Share This Page
Drug Sales Trends for hydroxychloroquine
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for hydroxychloroquine (2021)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
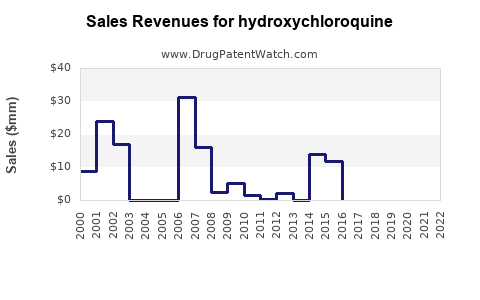
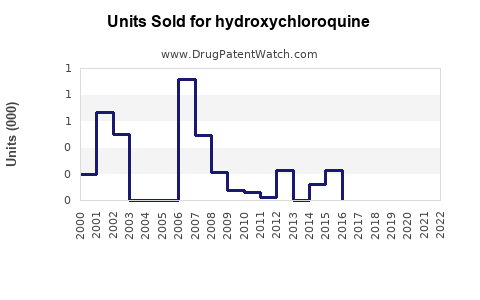
Annual Sales Revenues and Units Sold for hydroxychloroquine
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| HYDROXYCHLOROQUINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| HYDROXYCHLOROQUINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| HYDROXYCHLOROQUINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| HYDROXYCHLOROQUINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| HYDROXYCHLOROQUINE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Hydroxychloroquine: Market Trajectory and Sales Projections
Hydroxychloroquine (HCQ) is an antimalarial and antirheumatic drug with established therapeutic roles in treating malaria, systemic lupus erythematosus (SLE), and rheumatoid arthritis (RA). Its market presence is characterized by a long history of use, generic availability, and recent volatility linked to off-label investigations. This analysis projects HCQ's future market performance based on its established indications and explores potential impacts from ongoing research and regulatory considerations.
What is the Current Market Size and Segmentation for Hydroxychloroquine?
The global hydroxychloroquine market is estimated to have been valued at approximately USD 650 million in 2023. This market is segmented primarily by indication and distribution channel.
Market Segmentation by Indication:
- Rheumatoid Arthritis (RA): This segment represents the largest portion of the market, driven by the drug's efficacy in managing RA symptoms and slowing disease progression. The prevalence of RA, particularly in aging populations, supports sustained demand.
- Systemic Lupus Erythematosus (SLE): SLE treatment constitutes the second-largest segment. HCQ is a cornerstone therapy for managing cutaneous and systemic manifestations of lupus.
- Malaria: While historically a primary indication, the malaria segment has seen a decline in market share due to the development of newer antimalarial drugs and increasing drug resistance in certain regions. However, it remains a significant market in endemic areas.
- Other Indications: This includes off-label uses, such as certain dermatological conditions and its investigated, though not FDA-approved, role in treating COVID-19.
Market Segmentation by Distribution Channel:
- Hospital Pharmacies: Account for a significant share due to inpatient prescriptions and specialized treatment protocols.
- Retail Pharmacies: The largest segment, serving the broad outpatient market for chronic conditions like RA and SLE.
- Online Pharmacies: Experiencing steady growth as consumers increasingly opt for convenient home delivery of medications.
Key Market Drivers:
- Chronic Disease Prevalence: The rising incidence of autoimmune diseases like RA and SLE globally fuels consistent demand.
- Generic Availability: HCQ is available as a generic medication, making it a cost-effective treatment option, which is particularly relevant in healthcare systems facing budget constraints.
- Established Efficacy and Safety Profile: For its approved indications, HCQ has a well-documented history of efficacy and a generally manageable safety profile when used under medical supervision.
Key Market Restraints:
- Limited New Drug Development: The lack of significant new patent filings for HCQ indicates a mature product with limited scope for novel applications driving substantial market expansion through innovation.
- Off-Label Use Controversies: Investigations into its efficacy for conditions like COVID-19 led to significant market fluctuations and regulatory scrutiny, impacting physician confidence and prescribing patterns for off-label uses.
- Emergence of Biologics and Targeted Therapies: For RA and SLE, newer biologic and targeted synthetic disease-modifying antirheumatic drugs (DMARDs) offer alternative treatment options, potentially diverting market share from HCQ in some patient populations, especially those refractory to conventional therapies.
What are the Patent Expirations and Generic Competition Landscapes for Hydroxychloroquine?
Hydroxychloroquine is a well-established drug with its original compound patents long expired. The active pharmaceutical ingredient (API) is manufactured by numerous generic drug companies worldwide. This landscape is characterized by intense price competition among generic manufacturers.
