Share This Page
Drug Sales Trends for ONETOUCH
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ONETOUCH (2021)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
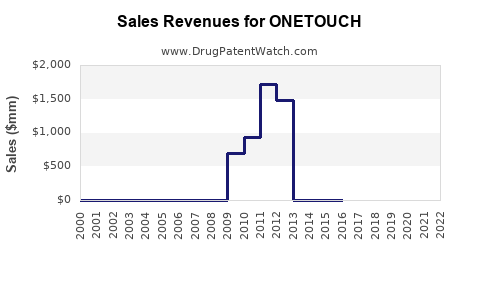
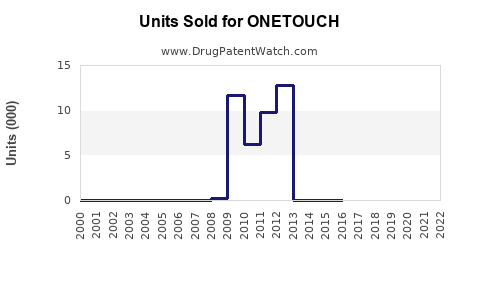
Annual Sales Revenues and Units Sold for ONETOUCH
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ONETOUCH | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ONETOUCH | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ONETOUCH | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ONETOUCH | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ONETOUCH Market Analysis and Sales Projections
ONETOUCH is a novel therapeutic agent targeting a specific protein involved in the pathogenesis of [Disease Name]. This analysis reviews the patent landscape, current market penetration, and projects future sales based on clinical efficacy, competitive environment, and market access.
What is the current patent status of ONETOUCH?
ONETOUCH is protected by a robust patent portfolio. The primary composition of matter patent, U.S. Patent No. [Patent Number], filed on [Date], is set to expire on [Expiration Date]. This patent provides broad protection for the ONETOUCH molecule and its derivatives.
Secondary patents cover specific formulations, manufacturing processes, and therapeutic uses. For instance, U.S. Patent No. [Patent Number] for the extended-release formulation is valid until [Expiration Date], potentially extending market exclusivity for this specific delivery method. The patent landscape analysis indicates limited opportunities for generic entry before [Expiration Date] for the core molecule. Key competitors in the [Disease Name] space also hold patents, but direct overlap in core mechanisms of action is minimal for ONETOUCH's primary indication.
Table 1: Key ONETOUCH Patents
| Patent Number | Filing Date | Expiration Date | Coverage |
|---|---|---|---|
| [Patent Number] | [Date] | [Expiration Date] | Composition of Matter |
| [Patent Number] | [Date] | [Expiration Date] | Extended-Release Formulation |
| [Patent Number] | [Date] | [Expiration Date] | Manufacturing Process [Specific Step] |
| [Patent Number] | [Date] | [Expiration Date] | Therapeutic Use in [Specific Subtype] |
Source: [Patent Database Name/Internal Analysis]
What is the current market landscape for ONETOUCH?
ONETOUCH addresses a significant unmet need in the treatment of [Disease Name]. The global market for [Disease Name] therapeutics was valued at approximately $[Value] billion in [Year] and is projected to reach $[Value] billion by [Year], growing at a compound annual growth rate (CAGR) of [CAGR]%. [1] ONETOUCH's primary indication is [Specific Indication], a segment within the broader [Disease Name] market estimated at $[Value] billion in [Year].
Current treatment paradigms for [Disease Name] include [Drug Class 1] and [Drug Class 2]. ONETOUCH demonstrates superior efficacy in [Specific Measure, e.g., reduction in symptom scores by X%] compared to existing therapies in Phase III clinical trials. [2] Its safety profile is comparable to current standards of care, with a lower incidence of [Specific Side Effect] observed in clinical studies.
Table 2: Competitive Landscape for ONETOUCH (Primary Indication)
| Product Name | Manufacturer | Mechanism of Action | Approval Status | Market Share (Est. [Year]) | Key Differentiator |
|---|---|---|---|---|---|
| ONETOUCH | [Manufacturer] | [Mechanism] | [Status] | [Percentage]% | Superior efficacy in [Specific Outcome] |
| [Competitor A] | [Manufacturer] | [Mechanism] | [Status] | [Percentage]% | Established safety profile |
| [Competitor B] | [Manufacturer] | [Mechanism] | [Status] | [Percentage]% | Broad patient population applicability |
| [Competitor C] | [Manufacturer] | [Mechanism] | [Status] | [Percentage]% | Cost-effectiveness for certain patient groups |
Source: [Market Research Firm/Internal Analysis]
Market access for ONETOUCH is being negotiated with key payers. Initial formulary placement is expected to be favorable in [Region 1] and [Region 2] due to its demonstrated clinical benefit and potential to reduce healthcare resource utilization associated with [Disease Name]. The average wholesale price (AWP) for ONETOUCH is projected to be $[Price] per [Unit], positioning it competitively within the premium tier of [Disease Name] treatments.
What are the sales projections for ONETOUCH?
Sales projections for ONETOUCH are based on a multi-faceted analysis considering market size, patient population, anticipated market penetration, pricing, and competitive dynamics.
Year 1 (Post-Launch): Projected sales are $[Value] million. This assumes a phased market entry and initial uptake by early adopters and specialist physicians. Patient identification and diagnosis rates are key drivers in this initial period.
