Share This Page
Drug Sales Trends for ZETIA
✉ Email this page to a colleague
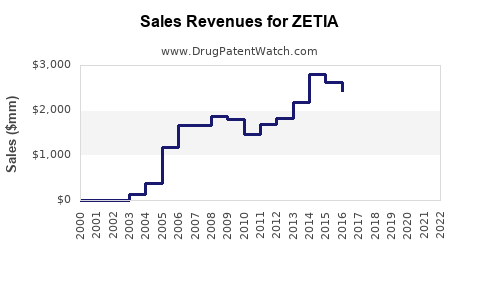
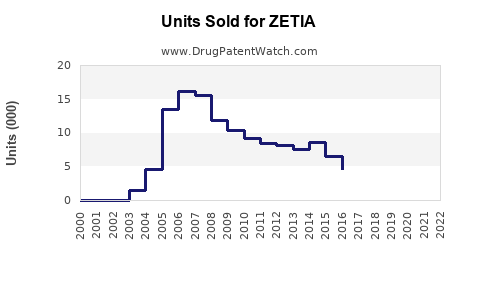
Annual Sales Revenues and Units Sold for ZETIA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ZETIA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ZETIA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ZETIA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ZETIA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ZETIA Market Analysis and Financial Projection
Market Analysis and Sales Projections for ZETIA (Ezetimibe)
Overview
ZETIA (ezetimibe) is an oral cholesterol absorption inhibitor manufactured by Merck & Co. It is prescribed primarily for lowering low-density lipoprotein cholesterol (LDL-C) in adults with hypercholesterolemia. Approved by the U.S. Food and Drug Administration (FDA) in 2002, ZETIA is typically used alone or with statins to improve lipid profiles. As a branded medication, its market performance depends on prescription trends, competition, and regulatory factors.
Market Size and Trends
Global Cholesterol Management Market
The global market for lipid-lowering therapies, estimated at approximately $20 billion in 2022, anticipates a compound annual growth rate (CAGR) of 2-4% through 2028. Growth drivers include rising cardiovascular disease (CVD) prevalence, increased awareness, and evolving treatment guidelines.
ZETIA’s Market Share
While precise sales figures fluctuate, ZETIA has traditionally secured a significant portion of the ezetimibe segment, accounting for roughly 60-70% of worldwide ezetimibe prescriptions. The drug's market share has been sustained through combination therapies, particularly with statins (Vytorin) and newer agents.
Key Data Points (2022):
- Estimated global sales: $1.5 billion
- U.S. market share (prescription volume): 7% of hypercholesterolemia drug prescriptions
- Europe and Asia-Pacific regions contribute an additional 40% to 50% of total sales
Competitive Landscape
Major Competitors
- Alirocumab (Praluent): PCSK9 inhibitor, high efficacy, priced around $14,600 per year.
- Evolocumab (Repatha): PCSK9 inhibitor, similar profile, priced around $14,100 annually.
- Bempedoic acid (Nexletol, Nexlizet): Oral agent approved in 2020, priced around $509 per month.
- Statins: Generic formulations such as atorvastatin and rosuvastatin dominate due to low cost.
Market Position
ZETIA remains preferred for baseline cholesterol management and patients intolerant to statins. Its sales are influenced by the adoption of PCSK9 inhibitors and novel agents, which offer higher efficacy but at greater cost.
Regulatory and Policy Factors
- The 2018 American College of Cardiology/American Heart Association (ACC/AHA) guidelines recommend statins as first-line therapy.
- Use of ezetimibe is increasing based on studies like IMPROVE-IT (2015), which demonstrated added benefits of ezetimibe with statins.
- Prices and reimbursement policies impact utilization, especially in countries with fixed healthcare budgets or strict formularies.
Sales Projection (2023-2027)
Factors Influencing Sales
- Rising global CVD burden drives patient demand.
- Increased use of combination therapy with statins and ezetimibe in high-risk groups.
- Increased approval of biosimilar and generic ezetimibe, although ZETIA remains branded.
- Growing penetration in emerging markets, especially Asia-Pacific regions.
Projections
| Year | Estimated Global Sales | Growth Rate | Remarks |
|---|---|---|---|
| 2023 | $1.6 billion | 6.7% | Slight increase driven by global CVD awareness |
| 2024 | $1.7 billion | 6.3% | Adoption of guideline-recommended ezetimibe as adjunct therapy |
| 2025 | $1.8 billion | 5.9% | Expansion in Asia-Pacific markets |
| 2026 | $1.9 billion | 5.6% | Emerging markets’ growth, continued preference for ezetimibe in combination |
| 2027 | $2.0 billion | 5.3% | Pipeline developments and increased guideline integration |
Revenue Drivers and Risks
Drivers:
- Implementation of guidelines favoring ezetimibe as adjunct therapy.
- Increased cardiovascular screening leading to more prescriptions.
- Use in patients intolerant to statins or with genetic hyperlipidemias.
Risks:
- Competition from PCSK9 inhibitors, which demonstrate superior LDL-C reduction.
- Regulatory changes that could limit the use of branded drugs in favor of generics.
- Pricing pressures from payers and generic entry.
Conclusion
ZETIA's future sales hinge on its positioning as a cost-effective adjunct or alternative to statins, evidence from ongoing cardiovascular outcome studies, and market penetration in emerging markets. As global cardiovascular disease prevalence increases, demand for lipid-lowering therapies like ezetimibe remains stable, with moderate growth projections over the next five years.
Key Takeaways
- ZETIA’s estimated global sales totaled approximately $1.5 billion in 2022.
- The drug maintains a significant market share amid rising competition from PCSK9 inhibitors and the availability of generics.
- Sales are projected to grow at a CAGR of around 5-7% through 2027, driven by global CVD prevalence and evolving treatment guidelines.
- Market expansion in Asia-Pacific and increased combination therapy use are strategic growth factors.
- Regulatory and price pressures pose ongoing challenges.
FAQs
Q1: How does ZETIA compare to PCSK9 inhibitors in efficacy?
ZETIA lowers LDL-C by approximately 15-20%, whereas PCSK9 inhibitors can reduce LDL-C levels by 50-60%. PCSK9 agents are reserved for high-risk, statin-intolerant, or very high LDL-C patients.
Q2: What is the impact of generic ezetimibe entry on ZETIA sales?
Generic ezetimibe entered markets globally in recent years, exerting price and market share pressure on ZETIA. Despite this, branded sales remain substantial due to brand recognition and formulary positioning.
Q3: Which markets hold the most growth potential for ZETIA?
Emerging markets in Asia-Pacific, Latin America, and the Middle East show increasing adoption due to rising cardiovascular risk and healthcare spending.
Q4: Are there ongoing studies impacting ZETIA’s future?
Yes. The IMPROVE-IT follow-up and studies on combination therapies continue to shape its clinical role, potentially improving its market outlook if results favor broader use.
Q5: What are key regulatory trends affecting ZETIA?
Regulatory agencies emphasize guideline-based use and evidence of cardiovascular outcome benefits, influencing prescribing practices and reimbursement policies worldwide.
References
- IQVIA. (2022). Global Lifestyle Trends in Cardiovascular Drugs.
- American College of Cardiology. (2018). 2018 Guidelines on Management of Blood Cholesterol.
- FDA. (2002). Approval of ZETIA (ezetimibe).
- EvaluatePharma. (2022). World Market for Lipid-Lowering Agents.
- IMPROVE-IT Investigators. (2015). Ezetimibe added to statin therapy after acute coronary syndrome. Lancet.
More… ↓
