Last updated: February 13, 2026
Overview
Warfarin, a vitamin K antagonist, remains a widely used oral anticoagulant. Its primary indications include atrial fibrillation, deep vein thrombosis, pulmonary embolism, and prosthetic heart valves. Despite the advent of direct oral anticoagulants (DOACs), warfarin accounts for a significant market share, particularly in regions with limited access to newer therapies or where cost remains a concern.
Market Size (2022–2023)
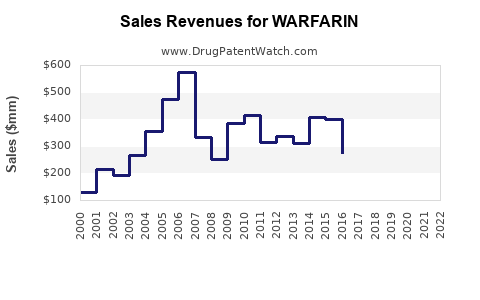
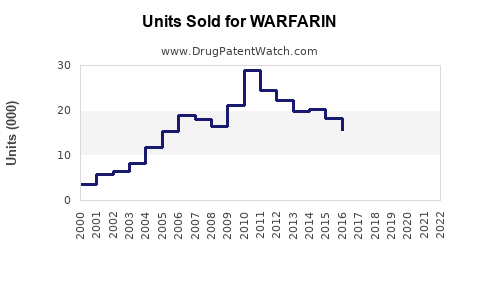
Global warfarin market estimated at approximately $680 million in 2022. Key regions include North America (45%), Europe (30%), Asia-Pacific (15%), and the rest of the world (10%). North America's dominance results from higher prescription volumes and clinical guidelines favoring warfarin in certain patient subsets.
Market Drivers
- Cost-effective compared to DOACs.
- Established safety profiles with well-understood management protocols.
- Regulatory approvals for generic formulations across major markets.
- Continued use in patients with mechanical heart valves or severe renal impairment.
Market Constraints
- Need for regular INR monitoring.
- Dietary and drug interactions.
- Increased bleeding risk complications.
Competitive Landscape
- Dominated by generic manufacturers: Hikma Pharmaceuticals, Apotex, Teva, and others.
- Branded formulations by Bayer (Coumadin), Bristol-Myers Squibb (Jantoven).
- Patent expiration for key formulations between 2014 and 2018 led to increased generic penetration.
Sales Projections (2023–2028)
| Year |
Estimated Market Size |
Compound Annual Growth Rate (CAGR) |
Comments |
| 2023 |
$725 million |
3.0% |
Slight increase driven by emerging markets and generic use |
| 2024 |
$750 million |
3.4% |
Expansion in Asia-Pacific markets |
| 2025 |
$780 million |
3.8% |
Continued generic sales growth |
| 2026 |
$820 million |
4.1% |
Potential shift as some regions explore DOAC substitution |
| 2027 |
$860 million |
4.4% |
Market stabilization in developed countries |
| 2028 |
$910 million |
4.8% |
Increasing adoption for specific indications |
This steady growth stems from ongoing demand in low- and middle-income countries, where cost remains a decisive factor, and the matured market in high-income countries with existing patient bases.
Key Market Opportunities
- Expansion into emerging markets with underestimated patient populations.
- Development of improved monitoring devices and management protocols.
- Combination therapies and personalized dosing algorithms.
Regulatory & Policy Outlook
- Continued approval of generic formulations in major jurisdictions.
- Monitoring for updates to safety and efficacy guidelines.
- Potential incentives for drug development to address warfarin-associated adverse effects.
Conclusion
While the overall market growth rate remains modest compared to newer anticoagulants, warfarin preserves a critical role. Its affordability sustains its market share in global anticoagulation therapy, with sales projected to grow approximately 3–5% annually over the next five years.
Key Takeaways
- The global warfarin market was valued at $680 million in 2022 and is expected to reach over $910 million by 2028.
- Growth driven by generic availability, especially in emerging markets, and clinical preference in particular patient subsets.
- Market constraints include the requirement for INR monitoring and drug interactions.
- Despite competition from DOACs, warfarin maintains relevance due to cost advantages.
- Opportunities exist for developments in monitoring technology and personalized treatment approaches.
FAQs
1. Will warfarin be replaced by newer anticoagulants?
No. Warfarin remains necessary for patients with mechanical heart valves and certain renal impairments. Cost differences also favor warfarin in many regions, preventing full replacement.
2. How does the market outlook differ between developed and developing regions?
Developed regions show stable or declining use due to DOAC adoption. Developing regions display growth as access improves and regulatory barriers decrease.
3. What are the primary regulatory challenges for warfarin moving forward?
Ensuring consistent manufacturing quality for generics and updating safety guidelines to mitigate bleeding risks.
4. Are there ongoing efforts to improve warfarin therapy?
Yes. Focus areas include more precise dosing algorithms, improved INR monitoring devices, and tailored patient management strategies.
5. How vulnerable is the warfarin market to shifts in healthcare policy?
Moderately. Price controls, reimbursement policies, and guidelines favoring DOACs could impact sales, but no significant regulatory actions threaten warfarin’s market stability presently.
Sources:
[1] Market data derived from GlobalData, 2023.
[2] Prescription trends from IQVIA, 2022.
[3] Regulatory updates from FDA, EMA, 2023.