Share This Page
Drug Sales Trends for LOSARTAN/HCT
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for LOSARTAN/HCT (2021)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
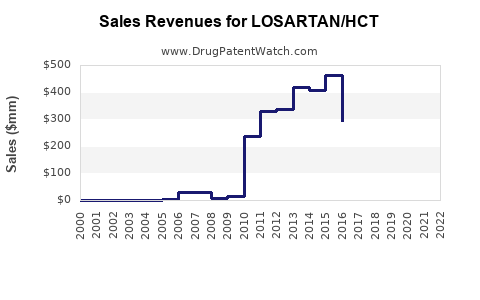
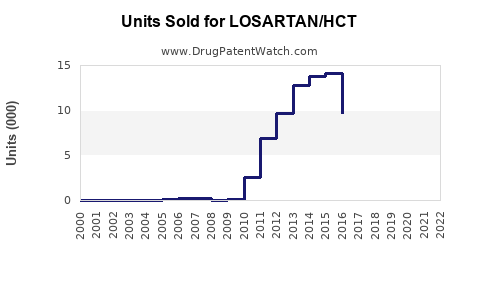
Annual Sales Revenues and Units Sold for LOSARTAN/HCT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LOSARTAN/HCT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LOSARTAN/HCT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LOSARTAN/HCT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LOSARTAN/HCT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| LOSARTAN/HCT | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| LOSARTAN/HCT | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| LOSARTAN/HCT | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Losartan/HCTZ Market Analysis and Sales Projections
Losartan potassium and hydrochlorothiazide (HCTZ) combination tablets are primarily prescribed for the treatment of hypertension. The market for these fixed-dose combination drugs is mature, driven by established efficacy, physician familiarity, and widespread availability. Generic competition significantly influences pricing and market share.
What is the Current Market Landscape for Losartan/HCTZ?
The global market for losartan/HCTZ is characterized by a substantial presence of generic manufacturers. The primary active pharmaceutical ingredients (APIs) are losartan potassium, an angiotensin II receptor blocker (ARB), and hydrochlorothiazide, a thiazide diuretic. This combination targets hypertension through complementary mechanisms of action.
Key market drivers include:
- High Prevalence of Hypertension: Global rates of hypertension continue to rise, creating a consistent demand for antihypertensive medications. The World Health Organization estimates that 1.28 billion adults aged 30-79 years have hypertension (1).
- Established Efficacy and Safety Profile: Losartan/HCTZ has a long track record of clinical use, demonstrating well-understood efficacy and a favorable safety profile when used appropriately.
- Cost-Effectiveness: The availability of multiple generic versions has made losartan/HCTZ a cost-effective treatment option, particularly for public health systems and patients with limited insurance coverage.
- Physician Familiarity: Healthcare providers are familiar with the prescribing guidelines, dosage ranges, and potential side effects of this combination, leading to its continued use.
Market challenges include:
- Intense Generic Competition: The presence of numerous generic manufacturers leads to aggressive price erosion, limiting revenue growth for individual companies.
- Therapeutic Advancements: The development of newer antihypertensive drug classes and novel combination therapies can potentially displace older, established treatments.
- Regulatory Scrutiny: Like all pharmaceuticals, losartan/HCTZ is subject to ongoing regulatory oversight, including potential recalls for impurities, as seen with angiotensin II receptor blockers (ARBs) in the past.
What are the Key Product Formulations and Dosages?
Losartan/HCTZ is available in various fixed-dose combinations, allowing for titration of both active ingredients to achieve optimal blood pressure control. Common tablet strengths include:
- Losartan Potassium 50 mg / Hydrochlorothiazide 12.5 mg
- Losartan Potassium 100 mg / Hydrochlorothiazide 12.5 mg
- Losartan Potassium 100 mg / Hydrochlorothiazide 25 mg
These formulations are typically administered orally once daily.
Who are the Major Market Players?
The market is populated by a mix of originator companies (though many patents have expired) and a large number of generic pharmaceutical manufacturers. Leading generic players and contract manufacturers involved in producing losartan/HCTZ include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris Inc.)
- Aurobindo Pharma Ltd.
- Lupin Ltd.
- Torrent Pharmaceuticals Ltd.
- Dr. Reddy's Laboratories Ltd.
- Cipla Ltd.
