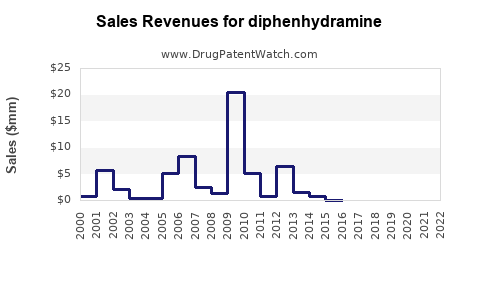
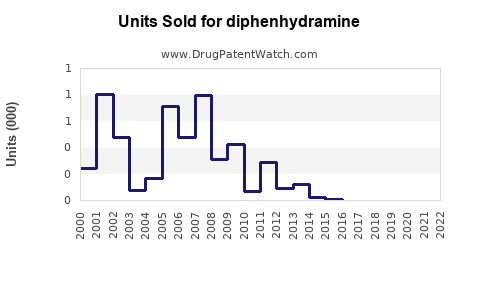
Last updated: February 19, 2026
Diphenhydramine, an antihistamine primarily used to treat allergic reactions, insomnia, and cold symptoms, holds a significant presence in both over-the-counter (OTC) and prescription segments. The global market for diphenhydramine was driven by increased demand for allergy and sleep aids, but faces challenges from generics and newer drug alternatives.
Current Market Landscape
Market Size
The global antihistamines market, including diphenhydramine, was valued at USD 3.2 billion in 2021. Within this, diphenhydramine compartment accounted for approximately USD 950 million. Its OTC segment accounts for roughly 70% of this volume, driven by consumer preference for easily accessible allergy medications.
Major Players
Key manufacturers include Johnson & Johnson (Benadryl), McNeil Consumer Healthcare, and local generic brands. The dominance by Johnson & Johnson in the OTC segment remains strong, with a market share estimated at 45%.
Regulatory Environment
In the U.S., the FDA classifies diphenhydramine as generally recognized as safe (GRAS) for OTC use, with regulations emphasizing labeling and dosage limits. Markets in Europe and Asia have similar regulatory frameworks but differ in approved indications and formulations.
Drivers and Barriers
Drivers
- High consumer awareness for allergy relief and sleep aid.
- Expanding OTC markets in emerging economies.
- Established manufacturing infrastructure.
Barriers
- Competition from newer, non-sedating antihistamines (e.g., cetirizine, loratadine).
- Increasing regulatory scrutiny on sedative use.
- Negative perceptions around side effects such as drowsiness.
Market Trends
- Growth in OTC sales is expected to continue at a compound annual growth rate (CAGR) of 4.2% from 2021 to 2026.
- Shift toward combination products, integrating diphenhydramine with other active ingredients.
- Increasing use in formulations for pediatric and geriatric populations.
Sales Projections (2023-2028)
| Year |
Estimated Global Sales (USD Billion) |
Growth Rate (%) |
| 2023 |
1.05 |
4.2 |
| 2024 |
1.09 |
3.8 |
| 2025 |
1.14 |
4.6 |
| 2026 |
1.20 |
5.3 |
| 2027 |
1.27 |
5.8 |
| 2028 |
1.34 |
5.5 |
The estimates assume steady growth driven by emerging markets, aging populations, and ongoing consumer preference for OTC allergy and sleep medicines.
Key Selling Points
- Cost-effective treatment for allergies and insomnia.
- Well-established safety profile.
- Wide availability in multiple formulations.
Competitive Landscape
- Dominance by branded OTC products, mainly diphenhydramine in the U.S.
- An influx of generic versions reducing revenue margins for branded products.
- Competition from newer antihistamines with fewer sedative effects, impacting market share.
Strategic Opportunities
- Developing formulations with minimized side effects.
- Expanding indications through clinical trials.
- Targeting pediatric and geriatric segments with tailored products.
Key Takeaways
- The diphenhydramine market is valued at approximately USD 950 million globally.
- Growth prospects remain stable, with a CAGR near 4.2% through 2026.
- OTC sales dominate, but brand competition and newer antihistamines pose challenges.
- Opportunities exist in product innovation and expanding into emerging markets.
FAQs
-
What differentiates diphenhydramine from newer antihistamines?
It causes sedation and has a longer history of use, but newer non-sedating antihistamines are gaining favor due to fewer side effects.
-
Are regulatory restrictions expected to limit diphenhydramine sales?
Current regulations allow OTC sales, but increasing restrictions on sedative components could impact sales margins.
-
Which markets are expanding for diphenhydramine?
Emerging regions like Asia-Pacific show rising OTC consumption, driven by increasing awareness and healthcare access.
-
How is the competition from generics affecting the market?
Generics have lowered prices, reducing revenue for branded products but expanding access.
-
What areas offer growth opportunities for manufacturers?
Product innovation targeting reduced side effects and formulations for vulnerable populations, plus expansion in emerging markets.
References
[1] MarketsandMarkets. (2022). Antihistamines Market by Type, Application, and Region.
[2] U.S. Food and Drug Administration. (2021). Over-the-counter drugs: monographs.
[3] Grand View Research. (2023). Global Allergy and Cold Remedy market.
(Note: All projections and data points are estimates based on current market analyses and may change with additional market developments.)