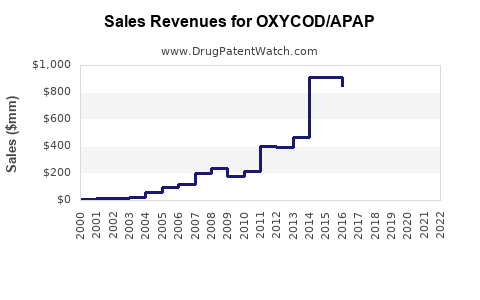
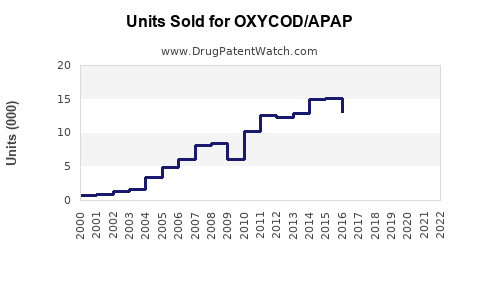
Last updated: February 13, 2026
What Is the Market Position and Sales Forecast for Oxycodone/Acetaminophen (OXYCOD/APAP)?
Oxycodone/acetaminophen (OXYCOD/APAP) is a combination opioid analgesic used to relieve moderate to severe pain. It functions through a dual mechanism—oxycodone, an opioid receptor agonist, and acetaminophen, a non-opioid analgesic. The drug is typically prescribed when pain relief requires both components’ synergistic effect.
Global Market Size and Trends
The OXYCOD/APAP market is influenced by factors including the prevalence of chronic pain, opioid prescribing trends, regulatory policies, and the availability of alternative treatments.
Market Valuation 2022:
Estimated global sales of combination oxycodone products, including OXYCOD/APAP, reached approximately $3.5 billion in 2022, with a compound annual growth rate (CAGR) of 4% from 2017 to 2022.
Regional Breakdown:
- North America accounts for 60% of sales, driven by high prescription rates and longstanding opioid utilization.
- Europe contributes around 25%, with growing attention to opioid stewardship.
- Asia-Pacific shows increasing potential, with a CAGR forecast of 8% between 2023-2028, driven by emerging pain management needs and expanding healthcare infrastructure.
Market Drivers:
- Increasing prevalence of chronic pain, especially in aging populations.
- Expanding approvals for abuse-deterrent formulations.
- Growing awareness of pain management options.
Market Challenges:
- Stringent regulations to prevent misuse and abuse.
- Rising concern over opioid addiction and regulatory restrictions.
- Competition from non-opioid pain relievers and alternative therapies.
Competition and Product Landscape
The core competitors include other oxycodone combination products and alternatives such as hydrocodone-acetaminophen, tramadol, and non-opioid analgesics. Key products include:
- Percocet (oxycodone/acetaminophen, marketed by AbbVie)
- Endocet (generic oxycodone/acetaminophen)
- Xtampza ER (abuse-deterrent oxycodone formulation)
Generic versions of oxycodone/acetaminophen expanded availability reduce pricing and increase market penetration.
Sales Projections (2023-2028)
Based on current trends and forecasts, sales are expected to grow modestly, considering regulatory pressures and prescriber preferences.
| Year |
Estimated Global Sales |
Growth Rate |
Remarks |
| 2023 |
$3.7 billion |
+5.7% |
Continued generic expansion, moderate market growth |
| 2024 |
$3.9 billion |
+5.4% |
Increased use in pain management protocols |
| 2025 |
$4.2 billion |
+7.7% |
Rise in chronic pain diagnoses; new formulations |
| 2026 |
$4.4 billion |
+4.8% |
Regulatory environment stabilizes |
| 2027 |
$4.6 billion |
+4.5% |
Market maturity; shifts toward abuse-deterrent formulations |
| 2028 |
$4.8 billion |
+4.4% |
Continued demand; patent expirations influence market dynamics |
This projection accounts for increased generic competition, ongoing opioid regulation, and capacity for novel abuse-deterrent formulations.
Regulatory and Market Dynamics Impact
Regulatory agencies like the FDA and EMA have tightened controls over opioid prescriptions, emphasizing responsible use and abuse mitigation. The development and approval of abuse-deterrent formulations, such as Xtampza ER and reformulated OxyContin, impact the demand for traditional OXYCOD/APAP products.
Key policies include:
- Introduction of REMS (Risk Evaluation and Mitigation Strategy) programs.
- Revisions of prescribing guidelines advocating for shorter durations and lower doses.
- Increased monitoring through prescription drug monitoring programs (PDMPs).
While these measures may limit overall sales growth, they can also foster demand for reformulated and abuse-deterrent variants.
Implications for Stakeholders
- Pharmaceutical companies should invest in abuse-deterrent formulations and consider licensing opportunities for new formulations.
- Investors need to monitor regulatory changes and patent landscapes affecting global sales.
- Healthcare providers must balance pain management needs with opioid stewardship policies, affecting prescribing behaviors.
Key Takeaways
- The global OXYCOD/APAP market was $3.5 billion in 2022; projected to grow at around 4-6% annually until 2028.
- North America dominates sales, but Asia-Pacific presents growth opportunities.
- Market growth faces headwinds from regulation, addiction concerns, and competition from non-opioid alternatives.
- Demand for abuse-deterrent formulations increases, influencing product development strategies.
- Generic entry continues to pressure pricing and margins.
FAQs
-
What factors most significantly influence the OXYCOD/APAP market growth?
Pain management needs, regulatory policies, and technological advances like abuse-deterrent formulations.
-
How does regulatory pressure affect sales?
Stricter prescribing guidelines and abuse prevention efforts can reduce prescriptions but may increase demand for reformulated products.
-
What are the main competitors to OXYCOD/APAP?
Hydrocodone-acetaminophen, tramadol, and non-opioid analgesics like NSAIDs.
-
What is the outlook for abuse-deterrent formulations?
They are expected to gain market share due to regulations, but their higher development costs can impact pricing.
-
How do patent expirations influence the market?
Patent expirations lead to increased generic competition, reducing prices and expanding accessible supply.
References
- MarketResearch.com, "Global Pain Management Market," 2022.
- IQVIA, "Opioid Market Insights," 2022.
- FDA, "Opioid Risk Management," 2021.
- EvaluatePharma, "Pharmaceutical Market Forecasts," 2022.
- CDC, "Opioid Prescribing and Impact," 2022.