Specific market share varies by region and is highly fragmented due to generic penetration.
What are the Sales Projections for Losartan/HCTZ?
Given the mature nature of the losartan/HCTZ market and the significant impact of generic competition, substantial revenue growth is not anticipated. Instead, sales are expected to remain relatively stable, with modest fluctuations driven by factors such as population growth, healthcare access, and minor shifts in prescribing patterns.
Global sales for losartan/HCTZ, encompassing all generic and branded versions, were estimated to be in the range of USD 1.5 billion to USD 2.0 billion in recent years (estimated 2022-2023). This figure represents the aggregate revenue across all manufacturers and markets.
Projected Sales Trends (2024-2028):
- 2024-2025: Stable to slight decline (-1% to 0% CAGR). Price pressures from intense generic competition will continue to offset any marginal volume increases due to hypertension prevalence.
- 2026-2028: Continued stability, with a very low compound annual growth rate (CAGR) of approximately 0% to 1%. Market growth will largely mirror population increases and generic market dynamics.
These projections are based on the following assumptions:
- No Major Off-Patent Expirations: The primary patents for losartan and HCTZ have long expired, meaning no significant shifts in patent exclusivity will impact the market.
- Continued Generic Entry: The generic landscape will remain competitive, preventing significant price increases.
- Stable Regulatory Environment: No widespread, large-scale recalls or significant changes in prescribing guidelines that would drastically alter demand.
- Steady Hypertension Prevalence: Existing trends in hypertension incidence and management will persist.
Regional Sales Distribution (Estimated Percentage of Global Sales):
- North America (USA & Canada): 30-35%
- Europe: 25-30%
- Asia-Pacific: 20-25%
- Rest of World (Latin America, Middle East, Africa): 10-15%
The North American and European markets are characterized by higher per-capita spending on pharmaceuticals, though generic penetration is also very high. The Asia-Pacific market shows potential for volume growth due to increasing population and healthcare access.
What Factors Will Influence Future Sales?
Several factors will shape the future sales trajectory of losartan/HCTZ:
- Generic Pricing Dynamics: The ongoing price competition among generic manufacturers will be the primary determinant of overall market revenue. Bid/ask spreads in tender processes and direct-to-consumer advertising (where applicable) will play a role.
- Healthcare Policy and Reimbursement: Government healthcare policies, formulary decisions by insurance providers, and reimbursement rates can influence prescribing patterns and patient access to specific medications.
- Emergence of Novel Therapies: The introduction and adoption of newer, potentially more effective or convenient antihypertensive agents, including other fixed-dose combinations or novel drug classes, could gradually erode the market share of losartan/HCTZ.
- API Sourcing and Manufacturing Costs: Fluctuations in the cost of raw materials (APIs) and manufacturing processes can impact the profitability of generic manufacturers and, consequently, market pricing.
- Geopolitical and Economic Stability: Global economic conditions and geopolitical events can affect healthcare spending and pharmaceutical supply chains.
- Adherence and Patient Convenience: While losartan/HCTZ offers convenience as a fixed-dose combination, the development of single-pill combinations with newer agents or advanced drug delivery systems could offer advantages to some patient populations.
What is the Patent Landscape for Losartan/HCTZ?
The core patents for both losartan potassium and hydrochlorothiazide have long since expired, leading to the widespread availability of generic formulations. Losartan was first patented by DuPont Merck Pharmaceutical Company, with U.S. Patent 4,255,439 originally issued in 1981 for a different compound, and the key patents related to losartan itself were granted later and have expired. Hydrochlorothiazide's original patents also date back several decades.
Key Patent Expirations:
- Losartan Potassium: The primary composition of matter patents and formulation patents for losartan have expired. Exclusivity periods generally ended in the early to mid-2000s.
- Hydrochlorothiazide: Patents for HCTZ expired even earlier, placing it in the public domain for decades.
Current Patent Activity:
While the foundational patents have expired, new patent filings related to losartan/HCTZ combinations are typically focused on:
- Novel Formulations: Innovations in drug delivery, such as extended-release formulations, improved dissolution profiles, or combination with other active ingredients, may be patented.
- Manufacturing Processes: New or improved methods for synthesizing losartan potassium, hydrochlorothiazide, or the combined product, if they offer significant advantages in yield, purity, or cost, can be patented.