Key Aspects of Patent and Competition Landscape:
- No Compound Patents: The fundamental patent protection for hydroxychloroquine expired decades ago. This means the molecule itself is in the public domain.
- Generic Dominance: The market is dominated by generic formulations. Major generic manufacturers in India, China, and established pharmaceutical markets in Europe and North America are key suppliers.
- Formulation Patents: While compound patents are expired, some companies may hold patents on specific extended-release formulations, novel delivery systems, or specific manufacturing processes. However, these are typically limited in scope and duration and have not historically created significant market exclusivity for HCQ.
- Intellectual Property (IP) Focus: Current IP activity related to HCQ is less about novel compound discovery and more focused on:
- Process Patents: Improvements in manufacturing efficiency or purity.
- Combination Therapies: Patents covering specific combinations of HCQ with other active pharmaceutical ingredients for synergistic effects in treating certain diseases, though this has been limited.
- Repurposing Studies: Patent applications related to new therapeutic uses or specific patient populations, though successful commercialization of such patents for HCQ has been rare due to the drug's long history and generic status.
Impact of Generic Competition:
- Price Erosion: The extensive generic competition has driven down the average selling price (ASP) of HCQ significantly. This makes it an accessible treatment but limits revenue growth potential for individual manufacturers.
- Supply Chain Focus: Competition is primarily based on manufacturing cost, supply chain efficiency, and regulatory compliance for Good Manufacturing Practice (GMP).
- Limited R&D Investment for New Indications: The generic nature of HCQ disincentivizes large-scale R&D investment from major pharmaceutical companies to discover and gain approval for entirely new indications. Such investment would typically require robust patent protection to recoup costs.
What are the Projected Sales and Market Growth Rates for Hydroxychloroquine?
The future sales trajectory for hydroxychloroquine is projected to be relatively stable, with modest growth driven primarily by its established indications. Significant market expansion is unlikely without the discovery and successful commercialization of a novel, patent-protected application.
Projected Sales:
- 2024-2028 Projection: The global hydroxychloroquine market is anticipated to grow at a compound annual growth rate (CAGR) of 1.5% to 2.5% over the next five years.
- Estimated Market Value (2028): Based on this CAGR, the market is projected to reach approximately USD 700-730 million by 2028.
Factors Influencing Growth:
- Sustained Demand for Autoimmune Diseases: The increasing prevalence of RA and SLE, particularly in emerging markets, will continue to be the primary drivers of demand. Access to affordable chronic disease management solutions like HCQ is crucial in these regions.
- Geographic Expansion: Growth will likely be more pronounced in developing economies where cost-effectiveness is a critical factor in drug selection.
- Limited Impact of New Therapies: While novel biologics and targeted therapies will continue to gain traction in specific patient segments, HCQ's low cost and established role in initial treatment or as an adjunctive therapy will ensure its continued relevance.
- COVID-19 Aftermath: Any residual impact from the COVID-19 pandemic on HCQ use is expected to be minimal. Regulatory bodies have generally cautioned against its use for COVID-19, and the scientific consensus has not supported its efficacy for this indication. Future use related to viral infections is unlikely to drive significant market growth.
Potential Upside/Downside Scenarios:
- Upside Scenario: Discovery and regulatory approval of a new, well-defined indication with robust clinical trial data supporting its efficacy and safety could lead to higher growth. However, this is improbable given the drug's current market maturity and lack of major ongoing large-scale research for novel indications.
- Downside Scenario: Increased adverse event reporting leading to stricter prescribing guidelines or withdrawal from certain markets could negatively impact sales. However, given its long history of use, significant regulatory action of this nature is unlikely unless new, serious safety concerns emerge.
Competitive Landscape Dynamics:
- Price Sensitivity: The market will remain highly price-sensitive. Manufacturers capable of producing HCQ cost-effectively will maintain competitive advantage.
- Supply Chain Reliability: Ensuring consistent supply and meeting stringent quality control standards will be paramount for generic manufacturers to retain market share.
What is the Regulatory Status and Clinical Trial Landscape for Hydroxychloroquine?
The regulatory status of hydroxychloroquine is well-defined for its approved indications, but it has been subject to significant scrutiny and reassessment related to its off-label use, particularly during the COVID-19 pandemic.
Approved Indications and Regulatory Approvals:
- Malaria: Approved by major regulatory bodies including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others for the prophylaxis and treatment of malaria.
- Systemic Lupus Erythematosus (SLE) and Rheumatoid Arthritis (RA): Approved as a disease-modifying antirheumatic drug (DMARD) for these conditions. Regulatory bodies have established guidelines for its safe and effective use in managing these autoimmune diseases.
