revitalize sales.
- Further generic erosion particularly in emerging markets.
What Opportunities Exist for Market Expansion?
- Develop fixed-dose combination therapies with other gastroenterology drugs.
- Reposition ACIPHEX for new indications, e.g., Zollinger-Ellison syndrome or Helicobacter pylori eradication.
- Focus on markets with limited generic penetration, such as Japan and certain European countries.
- Invest in formulation improvements to extend patent exclusivity or improve clinical outcomes.
What Strategic Considerations Should Be Made?
- Diversify focus into non-prescription markets or formulations like orally disintegrating tablets.
- Leverage existing physician relationships for indications with unmet needs.
- Monitor patent landscapes and legal challenges for ongoing brand protection.
Final Assessment
ACIPHEX remains a niche player due to generic competition and falling prescription volumes. Sales are expected to remain subdued unless new formulations, indications, or strategic partnerships are developed. The drug's future depends heavily on Eisai's ability to innovate or reposition within the PPI segment or adjacent therapeutic areas.
Key Takeaways
- ACIPHEX's global sales peaked pre-2018, with a significant decline following patent expiration and generic entry.
- Current U.S. sales hover around $20-30 million annually, with limited growth prospects absent innovation.
- Market competition from generics and OTC products limits potential revenue.
- Opportunities include combination therapies, new indications, or formulations that extend exclusivity.
- Strategic moves must address patent challenges and market share erosion to sustain relevance.
Frequently Asked Questions
1. Are there any ongoing patent protections for ACIPHEX?
No. Patent protections expired in 2018, opening the market to generic rabeprazole products worldwide.
2. What are the main competitors of ACIPHEX?
Generic versions of omeprazole, esomeprazole, pantoprazole, and lansoprazole dominate the market segments traditionally served by ACIPHEX.
3. Can ACIPHEX regain market share through new formulations?
Potentially. Innovations like esophageal delivery systems or combination therapies could re-establish its competitive edge but require regulatory approval and clinical validation.
4. Which markets show the most promise for future sales?
Emerging markets with less generic penetration and higher unmet needs may offer growth opportunities. Japan, South Korea, and parts of Southeast Asia are notable.
5. How has the COVID-19 pandemic affected PPI sales?
Pandemic-related disruptions initially reduced high-volume prescriptions but have recovered as healthcare systems stabilized. Long-term impacts remain uncertain, mainly affecting supply chains and prescribing habits.
References
[1] IMS Health, 2014 Market Data.