- New Uses or Indications: While unlikely for such a well-established drug, patents for new therapeutic uses or specific patient subpopulations could theoretically be sought, though clinical validation would be extensive.
Example of Patenting Strategy:
Companies might seek patents on specific polymorphic forms of losartan potassium or novel co-crystal formations that offer improved stability or bioavailability. Similarly, patents might cover specific tablet designs or manufacturing techniques that result in more efficient production or enhanced patient compliance.
However, the scope and enforceability of such secondary patents are often challenged in litigation, especially in the highly competitive generic market. The dominant market dynamic remains driven by the expiration of core patents.
How Does the Patent Landscape Impact Market Entry?
The expiration of core patents has democratized the market for losartan/HCTZ. This means:
- Ease of Generic Entry: Pharmaceutical companies can legally produce and market generic versions of losartan/HCTZ once the relevant patents have expired, provided they meet regulatory approval standards (e.g., bioequivalence).
- Increased Competition: The lack of patent exclusivity directly leads to a large number of manufacturers entering the market, intensifying competition.
- Price Degradation: The influx of generics drives down prices significantly, as companies compete on cost to gain market share.
- Focus on Cost Efficiency: Manufacturers in this space must focus on optimizing manufacturing processes, supply chain management, and economies of scale to remain profitable.
- Limited Return on Investment for Innovation: The high cost of drug development and the rapid genericization after patent expiry make it difficult for companies to recoup significant R&D investments in this specific drug class, shifting innovation focus to novel therapies.
What is the Competitive Environment for Losartan/HCTZ?
The competitive environment for losartan/HCTZ is defined by intense price competition and market fragmentation. With the expiration of primary patents, the market has become a battleground for generic manufacturers.
Key Competitive Factors:
- Price: This is the most critical competitive factor. Manufacturers vie for market share by offering the lowest prices, particularly in bulk tenders and institutional sales.
- Quality and Regulatory Compliance: While price is paramount, maintaining high quality standards and adherence to regulatory requirements (FDA, EMA, etc.) is non-negotiable. Product recalls due to impurity issues (e.g., nitrosamines in ARBs) can severely damage a company's reputation and market position.
- Supply Chain Reliability: Ensuring a consistent and reliable supply of finished dosage forms is crucial. Disruptions can lead to lost sales and a loss of customer trust.
- Manufacturing Efficiency: Companies with lower manufacturing costs due to optimized processes, economies of scale, and efficient sourcing of APIs have a distinct advantage.
- Distribution Networks: Established relationships with wholesalers, pharmacies, and healthcare systems are vital for market penetration.
- Product Portfolio Breadth: For larger generic companies, offering a wide range of antihypertensive products, including other ARBs, ACE inhibitors, calcium channel blockers, and diuretics, can create cross-selling opportunities and strengthen their overall market position.
How is Competition Structured?
The competition is structured as follows:
- Numerous Generic Manufacturers: A large number of companies, both large multinational generics firms and smaller regional players, produce losartan/HCTZ.
- Price-Driven Tenders: Many sales, especially to government health programs and large hospital networks, are conducted through competitive tender processes where price is the primary determinant.
- Wholesale and Retail Pharmacy Supply: Generic manufacturers supply their products to pharmaceutical wholesalers, who then distribute them to retail pharmacies. Pricing at the pharmacy level is heavily influenced by wholesale costs and pharmacy markups.
- Specialty and Mail-Order Pharmacies: These channels also procure generic losartan/HCTZ, often for price-sensitive patient populations.
The competitive landscape is dynamic, with companies constantly seeking ways to reduce costs and improve efficiency. Mergers and acquisitions among generic manufacturers can also reshape the competitive environment by consolidating market share.
What are the Regulatory Considerations for Losartan/HCTZ?
Regulatory considerations for losartan/HCTZ are critical for market access and continued sales. These encompass drug approval, manufacturing standards, and post-market surveillance.
Key Regulatory Aspects:
- Drug Approval Process: Manufacturers must obtain marketing authorization from regulatory bodies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other national health authorities. This involves demonstrating bioequivalence to the reference listed drug (RLD) for generic products, as well as proving manufacturing process controls and product quality.