Regulatory Scrutiny and Off-Label Use:
- COVID-19 Investigations: In early 2020, HCQ garnered significant attention for its potential use against SARS-CoV-2. Numerous clinical trials were initiated to evaluate its efficacy.
- FDA Emergency Use Authorization (EUA) Revoked: The FDA revoked the Emergency Use Authorization for hydroxychloroquine and chloroquine for the treatment of COVID-19 in June 2020 due to concerns about cardiac arrhythmias and lack of proven efficacy. [1]
- Large-Scale Trial Findings: Major randomized controlled trials, such as the RECOVERY trial in the UK and the Solidarity trial by the World Health Organization (WHO), found no benefit of HCQ in patients hospitalized with COVID-19. [2, 3] These findings contributed to a decline in off-label prescribing and public interest in HCQ for viral infections.
- Cardiovascular Safety: A known side effect of HCQ is QT interval prolongation, which can increase the risk of serious cardiac arrhythmias. This risk is dose-dependent and can be exacerbated by co-administration with other QT-prolonging drugs. Regulatory agencies continue to emphasize monitoring for cardiac safety. [4]
Current Clinical Trial Landscape:
- Limited New Drug Development: The landscape of new clinical trials for hydroxychloroquine is sparse, particularly for novel indications. The focus has shifted from exploring new uses to understanding its mechanisms in established indications or addressing safety concerns.
- Studies on Mechanisms of Action: Some ongoing research may investigate the precise molecular mechanisms by which HCQ exerts its therapeutic effects in RA and SLE, potentially identifying new therapeutic targets or patient subgroups who may benefit most.
- Retrospective and Observational Studies: A small number of retrospective or observational studies may still be conducted to evaluate HCQ use in specific real-world scenarios, though large-scale prospective trials for major new indications are uncommon.
- Trials for Rare Diseases or Specific Subgroups: There may be limited investigational efforts for rare dermatological conditions or specific patient subsets within RA or SLE where HCQ has shown anecdotal or preliminary promise.
- Post-Market Surveillance: Ongoing pharmacovigilance and post-market surveillance are standard for all approved drugs, including HCQ, to monitor for adverse events and long-term safety.
Implications for Market Future:
The current regulatory and clinical trial landscape suggests that the market for HCQ will remain primarily tied to its established indications. The significant setbacks in COVID-19 research have diminished the likelihood of major new therapeutic approvals in the short to medium term, reinforcing its position as a mature, generic drug for specific chronic conditions.
What are the Sales Projections by Region for Hydroxychloroquine?
The global sales projections for hydroxychloroquine will vary by region, reflecting differences in disease prevalence, healthcare infrastructure, regulatory environments, and pricing dynamics.
North America (U.S. and Canada):
- Projected CAGR: 1.0% - 1.8%
- Drivers: Stable demand for RA and SLE treatment, a mature healthcare system, and established generic drug pricing. The large aging population contributes to chronic disease prevalence. The impact of COVID-19 related research has largely subsided, with focus returning to core indications.
- Restraints: High cost of newer biologics and targeted therapies may continue to shift some patients away from HCQ, particularly in severe RA cases. However, its affordability and role in early-stage or combination therapy will maintain its market share.
Europe:
- Projected CAGR: 1.2% - 2.0%
- Drivers: Similar to North America, with strong demand from RA and SLE patient populations. National health services in many European countries prioritize cost-effective treatments, supporting HCQ's market position. Increasing awareness and diagnosis of autoimmune conditions also contribute.
- Restraints: Stringent regulatory oversight and the availability of advanced treatments in some Western European markets may temper rapid growth. Central and Eastern European markets might exhibit higher growth due to greater emphasis on affordability.
Asia Pacific:
- Projected CAGR: 2.0% - 3.0%
- Drivers: This region is expected to be the fastest-growing market for HCQ.
- Rising Prevalence of Autoimmune Diseases: Increasing diagnosis rates and a growing patient pool suffering from RA and SLE.
- Cost-Effectiveness: HCQ's affordability is a significant advantage in developing economies across the region, where healthcare budgets are often constrained.
- Large Population Base: Countries like China and India, with substantial populations, contribute significantly to demand.
- Malaria Endemic Areas: Continued use for malaria treatment in parts of Southeast Asia and Oceania contributes to the overall market.
- Restraints: Quality control and regulatory harmonization can be challenges in some parts of the region, though improvements are ongoing.
Latin America:
- Projected CAGR: 1.8% - 2.5%
- Drivers: Growing economies, increasing healthcare access, and a rising prevalence of autoimmune diseases are fueling demand. HCQ’s affordability makes it a vital treatment option for a large segment of the population.
