Last updated: February 14, 2026
RETIN-A (tretinoin) holds a significant position within dermatology, primarily used for acne treatment and hyperpigmentation. Its market dynamics are affected by competition, regulatory status, patent expiry, and evolving medical practices.
Current Market Position of RETIN-A
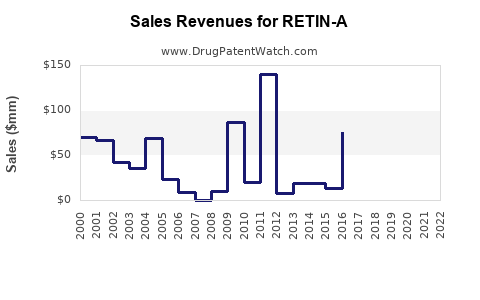
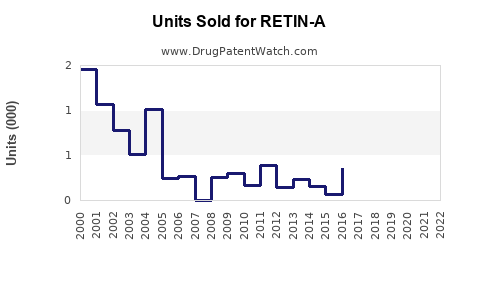
RETIN-A, approved by the FDA in 1971, remains a leading topical retinoid. Its extensive patent protection expired in the early 2000s, leading to increased generic availability. Collateral impacts include significant price decline and market share erosion for branded formulations.
- Estimated Global Market Size (2022): $1.2 billion
- Market Share (Branded RETIN-A): Approximately 25% of the total topical retinoids market.
- Generics' Share: Over 70%, with multiple manufacturers offering tretinoin formulations.
Key Drivers and Barriers
Drivers:
- Dermatology demand: Rising acne prevalence among adolescents and adults.
- Cosmetic use: Increased use for anti-aging and hyperpigmentation.
- Regulatory approvals: Continued approval for additional indications, expanding usage scope.
- Efficacy profile: Proven effectiveness with well-established safety profile.
Barriers:
- Generic competition: Pressure on pricing and margins.
- Side effect profile: Skin irritation limits patient adherence.
- Formulation limitations: New delivery systems (e.g., gels, creams with better tolerability) challenge traditional formulations.
- Market saturation: Mature product stage, slowing growth potential.
Sales Projections (2023–2027)
| Year |
Estimated Global Sales (USD billions) |
Growth Rate |
Comments |
| 2023 |
1.2 |
- |
Base year for projections |
| 2024 |
1.3 |
8.3% |
Ongoing acne management demand |
| 2025 |
1.4 |
7.7% |
Incremental growth from expanding use |
| 2026 |
1.5 |
7.1% |
Market penetration continues |
| 2027 |
1.6 |
6.7% |
Slight slowdown as market matures |
Assumptions:
- Steady growth of 6–8% annual driven by increased acne and anti-aging use.
- Market penetration of generics limits branded sales rebound but offsets volume increases.
- Novel formulations (e.g., improved tolerability) contribute to incremental gains.
Regional Outlook
| Region |
Market Share |
Growth Rate (2023–2027) |
Drivers |
| North America |
50% |
6.5% |
High acne prevalence, mature market |
| Europe |
25% |
7% |
Extended indications, aging population |
| Asia-Pacific |
15% |
10% |
Rising acne cases, increasing skincare awareness |
| Rest of World |
10% |
8% |
Expanding access to dermatological care |
Competitive Landscape and Future Trends
主要竞争者
- Generic manufacturers hold predominant market share.
- Branded players include Almirall (Epiduo), Rohto (Retin-A Micro), and other specialty dermatology firms.
- Increasing interest in combination therapies (e.g., tretinoin with antibiotics or bleaching agents).
未来发展趋势
- Launch of reformulated tretinoin products targeting improved tolerability.
- Expansion into new indications like photo-aging and skin cancers.
- Digital health initiatives for better patient adherence.
- Regulatory adaptations in emerging markets to broaden accessibility.
Implications for Stakeholders
- Pharmaceutical companies: Focus on developing formulation innovations to extend lifecycle and improve patient outcomes.
- Investors: Brand stability diminishes with patent expiry; opportunities arise in generics and over-the-counter markets.
- Healthcare providers: Personalizing tretinoin use based on tolerability and patient preferences can sustain long-term treatment adherence.
Key Takeaways
- RETIN-A faces intense generic competition, constraining branded sales growth.
- The global market is projected to grow at approximately 6–8% annually over the next five years.
- Emerging markets and anti-aging applications are key growth areas.
- Innovation in formulation and combination therapies will shape future market dynamics.
FAQs
1. How does patent expiry impact RETIN-A revenues?
Patent expiry in the early 2000s led to widespread generic manufacture, reducing prices and branded sales. The market now hinges on generics, with branded RETIN-A holding a niche.
2. What factors limit the growth of tretinoin?
Side effects such as skin irritation, formulation limitations, and market saturation limit significant growth. The emergence of newer formulations targeting tolerability also challenges traditional tretinoin products.
3. Which regions offer the greatest growth potential?
Asia-Pacific shows the highest projected growth, driven by rising skin disease awareness and increasing access to dermatologic therapies.
4. How are new formulations affecting the market?
Formulations improving tolerability or combining tretinoin with other agents can expand usage and patient adherence, creating incremental revenue opportunities.
5. What are the main opportunities for new entrants?
Innovation in delivery systems, combination products, and targeting unmet needs in emerging markets constitute the main opportunities.
Citations
- Market Research Future. Global Topical Retinoids Market Analysis, 2022-2027.
- IQVIA. Dermatology Market Reports, 2022.
- U.S. Food and Drug Administration. Retin-A (tretinoin) Approval and Indications.
- Company Financial Reports. (Almirall, Rohto)