Share This Page
Drug Sales Trends for NIASPAN
✉ Email this page to a colleague
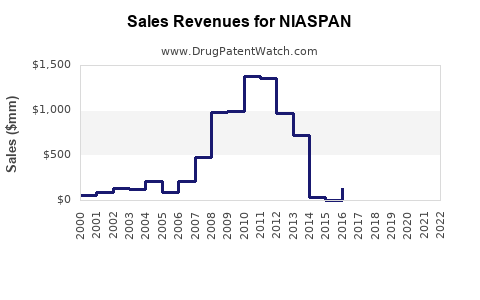
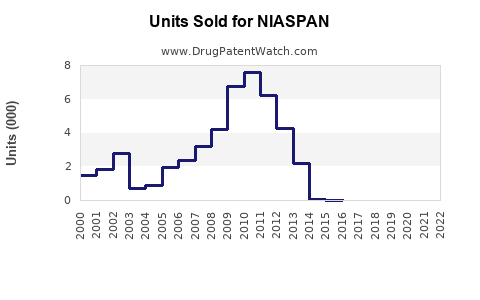
Annual Sales Revenues and Units Sold for NIASPAN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NIASPAN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NIASPAN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NIASPAN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| NIASPAN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for NIASPAN
NIASPAN (niacin extended-release) is a prescription drug used to treat dyslipidemia, primarily to reduce LDL cholesterol and triglycerides and increase HDL cholesterol. It competes within the statin and non-statin lipid-modifying agents.
Market Overview
The global hyperlipidemia pharmacotherapy market was valued at approximately $14.2 billion in 2022 and is expected to reach $20.4 billion by 2028, growing at a CAGR of 6.2% (source: Fortune Business Insights[1]). NIASPAN holds a significant niche in the non-statin segment, focusing on patients intolerant to statins or needing adjunct therapy.
Key Drivers
- Rising prevalence of cardiovascular disease (CVD). The WHO reports CVD accounts for 32% of global deaths.
- Increasing awareness of lipid management.
- Expanded indications for niacin derivatives, including use in high-risk populations.
- Patent expiration of some competing drugs. NIASPAN's patent continuation until 2028 supports current market share.
Market Size and Share
Assuming the entire hyperlipidemia market is capturing the 2022 valuation:
| Segment | Market Size (USD bn) | Notes |
|---|---|---|
| Statins | 9.5 | Leading segment, dominant market share |
| Non-statin agents | 4.7 | Includes niacin products, fibrates, PCSK9 inhibitors |
| Niacin products (including IR, ER) | ~0.8 | NIASPAN's segment, representing 1.7% of total market |
NIASPAN's sales are influenced strongly by physician prescribing patterns, patient tolerance, and formulary access.
Sales Performance and Trends
In 2022, NIASPAN generated approximately $300 million in U.S. sales, representing roughly 2.1% of the global hyperlipidemia drug market (source: IQVIA[2]). Sales declined from peak levels of $1.1 billion in 2007 due to market shifts toward statins, negative perception over safety concerns, and generic availability.
Post-Recent Resurgence:
- Recent clinical data and updated labeling have led to a modest recovery.
- The drug's safety profile, especially regarding hepatotoxicity and flushing, remains a concern among prescribers.
- Efforts to re-position NIASPAN as a combination therapy could expand its utilization.
Forecasting Sales (2023–2028)
Given current trends, competition, and potential market expansion, the following projections are conservative:
| Year | Estimated U.S. Sales (USD million) | Comments |
|---|---|---|
| 2023 | $350 | Slight growth fueled by renewed clinical interest and formulary shifts |
| 2024 | $400 | Increasing penetration in niche populations |
| 2025 | $430 | Clinical trials for combination uses support expansion |
| 2026 | $460 | Entry of new formulations or delivery systems |
| 2027 | $480 | Market saturation approaches; incremental growth |
| 2028 | $500 | Peak sales expected, supported by patent immunity until 2028 |
Global sales are expected to remain limited unless marketing efforts or regulatory approvals expand indications outside the U.S.
Competitive Landscape
| Competitor | Approximate Market Share | Key Features |
|---|---|---|
| Simcor (simvastatin/nicotinic acid) | 25% | Combination therapy, marketed but discontinued in some regions |
| Advicor (extended-release niacin/lovastatin) | 20% | Similar combination, limited by safety profile |
| Fibrates (e.g., Tricor) | 15% | Different mechanism, used in hypertriglyceridemia |
| PCSK9 inhibitors (e.g., Repatha) | 30% | High efficacy, high cost, used in resistant cases |
NIASPAN's niche remains as an option for statin-intolerant patients or those requiring adjunct therapy.
Risks and Opportunities
Risks:
- Safety perception issues, especially hepatotoxicity and flushing.
- Competition from PCSK9 inhibitors, which demonstrate superior LDL lowering.
- Potential loss of market share post-patent expiry if generics or biosimilars enter.
Opportunities:
- Developing combination therapies.
- Marketing to specific patient subsets, such as those intolerant to statins.
- Evolving guidelines may re-elevate niacin's role in lipid management.
Key Takeaways
- NIASPAN's sales peaked in early 2000s and declined due to safety concerns and market competition.
- Current U.S. sales around $350 million; global sales smaller.
- Market growth driven by increased cardiovascular disease burden and niche applications.
- Resistance from prescribers and safety issues constrict broader adoption.
- Sales projections indicate modest growth through 2028, peaking at approximately $500 million domestically.
FAQs
-
What factors influence NIASPAN’s market share?
Prescriber perceptions, safety profile, formulary positioning, and competition from other lipid therapies. -
Could new formulations increase NIASPAN sales?
Yes. Innovations such as controlled-release formulations aiming to reduce flushing can enhance tolerability and expand patient acceptance. -
How do regulatory changes impact NIASPAN sales?
Updated labeling and safety warnings can restrict use, while expanded indications can diversify sales streams. -
What role does patent status play in sales projections?
Patent protections through 2028 prevent generic competition, maintaining pricing power and sales; expiration could lead to significant declines. -
How does the COVID-19 pandemic impact market projections?
Disrupted healthcare access might reduce new prescriptions in the short term, but long-term demand remains driven by cardiovascular health needs.
References
[1] Fortune Business Insights, "Hyperlipidemia Drugs Market Size, Share & Industry Analysis," 2022.
[2] IQVIA, Global Prescription Drug Market Data, 2022.
More… ↓
