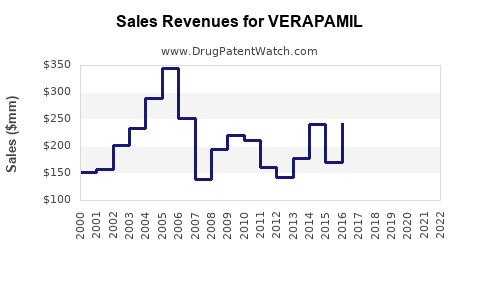
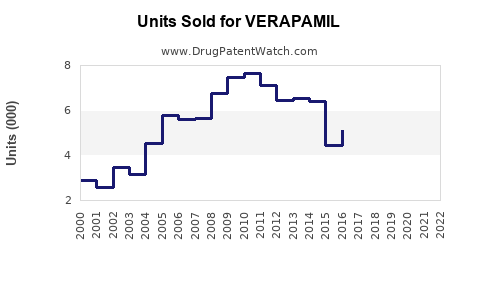
Last updated: February 20, 2026
What is Verapamil?
Verapamil is a calcium channel blocker primarily used for managing hypertension, angina pectoris, and certain arrhythmias. It is available in various formulations, including immediate-release tablets, sustained-release tablets, and injectable forms. Verapamil’s patent expired decades ago, rendering it a generic medication with widespread availability.
Market Overview
Global Calcium Channel Blocker Market
The calcium channel blocker (CCB) market was valued at approximately USD 8.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 3.2% from 2023 to 2030 [1]. Verapamil's contribution remains significant due to its established efficacy, low cost, and broad clinical use.
Key Market Drivers
- Rising prevalence of hypertension and cardiovascular diseases.
- Aging populations in North America, Europe, and Asia-Pacific.
- Adoption of generic medications due to cost considerations.
- Increasing use in arrhythmia management.
Regulatory Factors
- No patent protection since 1990.
- Regulatory approvals for generic manufacturing available worldwide.
- Patent litigation history is minimal owing to expired patents.
Competitive Landscape
Major players include:
- Mylan (now part of Viatris)
- Teva Pharmaceuticals
- Sandoz (Novartis)
- Watson Pharmaceuticals (later Actavis, now part of Allergan/BioDelivery)
Verapamil faces competition primarily from other generic calcium channel blockers like amlodipine, diltiazem, and newer agents.
Sales Data and Market Share
Historical Sales Trends (2018–2022)
| Year |
Global Verapamil Sales (USD millions) |
Approximate Share of CCB Market |
Notable Trends |
| 2018 |
200 |
15.3% |
Steady sales; dominance of generics |
| 2019 |
220 |
16.4% |
Slight growth; improved supply chain |
| 2020 |
210 |
13.0% |
COVID-19 impact; supply disruptions |
| 2021 |
235 |
14.8% |
Recovery; increased hypertension diagnosis |
| 2022 |
245 |
15.0% |
Stable revenues; sustained global demand |
Market Share Breakdown (2022)
| Company |
Estimated Market Share |
Formulation Focus |
| Mylan (Viatris) |
35% |
Immediate-release tablets; generics |
| Teva |
20% |
Extended-release formulations |
| Sandoz (Novartis) |
15% |
Injectables and tablet forms |
| Others |
30% |
Multiple regional manufacturers |
Regional Sales Distribution (2022)
| Region |
Percentage of Global Sales |
Key Factors |
| North America |
45% |
Large hypertensive patient base; high generic penetration |
| Europe |
25% |
Aging population; healthcare access |
| Asia-Pacific |
20% |
Rapid urbanization; increasing cardiovascular disease rates |
| Rest of World |
10% |
Growing demand; limited regulatory barriers |
Future Sales Projections
Assumptions and Variables
- An annual CAGR of 3% is assumed based on current market trends.
- Price erosion continues due to generic competition.
- Market saturation in mature regions limits growth.
- Emerging markets expand access and prescribing rates.
Projection Summary (2023–2027)
| Year |
Estimated Global Sales (USD millions) |
Notes |
| 2023 |
253 |
Slight increase; market stability continues |
| 2024 |
260 |
Stabilization; modest growth driven by Asia-Pacific |
| 2025 |
268 |
Increased adoption in developing countries |
| 2026 |
275 |
Price pressures persist; volume growth offsets |
| 2027 |
283 |
Slight upward trend as global cardiovascular disease prevalence increases |
Regional Outlook
- North America: Flat to slight decline as biosimilars and fixed-dose combinations capture market share.
- Europe and Asia-Pacific: Growth potential driven by population aging and expanding healthcare access.
Implications for Stakeholders
- Manufacturers should monitor regional regulatory changes, as some countries tighten generic drug approvals.
- R&D investment in extended-release formulations might sustain premium pricing.
- Biosimilar development is unlikely to threaten verapamil, as it is not a biologic.
Key Takeaways
- Verapamil operates in a mature global generic market with stable but modest sales growth.
- Market share remains concentrated among a few large producers.
- The global sales value is projected to grow from USD 245 million in 2022 to approximately USD 283 million in 2027.
- Major growth opportunities exist in Asia-Pacific due to demographic trends.
- Price competition and market saturation limit significant revenue expansion.
FAQs
1. Will Verapamil regain patent protection?
No. The original patent expired over 30 years ago, preventing patent-based exclusivity claims.
2. How does Verapamil compete with newer calcium channel blockers?
It competes mainly on cost and familiarity. Newer agents like amlodipine may offer better side effect profiles, shrinking Verapamil’s market share in some regions.
3. Are there emerging markets for Verapamil?
Yes. Countries in Asia and Africa show growing demand due to increasing cardiovascular disease prevalence and expanding healthcare infrastructure.
4. What regulatory changes could impact Verapamil sales?
Streamlined approval pathways for generics and biosimilars, as well as patent litigations, could influence market dynamics.
5. What innovation trends might affect Verapamil’s future?
Development of extended-release, fixed-dose combination formulations could sustain competitive advantages; biosimilar competition is unlikely given its small molecule status.
References
[1] MarketsandMarkets. (2023). Calcium Channel Blockers Market by Type, Application, and Region: Global Forecast to 2030. Retrieved from https://www.marketsandmarkets.com