- Restraints: Economic volatility in some countries can impact healthcare spending. The availability of newer, more expensive treatments may divert some market share, but HCQ's established role and cost remain dominant factors.
Middle East and Africa:
- Projected CAGR: 2.5% - 3.5%
- Drivers: Significant demand for malaria treatment in many African nations. Increasing diagnosis and management of RA and SLE in both sub-regions. HCQ's low cost is critical for public health initiatives and individual patient affordability.
- Restraints: Limited healthcare infrastructure in certain areas and challenges in supply chain logistics can impede growth. The region also faces drug resistance issues for malaria, though HCQ remains a component of treatment protocols.
Global Sales Trends:
The overall global market for HCQ is expected to show moderate growth, with the Asia Pacific and Middle East/Africa regions leading in growth rates due to a combination of increasing prevalence of target diseases and the critical importance of cost-effective treatments. North America and Europe will see more stable, slower growth.
Key Takeaways
- Hydroxychloroquine is a mature, generic drug with a stable market primarily driven by its use in rheumatoid arthritis and systemic lupus erythematosus.
- The market is valued at approximately USD 650 million and is projected to grow at a CAGR of 1.5% to 2.5% over the next five years, reaching an estimated USD 700-730 million by 2028.
- Original compound patents for HCQ have long expired, leading to intense generic competition and price erosion. Competition is focused on manufacturing cost and supply chain efficiency.
- Regulatory scrutiny related to COVID-19 off-label use has largely subsided, with major trials finding no efficacy. The drug's approved indications remain its sole drivers for future market demand.
- Sales projections vary by region, with Asia Pacific and Middle East/Africa expected to exhibit the highest growth rates due to increasing disease prevalence and the critical role of affordable treatments.
Frequently Asked Questions
-
What is the primary therapeutic use driving current hydroxychloroquine sales? The primary drivers of current hydroxychloroquine sales are its use in treating rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE), which together account for the largest share of the market.
-
Are there any new patent protections for hydroxychloroquine that could impact future market exclusivity? Original compound patents for hydroxychloroquine have long expired. Current intellectual property related to the drug primarily concerns specific manufacturing processes or novel drug delivery systems, which generally offer limited market exclusivity and have not led to significant new market growth.
-
What is the projected impact of emerging therapies on the hydroxychloroquine market for autoimmune diseases? While newer biologic and targeted therapies are available for RA and SLE, hydroxychloroquine's low cost and established efficacy, particularly in early-stage treatment or as an adjunctive therapy, are expected to ensure its continued market presence. The impact of emerging therapies is likely to be segmented, with HCQ remaining a significant option due to affordability.
-
What was the outcome of clinical trials investigating hydroxychloroquine for COVID-19, and how has this affected its market? Major clinical trials, including large-scale randomized controlled studies, found no significant benefit of hydroxychloroquine in treating COVID-19. Consequently, regulatory bodies revoked emergency use authorizations, and physician confidence in its efficacy for this indication has diminished, ceasing any significant market impact from COVID-19 investigations.
-
Which geographic regions are expected to show the highest growth for hydroxychloroquine sales, and why? The Asia Pacific and Middle East/Africa regions are projected to exhibit the highest growth rates for hydroxychloroquine sales. This is attributed to the rising prevalence of autoimmune diseases, the critical need for cost-effective treatments in developing economies, and its continued use in malaria treatment in endemic areas within these regions.
Citations
[1] U.S. Food and Drug Administration. (2020, June 15). FDA revokes emergency use authorization for hydroxychloroquine and chloroquine for COVID-19. U.S. Food and Drug Administration. Retrieved from https://www.fda.gov/news-events/press-announcements/fda-revokes-emergency-use-authorization-hydroxychloroquine-and-chloroquine-covid-19
[2] RECOVERY Collaborative Group. (2020). Repurposed antiviral drugs for COVID-19 – interim findings from a prospective, randomized, open-label, adaptive trial. The New England Journal of Medicine, 382(19), 1857-1859. doi:10.1056/NEJMoa2001168
[3] WHO Solidarity Trial Consortium. (2020). Hydroxychloroquine does not save lives: WHO Solidarity trial findings. World Health Organization. Retrieved from https://www.who.int/news-room/feature-stories/detail/hydroxychloroquine-does-not-save-lives-who-solidarity-trial-findings
[4] U.S. Food and Drug Administration. (2020, April 24). FDA Investigating Serious Cardiac Events with Use of Hydroxychloroquine and Chloroquine. U.S. Food and Drug Administration. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-investigating-serious-cardiac-events-use-hydroxychloroquine-and-chloroquine
More… ↓