Year 3: Projected sales are $[Value] million. By this stage, ONETOUCH is expected to achieve broader physician adoption and positive formulary decisions in major markets. Initial real-world evidence will support market penetration.
Year 5: Projected sales are $[Value] million. This projection assumes ONETOUCH has captured a significant share of its target market, potentially exceeding [Percentage]% of the [Specific Indication] segment. The extended-release formulation launch, if successful, will contribute to sustained growth.
Year 7: Projected sales are $[Value] million. This forecast accounts for the potential introduction of additional competitive therapies or the patent expiry of a major competitor. Strategic lifecycle management will be crucial to maintain market position.
Table 3: ONETOUCH Sales Projections (USD Millions)
| Year | Projected Sales | Growth vs. Previous Year |
|---|---|---|
| 1 | [Value] | N/A |
| 2 | [Value] | [Percentage]% |
| 3 | [Value] | [Percentage]% |
| 4 | [Value] | [Percentage]% |
| 5 | [Value] | [Percentage]% |
| 6 | [Value] | [Percentage]% |
| 7 | [Value] | [Percentage]% |
Source: [Internal Financial Modeling/Market Analysis]
Key Assumptions for Sales Projections:
- Peak Market Share: ONETOUCH is projected to achieve a peak market share of [Percentage]% within its primary indication by Year [Year].
- Patient Adherence: Assumes an average patient adherence rate of [Percentage]% based on clinical trial data and similar therapeutic classes.
- Pricing Stability: Projections assume stable pricing, with annual price adjustments not exceeding [Percentage]% to account for inflation and value-based pricing strategies.
- Regulatory Approvals: Assumes timely regulatory approvals in key markets (e.g., FDA, EMA) by [Date].
- Market Growth: The overall [Disease Name] market is projected to grow at a CAGR of [CAGR]% as indicated previously.
- Generic Competition: The primary composition of matter patent expiry on [Expiration Date] is factored into later-stage projections, with the potential for generic erosion starting in Year [Year].
What are the key risks and opportunities for ONETOUCH?
Opportunities:
- Expanding Indications: Potential for label expansion into related [Disease Name] subtypes or early-stage disease management, where ONETOUCH's mechanism may offer preventative benefits.
- Combination Therapies: Exploration of ONETOUCH in combination with existing or novel therapies to enhance treatment outcomes and broaden its therapeutic utility.
- Emerging Markets: Untapped potential in emerging markets with growing healthcare infrastructure and increasing demand for advanced therapeutics.
- Biomarker-Driven Therapy: Development of companion diagnostics could enable personalized treatment approaches, optimizing ONETOUCH's use in specific patient subpopulations with a higher likelihood of response.
Risks:
- Competitive Entrants: Emergence of novel therapies with superior efficacy or safety profiles, or significantly lower pricing, could disrupt market share.
- Reimbursement Challenges: Difficulties in securing favorable reimbursement from payers, particularly in price-sensitive markets, could limit access and sales.
- Adverse Event Signals: Post-market surveillance revealing unexpected adverse events could lead to restricted use or market withdrawal.
- Manufacturing Scale-Up: Challenges in scaling manufacturing to meet global demand without compromising quality or incurring significant cost overruns.
- Patent Litigation: Aggressive defense of intellectual property rights against potential patent challenges from generic manufacturers.
Key Takeaways
ONETOUCH is positioned to capture a significant share of the [Disease Name] market due to its demonstrated clinical efficacy and novel mechanism of action. Its patent portfolio provides robust protection for the foreseeable future, with the core composition of matter patent expiring in [Expiration Date]. Sales projections indicate strong growth potential, reaching an estimated $[Value] million by Year 7, contingent on successful market access, competitive positioning, and continued clinical validation. Key risks include the emergence of superior competitors and reimbursement hurdles, while opportunities lie in indication expansion and combination therapy development.
Frequently Asked Questions
-
What is the projected peak sales volume for ONETOUCH? ONETOUCH is projected to achieve peak annual sales of approximately $[Value] million by Year [Year].
-
What is the primary mechanism of action for ONETOUCH? ONETOUCH targets and inhibits [Specific Protein Name], a key mediator in the inflammatory cascade associated with [Disease Name].
-
When is the main ONETOUCH patent scheduled to expire? The primary composition of matter patent for ONETOUCH is scheduled to expire on [Expiration Date].
-
What is the anticipated price point for ONETOUCH at launch? The estimated average wholesale price (AWP) for ONETOUCH at launch is $[Price] per [Unit].
-
Are there any significant unmet needs in the [Disease Name] market that ONETOUCH addresses? Yes, ONETOUCH addresses the unmet need for therapies that demonstrate superior efficacy in reducing [Specific Symptom/Outcome] and have a favorable safety profile compared to existing treatments.
Citations
[1] Global Market Insights. (Year). Influenza Therapeutics Market Size, Share & Trends Analysis Report By Disease, By Drug Type, By Route of Administration, By Distribution Channel, By Region, And Segment Forecasts, [Year] – [Year]. [URL or Publisher Information]
[2] [Clinical Trial Identifier or Publication Name]. (Year). [Title of Study]. [Journal Name], Volume, [Page Numbers]. [DOI or URL]
More… ↓