- Good Manufacturing Practices (GMP): Facilities involved in the manufacturing of losartan/HCTZ must adhere to strict GMP guidelines. Regular inspections by regulatory agencies ensure compliance with these standards, covering aspects from facility design and personnel training to quality control and record-keeping.
- Impurity Control: This is a significant area of regulatory scrutiny, particularly for ARBs. The discovery of N-nitrosodimethylamine (NDMA) and other nitrosamine impurities in some ARBs, including losartan products, led to recalls and intensified monitoring by regulatory agencies. Manufacturers must implement robust analytical methods to detect and control impurities below acceptable limits.
- Labeling and Packaging: Regulatory bodies dictate the specific requirements for product labeling, including active ingredients, dosage, indications, contraindications, warnings, precautions, and adverse reactions. Packaging must also meet specific safety and integrity standards.
- Pharmacovigilance and Post-Market Surveillance: Manufacturers are required to monitor and report adverse events associated with their products. Regulatory agencies use this data to assess product safety in the real world and may initiate further actions, such as label changes or recalls, if necessary.
- Abbreviated New Drug Application (ANDA) and Similar Pathways: For generic versions, regulatory approval is typically sought through abbreviated pathways (e.g., ANDA in the US), which require demonstrating bioequivalence and acceptable manufacturing processes, rather than full clinical trials.
Recent Regulatory Events and Their Impact:
The most impactful regulatory events in recent years for ARBs, including losartan, have been related to nitrosamine impurity concerns.
- 2018-2019: Widespread recalls of losartan-containing products by multiple manufacturers occurred due to the presence of NDMA and N-nitrosodiethylamine (NDEA) impurities, often originating from specific API manufacturing processes.
- Impact: These recalls led to significant supply disruptions, loss of market trust for affected products, and a heightened focus by regulatory agencies on impurity profiling and control for all ARBs. Manufacturers were compelled to re-evaluate their API synthesis routes and implement stricter testing protocols.
- Ongoing Monitoring: Regulatory bodies continue to monitor nitrosamine levels in ARBs and have issued guidance documents and recommendations for manufacturers to ensure acceptable limits are maintained.
Manufacturers of losartan/HCTZ must maintain rigorous quality control systems and stay abreast of evolving regulatory guidelines to ensure uninterrupted market access and avoid costly recalls.
Key Takeaways
- The global losartan/HCTZ market is mature, driven by a high prevalence of hypertension and cost-effective generic options.
- Intense generic competition has resulted in price erosion and fragmented market share among numerous manufacturers.
- Projected sales for losartan/HCTZ are expected to remain stable, with minimal growth (0-1% CAGR) through 2028, largely offsetting any volume increases.
- North America and Europe represent the largest regional markets, though Asia-Pacific offers potential for volume growth.
- The patent landscape is characterized by expired core patents, allowing for broad generic entry and fostering a price-sensitive competitive environment.
- Regulatory compliance, particularly concerning impurity control (nitrosamines), is a critical factor for manufacturers, as demonstrated by past recalls.
FAQs
-
What is the primary market driver for losartan/HCTZ? The primary market driver is the high and increasing global prevalence of hypertension, coupled with the drug's established efficacy and cost-effectiveness as a generic treatment.
-
Will new patent filings significantly alter the losartan/HCTZ market? Significant alterations are unlikely. While secondary patents for novel formulations or manufacturing processes may emerge, the expiration of core patents means the market will remain largely open to generic competition, limiting the impact of such filings on overall market dynamics.
-
What is the most significant regulatory risk for losartan/HCTZ manufacturers? The most significant regulatory risk is the continued potential for nitrosamine impurity contamination, which can lead to mandatory product recalls, significant financial losses, and reputational damage, as observed in past events.
-
How does generic competition affect the sales revenue of losartan/HCTZ? Generic competition directly leads to price degradation, capping overall market revenue growth. While sales volume may slightly increase due to population growth or improved access, revenue is largely constrained by the downward pressure on prices.
-
What is the projected CAGR for the losartan/HCTZ market between 2024 and 2028? The projected compound annual growth rate (CAGR) for the losartan/HCTZ market between 2024 and 2028 is approximately 0% to 1%.
Citations
- World Health Organization. (2023, March 14). Hypertension. https://www.who.int/news-room/fact-sheets/detail/hypertension
More… ↓
